INTRODUCTION
Pain relief after surgery remains one of the most significant medical challenges,
and inadequate treatment may delay hospital discharge and patient recovery. In
addition, postoperative pain is acute and, when improperly conducted, may lead
to the development of chronic pain and greater use of analgesics, including
opioids, and their consequences1,2.
The infiltration of surgical incisions with local anesthetics (LA), mainly those
long-lasting, has been increasingly used in different types of surgeries, and
the results described in studies are encouraging1,2. The
effects of this application have been tested in several experimental
models3-7. However, the impact of LA infiltration on
surgical incision healing has not yet been fully established.
Aesthetic surgeries sometimes are significant, and consequently, there may be
severe pain in the postoperative period. The infiltration of LA could improve
this pain; however, it is necessary to assess whether this infiltration
influences healing, which could harm the final result.
OBJECTIVE
This study aimed to evaluate the interference of the infiltration of long-lasting
LAs in the surgical incision on the inflammatory infiltrate and on the fibrous
scar areas in rats.
METHODS
This study was carried out in a public tertiary hospital (Hospital de
Clínicas, Universidade Federal do Triângulo Mineiro, Uberaba,
Minas Gerais, Brazil) from January 2018 to January 2020 approved by
the committee on ethics in animal use under protocol number 314. All the animals
were treated following the recommendations of the institutional animal care
committee.
We followed the committee’s recommendations on ethics in animal use to
comply with the principle of 3R (reduction, refinement, and replacement). No
sample calculation was performed. Forty Wistar rats, 20 males and 20 females,
were divided into four groups of 10, with the same number of females and males
in each group. The animals did not present a difference in their weight, ranging
from 152 to 378 grams, with an average of 264 grams (±0.06).
The rats were placed in cages with controlled temperature (24±1°C)
and in cycles of 12 hours with light-12 hours in the dark. They were fed a
standard diet with water ad libitum for 12 hours before the
experimental protocol.
The surgical procedure was performed under general anesthesia, induced by
xylazine hydrochloride 2%, 5mg/kg, associated with ketamine, 70mg/kg,
administered intraperitoneally. When anesthesia was confirmed (loss of tail
reflex, paws and muscle relaxation), the dorsal area of the animal was
trichotomized, cleaned with povidone- iodine and dried with sterile gauze after
2 minutes. Two linear incisions of about 2cm each were made on the dorsal
region, symmetrical concerning the midline, reaching the subcutaneous tissue.
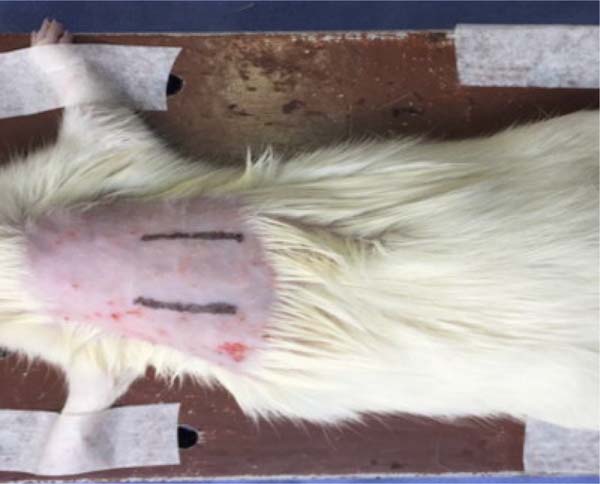
The left incision was infiltrated with doses of 1.8ml of LA (Figure 1), and the right incision did not
receive infiltration (control group). Another group received 1,8ml of 0.9%
saline solution (SS) application in the left incision and nothing in the right.
Thus, six application points (0.3ml per point) distributed along the 2cm
incision were made.
Figure 1 - Sutured surgical incisions, symmetrical in relation to midline, on
the dorsal area of the test animal.
Figure 1 - Sutured surgical incisions, symmetrical in relation to midline, on
the dorsal area of the test animal.
The groups were named according to the substance applied in the incision:
- Bupivacaine - infiltration of bupivacaine 0.25%;
- Levobupivacaine - infiltration of enantiomeric excess
levobupivacaine 0.25%;
- Ropivacaine - infiltration of ropivacaine 0.25%;
- Saline solution - infiltration of saline solution 0.9%.
There was no evaluation of anesthetic equipotency. Therefore, we chose to
standardize the volume and number of application points in the groups.
Two minutes after infiltration, the incisions were sutured using a 4.0 prolene
suture thread. There was an observation period of 2 hours after the application
of the local anesthetic. The test animals received an intramuscular tramadol
injection (2mg/kg) during the postoperative period every 12 hours for three
days. The surgical wound was treated once a day, and the animals were sacrificed
at the end of the seventh day.
Immediately after sacrifice, tissue samples from the two incisions with an
average diameter of 2cm2 were removed and fixed in 4% buffered
formaldehyde for approximately 4 hours. Posteriorly, they were cleaved, and
samples of the scar were processed and embedded in paraffin to make histological
sections of about 5 micrometers (µm) thick. Next,
histological sections were stained using the hematoxylin-eosin (HE) techniques
to evaluate the inflammatory infiltrate and the Masson’s trichrome (MT)
staining for the evaluation of the fibrous scar. The slides were analyzed under
a standard light microscope (OLYMPUS® BX40) by a single previously
trained observer, who was unaware of the group to which each animal belonged
(blind).
To perform the morphometric analysis of the area of the inflammatory infiltrate
(HE) and the fibrous scar (MT), we selected the areas with the largest
inflammatory infiltrate around the surgical scar (HE) and the focus with the
largest scar diameter (MT), respectively. To determine the area of the
inflammatory infiltrate and the scar, Image J® 1.32j semiautomatic image
analyzer system (National Institutes of Health, U.S.) was used, using
µm as reference unit and the 100X magnification
field of light microscope Olympus BX41 (Olympus®, São Paulo,
Brazil).
Biostat® program, version 5.0, was used for statistical analysis.
Comparisons among the different groups were made using a one-way ANOVA test with
Bonferroni post-test and Kruskal-Wallis test with Student-Newman-Keuls
post-test. The results were considered significant when the probability of
rejection of the null hypothesis was less than 5%
(p<0.05).
RESULTS
Morphologically evaluating, the inflammatory infiltrate was predominantly
mononuclear, permeated by occasional polymorphonuclear cells, especially
neutrophils. Table 1 shows the results of
the inflammatory infiltrate area in the different groups.
The inflammatory infiltrate area was significantly larger in the bupivacaine,
ropivacaine, and levobupivacaine groups compared with their controls (incisions
that did not receive any infiltration). When comparing the groups with each
other, the bupivacaine group had a significantly larger inflammatory infiltrate
than the levobupivacaine and ropivacaine groups. In addition, the SS 0.9% group
presented a substantially larger area of infiltrating than its control group.
However, no difference was observed between the SS 0.9% group and the LA groups
tested.
Figure 2 illustrates the inflammatory
infiltrate area in four different animals. Table 2 shows evaluation data of the fibrous scar area.
When evaluating the fibrous scar area, a significant difference was observed
between the levobupivacaine and ropivacaine groups and their controls. Moreover,
the fibrous scar area was larger when these two anesthetics were applied.
However, when comparing the LA groups with the SS 0.9% group, no difference was
found.
Figure 3 shows the fibrous scar in four
different animals.
DISCUSSION
LA infiltration into the skin and subcutaneous tissue is widely used for
analgesic purposes after surgical correction in different types of
surgery1,2. Long-lasting LAs, such as
bupivacaine, ropivacaine, and the enantiomeric excess levobupivacaine,
corresponds to 75% of the levorotatory isomer and 25% of the dextrorotatory
isomer are regularly used. Theoretically, the infiltration of these anesthetics
is the most rational method of analgesia since they block the nociceptive
afferents and, therefore, pain and secondary stress8-10.
There has been controversy over the interference of LA in the healing of surgical
wounds. The effects of injection or topical application of LA have been tested
in several experimental models, using healing time, tensile strength measures,
inflammatory process area, regeneration, granulation tissue, and
angiogenesis3-7. These studies used rodents
whose skin heals rapidly, less susceptible to inhibitory agents and systemic
factors that may limit healing3.
The healing of surgical wounds is when the tissue at the lesion site is replaced
by vascularized connective tissue. The first healing step is the onset of an
inflammatory reaction that will reabsorb extravasated blood and tissue
degradation products. Subsequently, there is a proliferation of capillaries and
fibroblasts that will form the cicatricial connective tissue. The final volume
of the scar depends on the stimuli that regulate the activity of the cells that
produce the extracellular matrix and on balance between the matrix synthesis and
degradation11. There
is evidence that the intensity and type of the inflammatory infiltrate will
define the extent of the scar12. In the present study, the evaluation was carried out seven
days after the incision. At this time, healing is probably in the
granulation/proliferation stage, which is characterized by fibroblast migration
and collagen production in rats13. However, even in this stage, significant inflammatory
infiltration around the scar was observed in all groups.
Table 1 - Inflammatory infiltrate area in the surgical wound after applying
local anesthetics and saline solution in the different animal
groups.
| Animal Group |
Median area (µm2)
|
Minimum area (µm2)
|
Maximum area (µm2)
|
| Bupivacaine |
1595646.75a |
353178.5 |
3785935.5 |
| Bupivacaine control |
10301715 |
181919 |
2865686.5 |
| Levobupivacaine |
975600b.e |
661763 |
1829388 |
| Levobupivacaine control |
762221.8 |
97105 |
971325 |
| Ropivacaine |
1278801.3c.f |
379043.5 |
4122721.5 |
| Ropivacaine control |
340497 |
340497 |
565769 |
| SS
0.9%
|
1440765.3d |
832288 |
1959811 |
| SS 0.9% control |
749901.3 |
74383 |
1054869 |
Table 1 - Inflammatory infiltrate area in the surgical wound after applying
local anesthetics and saline solution in the different animal
groups.
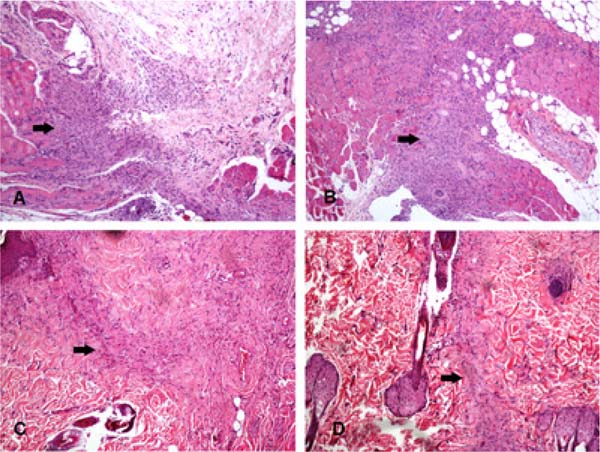
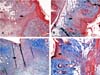
Figure 2 - Area of inflammatory infiltrate (arrows) in four different animals.
Note that in A (bupivacaine) and B (ropivacaine) the area is larger than
in C (levobupivacaine), and D (SS 0,9%) (hematoxylin-eosin, 100X). SS:
Saline Solution.
Figure 2 - Area of inflammatory infiltrate (arrows) in four different animals.
Note that in A (bupivacaine) and B (ropivacaine) the area is larger than
in C (levobupivacaine), and D (SS 0,9%) (hematoxylin-eosin, 100X). SS:
Saline Solution.
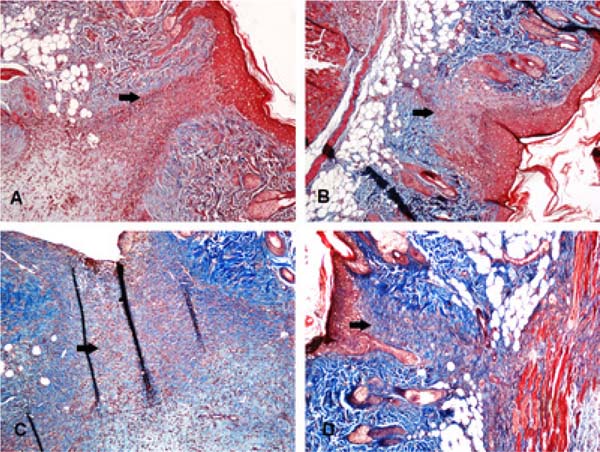
Figure 3 - Fibrous scar area stained in red (arrows) in four different animals.
Note that the area is larger in A (levobupivacaine) and B (ropivacaine)
than in C (SS 0,9%), and D (Bupivacaine) (Masson’s trichrome,
100X). SS: Saline Solution.
Figure 3 - Fibrous scar area stained in red (arrows) in four different animals.
Note that the area is larger in A (levobupivacaine) and B (ropivacaine)
than in C (SS 0,9%), and D (Bupivacaine) (Masson’s trichrome,
100X). SS: Saline Solution.
Table 2 - Fibrous scar area on the surgical wound after applying local
anesthetics and saline solution in the different groups of
animals.
| Animal Group |
Median area (µm2)
|
Minimum area (µm2)
|
Maximum area (µm2)
|
| Bupivacaine |
449402 |
146280 |
2016103.5 |
| Bupivacaine control |
605389.5 |
47813.5 |
1251420.5 |
| Levobupivacaine |
818835.5a.c |
346804 |
4741842 |
| Levobupivacaine control |
450434.0 |
116934 |
779886 |
| Ropivacaine |
885409b.d |
295817 |
2599696 |
| Ropivacaine control |
321682 |
207731 |
725589 |
| SS
0.9%
|
530607.8 |
235281.5 |
1997949 |
| SS 0.9% control |
501198.5 |
140366 |
1089956 |
Table 2 - Fibrous scar area on the surgical wound after applying local
anesthetics and saline solution in the different groups of
animals.
Levobupivacaine was associated with the smallest area of inflammatory infiltrate
and one of the largest areas of a fibrous scar. This observation contradicts the
conception that the intensity of the inflammatory infiltrate defines the extent
of scar formation12. It is
possible that the type of inflammatory infiltrate, rather than its intensity,
may be associated with a larger or smaller fibrous scar area, which was not
evaluated in the current study. The presence of inflammatory infiltrate
interferes directly in the scar formation; however, different types of
inflammatory cells may be present, and these cells may, or not, stimulate the
production of extracellular matrix. Another possibility would be that the volume
or trauma of the application, not the LA itself, would interfere with the
inflammatory infiltrate and the fibrous scar11, since there was no difference between the LA group and
the group that received SS, an apparently inert solution.
The application of long-lasting LA in the incision for postoperative analgesia
has been associated with reduced cytokine-induced alterations, and in addition,
it minimizes hyperalgesia14.
Although some studies suggest that these drugs also have anti-inflammatory
properties15, our
results demonstrate that bupivacaine and ropivacaine, particularly the latter,
are associated with larger inflammatory infiltrate. Furthermore, bupivacaine was
associated with an area of infiltrating significantly larger than its
levorotatory isomer, levobupivacaine.
A study using rats to evaluate inflammatory and cicatricial processes of the
wound after long-lasting LA infiltration, assessing histology and tensile
strength on the third and the fourteenth days, observed that, on the third day,
there was a significant increase of macrophages in the group receiving
bupivacaine. It was also observed that the collagen concentration was increased
in the animals infiltrated with bupivacaine compared with ropivacaine and SS.
There was no difference in the scar inflammatory response, presence of collagen,
and tensile strength on the fourteenth day. The authors concluded that the
alterations caused by LA infiltration do not extend beyond the third day, and
therefore, do not impair the wound healing process in rats5. Our results contradict this
supposition.
Another study on the effects of LA infiltration on the eighth day of wound
healing in rats demonstrated that both bupivacaine and lidocaine reduced
collagen production and resistance to scar rupture, causing significant edema,
vascularization, and inflammation when compared with the controls4. Similarly, in the present
study, we observed that bupivacaine was associated with the largest area of
inflammatory infiltrate. Concerning the fibrous scar, bupivacaine seems not to
interfere in the fibrous scar. On the other hand, its levorotatory isomer,
levobupivacaine, and ropivacaine were associated with larger fibrous scar
areas.
A study evaluating the effect of lidocaine and bupivacaine on wound healing in
rats suggested that although these anesthetics influenced local inflammation and
proteolytic factors, no effect on wound healing was observed16. These findings partially
agree with ours.
Another study, using a methodology similar to ours, compared the amount of
collagen and the number of mast cells through morphometry in rats after
injection of lidocaine with epinephrine or with buffer. The authors concluded
that lidocaine interferes with collagen and reduces the initial amount of mast
cells in the surgical wound17.
Two experimental studies in rats associating levobupivacaine infiltration with
ibuprofen and norepinephrine showed greater regeneration of the dermis and
epidermis, granulation tissue and angiogenesis than in the control group,
suggesting an increase in the regenerative/healing process6 and increased angiogenesis and
tensile strength of the scar7.
However, unlike our study, levobupivacaine was associated with a non-hormonal
anti-inflammatory and a vasopressor, which could interfere with healing.
In rabbits with infiltration of lidocaine and bupivacaine, other authors found no
histopathological difference when these LAs were compared with saline solution
and suggested that these LAs do not affect healing18.
LAs act by directly inhibiting the nociceptive fibers of the skin. These fibers,
together with the melanocytes, neuropeptides, and interleukins, are part of the
cutaneous neuroendocrine system that, among other functions, modulate surgical
healing, especially inflammation. LAs block neural impulses temporarily by
inhibiting the neuroendocrine response to wound healing stimuli and related
signaling, interfering negatively in wound healing. One of the neuropeptides
that plays an essential role in the neuroendocrine system is the substance P,
which controls mast cells’ degranulation and the release of inflammatory
proteins in the surgical site. These cells act on wound healing by promoting
inflammatory response, angiogenesis, and resorption of the extracellular matrix.
In addition, they regulate growth factors and interleukins, essential for
inflammation and the proliferative phase of wound healing19,20.
Amongst the different wound healing proteins, collagen correlates more closely
with scar tissue strength. The amount of collagen in the scar site depends on
the cicatricial process and may undergo LA interference due to inhibition during
the neurogenic inflammatory phase21.
How SS 0,9% infiltration was associated with a larger area of inflammatory
infiltrate and fibrous scar, we question whether, in addition to the action of
LA or the SS 0.9% in the scar site, the mechanical effect (distension) caused by
the infiltration interferes with the wound healing, possibility already
described11.
When comparing bupivacaine with its isomer, levobupivacaine, the former was
associated with the largest infiltrate area, and the last was associated with
the largest fibrous scar areas and smallest area of infiltrate. Thus, this
modification in the bupivacaine molecule could be responsible for the lower
inflammation, and larger fibrous scar observed in the wound infiltrated with
levobupivacaine.
CONCLUSION
The present study results indicate that the volume applied, or the infiltration
trauma may have been responsible for the larger area de infiltrate and fibrous
scar, not the LA itself. Complementary studies, including studies in humans,
evaluating the equipotency between the LA and variation in the volume applied,
are necessary to better understand the associated mechanisms, particularly in
the evaluation of the area of inflammatory infiltrate and fibrosis in different
stages (days) of healing, the type of inflammatory infiltrate and the cytokines
involved.
REFERENCES
1. Lee NH, Ryu K, Song T. Postoperative analgesic efficacy of
continuous wound infusion with local anesthetics after laparoscopy (PAIN): a
randomized, double-blind, placebo-controlled trial. Surg Endosc. 2020
Fev;35(2):562-8.
2. Velanovich V, Rider P, Deck K, Minkowitz HS, Leiman D, Jones N, et
al. Safety and efficacy of bupivacaine HCL collagen-matrix implant (INL-001) in
open inguinal hernia repair: results from two randomized controlled trials. Adv
Ther. 2019 Jan;36(1):200-16.
3. Brower MC, Johnson ME. Adverse effects of local anesthetic
infiltration on wound healing. Reg Anesth Pain Med. 2003
Mai/Jun;28(3):233-40.
4. Hancı V, Hakimoğlu S, Özaçmak H, Bektas
S, Özaçmak HS, Özadamar SO, et al. Comparison of the
effects of bupivacaine, lidocaine, and tramadol infiltration on wound healing in
rats. Rev Bras Anestesiol. 2012;62(6):799-810.
5. Abrão J, Fernandes CR, White PF, Shimano AC, okubo R, Lima
BP, et al. Effect of local anaesthetic infiltration with bupivacaine and
ropivacaine on wound healing: a placebo-controlled study. Int Wound J. 2014
Ago;11(4):379-85.
6. Zongwen G, Feng C, Xuemei C, Wang D, Li X, Li T. Local infiltration
of the surgical wounds with levobupivacaine, dexibuprofen, and norepinephrine to
reduce postoperative pain: a randomized, vehicle- controlled, and preclinical
study. Biomed Pharmocother. 2017 Ago;92:459-67.
7. Korat PS, Kapupara PP. Local infiltration of the surgical wound with
levobupivacaine, ibuprofen, and epinephrine in postoperative pain: an
experimental study. Biomed Pharmacother. 2017 Dez;96:104-11.
8. Sakellaris G, Petrakis I, Makatounaki K, Arbiros I, Karkavitsas N,
Charissis G. Effects of ropivacaine infiltration on cortisol and prolactin
responses to postoperative pain after inguinal hernioraphy in children. J
Pediatr Surg. 2004 Set;39(9):1400-3.
9. Paladini G, Di Carlo S, Musella G, Petrucci E, Scimia P, Ambrosoli
A, et al. Continuous wound infiltration of local anesthetics in postoperative
pain management: safety, efficacy and current perspectives. J Pain Res.
2020;31:285-94.
10. Joshi GP, Machi A. Surgical site infiltration: a neuroanatomical
approach. Best Pract Res Clin Anaesthesiol. 2019
Set;33(3):317-24.
11. Pereira FEL. Reparo de lesões. In: Brasileiro Filho G, ed.
Bogliolo patologia. 9ª ed. Rio de Janeiro: Guanabara Koogan; 2018. p.
181-93.
12. Werner S, Grose R. Regulation of wound healing by growth factors and
cytokines. Physiol Rev. 2003 Jul;83(3):835-70.
13. Vidmar J, Chingwaru C, Chingwaru W. Mammalian cell models to advance
our understanding of wound healing: a review. J Surg Res. 2017
Abr;210:269-80.
14. Gordon SM, Brahim JS, Dubner R, McCullagh LM, Sang C, Dionne RA.
Attenuation of pain in a randomized trial by suppression of peripheral
nociceptive activity in the immediate postoperative period. Anesth Analg. 2002
Nov;95(5):1351-7.
15. Cassuto J, Sinclair R, Bonderovic M. Anti-inflammatory properties of
local anesthetics and their present and potential clinical implications. Acta
Anaesthesiol Scand. 2006 Mar;50(3):265-82.
16. Waite A, Gilliver SC, Masterson GR, Hardman MJ, Ashcroft GS.
Clinically relevant doses of lidocaine and bupivacaine do not impair cutaneous
wound healing in mice. Br J Anaesth. 2010 Jun;104(6):768-73.
17. Rodrigues FV, Hochman B, Wood VT, Simões MJ, Juliano Y.
Effects of lidocaine with epinephrine or with buffer on wound healing in rat
skin. Wound Repair Regen. 2011;19(2):223-8.
18. Vasseur PB, Paul HA, Dybdal N, Crumley L. Effects of local
anesthetics on healing of abdominal wounds in rabbits. Am J Vet Res. 1984
Nov;45(11):2385-8.
19. Robson MC. The role of growth factors in the healing of chronic
wounds. Wound Repair Regen. 1997 Jan/Mar;5(1):12-7.
20. Myllyharju J, Kivirikko KI. Collagens and collagen-related diseases.
Ann Med. 2001 Fev;33(1):7-21.
21. Steinhoff M, Ständer S, Seeliger S, Ansel JC, Shmelz M, Luger
T. Modern aspects of cutaneous neurogenic inflammation. Arch Dermatol. 2003
Nov;139(11):1479-88.
1. Federal University of Triângulo Mineiro,
Uberaba, MG, Brazil.
2. University of Uberaba, Uberaba, MG,
Brazil.
Corresponding author: Renata Margarida
Etchebehere, RuaGetúlio Guaritá 140, Bairro Abadia,
Uberaba, MG, Brasil, Zip Code 38025-440, E-mail:
renata.etchebehere@uftm.edu.br
Article received: February 17, 2021.
Article accepted: April 19, 2021.
Conflicts of interest: none.
Institution: Hospital de Clínicas, Federal University of the
Triângulo Mineiro, Uberaba, MG, Brasil..