INTRODUCTION
Following the introduction of radical mastectomy by Halsted1, breast cancer surgery underwent a substantial evolution
during the 20th century as a result of the incorporation of new
techniques, new technologies, and the biomolecular analysis of tumors. Until the
1970s, the gold standard surgical treatment was radical mastectomy, which was
considered a great success at the time.
However, the fear of mutilation and loss of quality of life felt by many women
has motivated the search for less aggressive techniques for the locoregional
control of the disease. Thus, interventions such as modified radical mastectomy,
simple mastectomy, and conservative breast surgery have been included in the
oncological surgery repertoire, with comparable results in terms of disease-free
survival2 while offering the patients
an improved perception of quality of life and body image3.
Advances in breast reconstruction surgery have occurred parallel to the evolution
of ablative treatment of mastectomy patients. Surgery with autologous flaps
advanced greatly with the use of microsurgery and the description of
angiosomes4 and perforator-based
flaps, which became excellent options for reconstruction, causing little or no
damage to the donor area. The advent and implementation of novel materials
developed in recent years, such as anatomical implants, expanders, and acellular
dermal matrices, has also contributed to the success of breast reconstruction
procedures.
Although breast reconstruction has obvious and significant benefits for the
quality of life of mastectomy patients5,
more than 60% of these women do not undergo the procedure6. The decision to have breast reconstruction involves a
number of variables and may be associated with sociodemographic and ethnic
factors or even medical conditions. Since 2013, a law in Brazil states that any
woman undergoing mastectomy within the Unified Health System (SUS) should be
guaranteed immediate reconstruction, in the context of favorable medical
circumstances7.
Mastectomy with immediate breast reconstruction may prevent patients from
experiencing a period of psychosocial stress, negative body image, and sexual
dissatisfaction, as compared to late reconstruction8. Immediate breast reconstruction is typically performed
using implants or expanders, especially in light of the increasing popularity of
skin-sparing mastectomy, nipple-areola-complex (NAC)-sparing mastectomy, and
even prophylactic mastectomy. It is estimated that reconstruction with implants
will increase by an average of 5% per year9, owing to the acceptable rate of complications and proven
oncological safety of this technique10.
The use of acellular dermal matrices has also contributed to the increased number
of immediate implant-based reconstructions, as it provides better implant
coverage, expansion of the submuscular pocket11, and reduced rates of capsular contracture12. However, most published studies have reported on the
use of matrices of human origin. Since Brazilian laws do not permit the use of
this type of product and the cost of dermal matrices of animal origin is still
high in Brazil, new materials have been implemented with the aim of obtaining
the obvious cosmetic benefits of these matrices.
The surgical use of synthetic meshes has been widely studied, and they have been
shown to be safe, biocompatible, and hypoallergenic, with a low rate of
complications13. Therefore, these
materials may be effective replacements for dermal matrices in implant-based
breast reconstruction surgery.
OBJECTIVE
This study aimed to report the author’s experience using a technique of
implant-based breast reconstruction with synthetic mesh as an alternative to
acellular dermal matrices.
METHOD
Patient selection
This was a retrospective analysis of 12 consecutive patients (20
reconstructed breasts) who underwent immediate or delayed breast
reconstruction using the described technique with implants and synthetic
mesh between November 2015 and December 2016.
All patients were operated by the author at the private clinic and at the
breast center of the Integrated Oncology Center (Centro de Oncologia
Integrado - COI) in Rio de Janeiro, RJ.
Decisions regarding patient selection, mastectomy technique indication,
incision location, and possibility of immediate breast reconstruction with
implants were made in consultation with the mastology team.
All patients underwent preoperative evaluation with detailed anamnesis,
physical examination, laboratory testing, and X-rays. The following
demographic data were obtained: age, history of the current disease, history
of comorbidities, smoking habits, and previous neoadjuvant chemotherapy or
radiotherapy. Preoperative photographic documentation was also included.
Physical examination included breast palpation and the measurement of breast
width, height, and projection. Assessment of the contralateral breast (in
cases of unilateral reconstruction), asymmetries, ptosis, thorax shape, and
patient’s biotype was also performed. Together with the mastectomy
technique, these data were essential in the estimation of the implant
volume. All patients underwent breast reconstruction with textured
anatomical implants Mentor® (Santa Barbara, CA) and
semi-absorbable mesh ULTRAPRO® (Ethicon, a Johnson & Johnson
company, Amersfoort, The Netherlands).
Surgical technique
The location and type of incision are discussed with the mastology team,
taking into account the tumor location, breast shape, ptosis, and patient’s
expectations.
At the end of the mastectomy (skin-sparing, areola-sparing, or prophylactic),
in cases of immediate reconstruction, the skin flap is assessed, as well as
the patient’s oncological status. The flap is assessed clinically (color,
capillary filling, and thickness). If there are signs of vascular injury,
immediate reconstruction with implants is aborted and two-stage
reconstruction with an expander is performed. In cases of the node-positive
axilla, an indication for intraoperative adjuvant radiotherapy is another
essential factor in determining the choice of technique. Due to the high
incidence of capsular contracture in patients with implants who undergo
radiotherapy, tissue expander-based reconstruction is another option.
Reconstruction begins with the detachment of the pectoralis major through its
free margin and from the sternum and inframammary fold, without sectioning
it and maintaining its attachment to the fascia. The size of the subpectoral
pocket is calculated based on the desired volume of the implant and the
patient’s anatomy. A mold with the desired volume of the implant is then
inserted in the partial submuscular pocket to gauge the size of the fascia
of the serratus anterior muscle that will be lifted to accommodate the
lateral portion of the implant, where the mesh will be sutured.
After the dissection of the fascia of the serratus anterior muscle, the
patient is seated on the operating table, the mold is once again inserted,
and the incision is closed temporarily with a skin stapler. At this point,
symmetry, contour, and position of the inframammary fold are assessed and
the ideal implant is selected. The submuscular pocket is then irrigated with
antibiotic solution (cefazolin + garamycin), and a vacuum suction drain and
the implant are inserted.
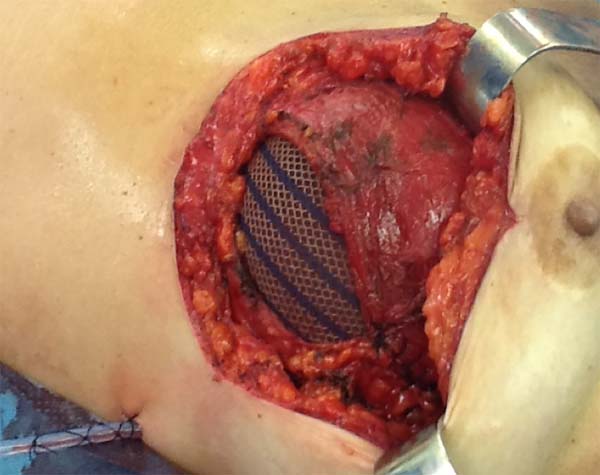
The superior-lateral portion of the pectoralis major is sutured to the fascia
of the serratus anterior muscle with Vicryl 2-0, and the mesh is placed over
the exposed portion of the implant. The mesh is then sutured with PDS thread
2-0 to the lateral margin of the pectoralis major and the fascia of the
serratus anterior muscle to support the implant laterally and inferiorly
(Figure 1). Intramuscular local
anesthesia with bupivacaine solution (20 mL) is administered to reduce
postoperative pain. The skin is then sutured in three layers, and the
incision is covered with Steri-Strip® (Figures 2 and 3)
and a surgical bra.
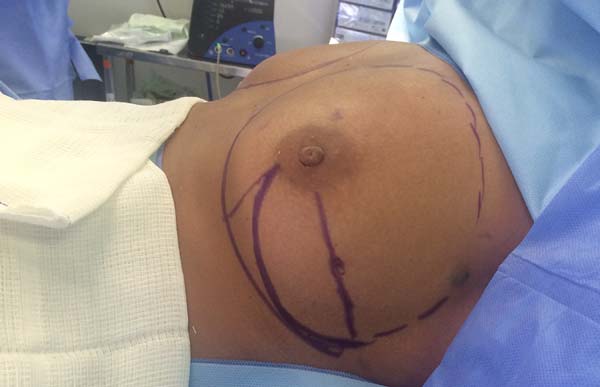
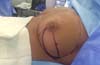
Figura 1 - Demonstration of the use of a mixed mesh sutured between the
pectoralis major and serratus anterior muscles.
Figura 1 - Demonstration of the use of a mixed mesh sutured between the
pectoralis major and serratus anterior muscles.
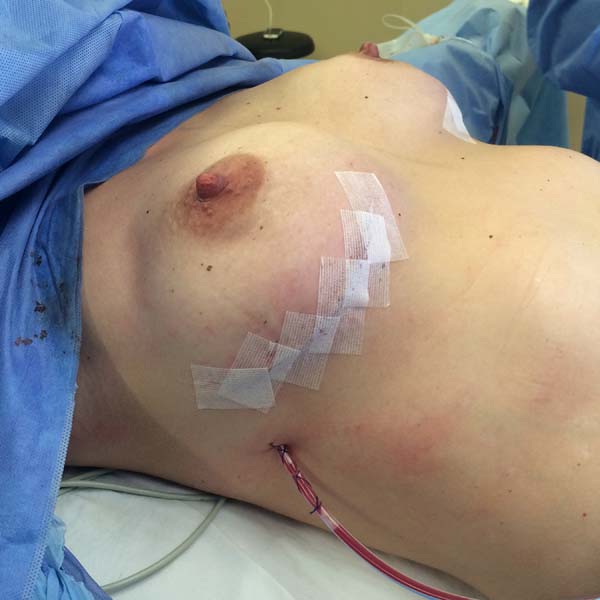
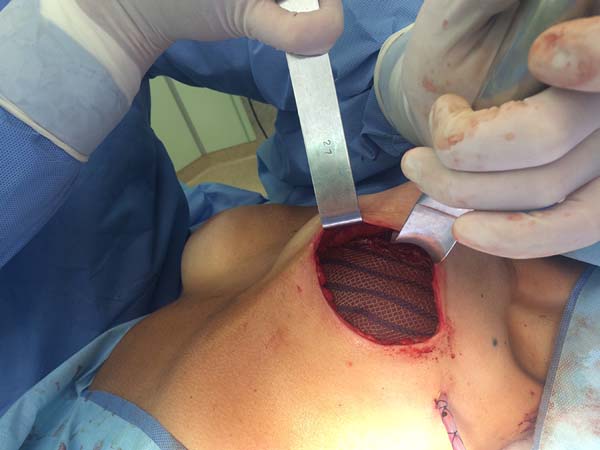
Figura 2 - Perioperative lateral view of a patient undergoing
prophylactic bilateral adenomastectomy with implant and mixed
mesh.
Figura 2 - Perioperative lateral view of a patient undergoing
prophylactic bilateral adenomastectomy with implant and mixed
mesh.
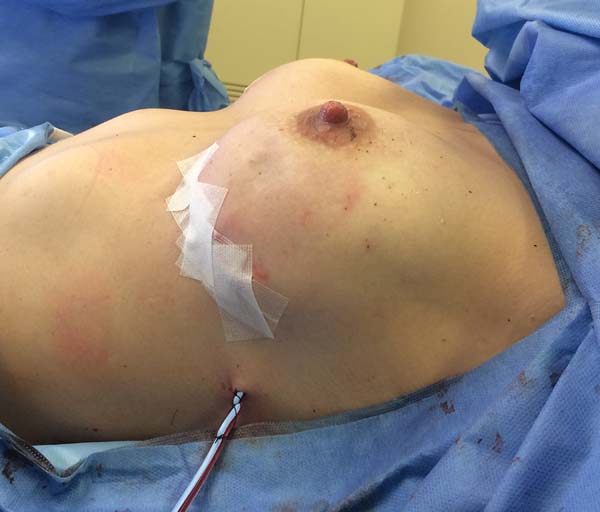
Figura 3 - Perioperative lateral view of a patient undergoing
prophylactic bilateral adenomastectomy with implant and mixed
mesh.
Figura 3 - Perioperative lateral view of a patient undergoing
prophylactic bilateral adenomastectomy with implant and mixed
mesh.
In one case of secondary reconstruction, the technique had to be slightly
modified owing to insufficient muscle coverage. In that case, because the
pectoralis major had been sectioned during the primary surgery, the lower
lateral portion of the mesh was sutured to the serratus anterior muscle and
the aponeurosis of the rectus abdominis muscle, while the upper portion was
sutured to the small lobe of the remaining pectoralis major (Figure 4). In addition, capsulotomy or
capsulectomy was performed in these cases.
Figure 4 - Perioperative view of a patient undergoing secondary breast
reconstruction. Owing to insufficient muscle coverage, the
inferior portion of the mesh was sutured in the aponeurosis of
the rectus abdominis muscle, laterally to the fascia of the
serratus anterior muscle and superiorly to the pectoralis
major.
Figure 4 - Perioperative view of a patient undergoing secondary breast
reconstruction. Owing to insufficient muscle coverage, the
inferior portion of the mesh was sutured in the aponeurosis of
the rectus abdominis muscle, laterally to the fascia of the
serratus anterior muscle and superiorly to the pectoralis
major.
Postoperative period
The patients were discharged from the hospital between 24 and 48 hours after
surgery and administered antibiotics. Rest with moderate activity for a
period of 30-45 days was recommended, along with the use of a surgical
bra.
The patients were monitored weekly during the first postoperative month and
then again at three and six months postoperatively. The drains were removed
when drainage was less than 30 mL/day.
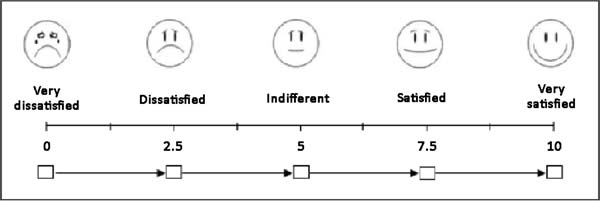
At postoperative 6 months, the patients were photographed and asked to
describe their level of satisfaction regarding the surgical technique that
was used by giving each parameter a score from 0 to 10, based on a visual
analog scale (Figure 5). The following
parameters were assessed: sensitivity, appearance, texture, symmetry, and
scar quality. In addition, the patients were asked whether they would choose
a different technique, with response options of “yes”, “maybe”, and
“no”.
Figure 5 - Visual analog scale used in the analysis of the patient’s
degree of satisfaction.
Figure 5 - Visual analog scale used in the analysis of the patient’s
degree of satisfaction.
Lastly, the potential complications of the technique, including infection
(requiring intravenous antibiotics), necrosis of the mastectomy flap or of
the NAC, seroma, hematoma, suture dehiscence, rippling, and
capsular contracture, were analyzed.
RESULTS
Twelve patients (20 breasts) underwent breast reconstruction with mixed mesh and
implants between November 2015 and December 2016. The mean age of the patients
was 55.6 years (35-67 years), and the mean body mass index (BMI) was 25.6
kg/m2 (19-29 kg/m2). The most common comorbidities
were hypertension (16%) and hypothyroidism (8%). One patient (8%) was a smoker,
two patients (16%) had a previous history of radiotherapy, one patient (8%) had
a history of neoadjuvant chemotherapy, and three patients (25%) had undergone
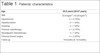
breast surgery. The mean follow-up time was 14 months (Table 1).
Table 1 - Patients’ characteristics.
| Age |
55.6 years (36-67 years) |
| BMI |
25.6 kg/m2 (19-29
kg/m2).
|
| Hypertension |
n = 2 (16%) |
| Hypothyroidism |
n = 1 (8%) |
| Smoking |
n = 1 (8%) |
| Previous radiotherapy |
n = 2 (16%) |
| Neoadjuvant chemotherapy |
n = 1 (8%) |
| Previous breast surgery |
n = 3 (25%) |
| Follow-up |
14 months (6-18 months) |
Table 1 - Patients’ characteristics.
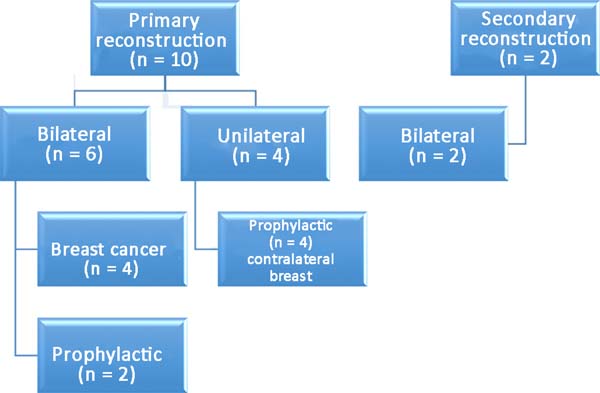
Among the patients, 83% (n=10) underwent primary breast reconstruction and 17%
(n=2) underwent secondary breast reconstruction (Figure 6). The secondary reconstructions were performed in patients
who had undergone conservative surgery for breast cancer with adjuvant
radiotherapy and who exhibited severe asymmetry. Bilateral reconstructions
accounted for 66% (n=8) of the cases, whereas unilateral reconstructions
comprised 34% (n=4) of the cases. Considering only the bilateral
reconstructions, 75% (n=6) were primary and 25% (n=2) were secondary
reconstructions. Of the bilateral primary reconstructions, 66% (n=4) were
NAC-sparing mastectomies for breast cancer and 34% (n=2) were prophylactic
mastectomies in patients with the BRCA1 gene mutation.
Figura 6 - Distribution of patients according to the type of
reconstruction.
Figura 6 - Distribution of patients according to the type of
reconstruction.
In patients with the gene mutation, prophylactic adenomastectomy was associated
with videolaparoscopic bilateral oophorectomy. The mean duration of unilateral
surgeries was 100 minutes (80-120 min), while that of bilateral surgeries was
180 minutes (100-240 min). The mean volume of the used implants was 345 mL
(270-415 mL). The mean duration of hospitalization was 36 hours (12-72 h). The
mean time of drain use was 12.2 days.
The complications are listed in Table 2.
Three breasts (15%) had minor complications. One patient (5%) had a hematoma of
moderate volume in the immediate postoperative period, which was treated
conservatively with drainage in an outpatient setting. Another patient exhibited
rippling at six months postoperatively, with spontaneous
improvement after 12 months (Figures 7 and
8). Lastly, one patient had
epidermolysis and suture dehiscence after unilateral adenomastectomy and breast
reconstruction with implant and mesh. This patient was treated with surgical
debridement and resuture because she resided in a different city and was not
able to attend frequent follow-ups.
Table 2 - Postoperative complications.
| |
20 breasts |
| Infection |
0 |
| Flap necrosis |
0 |
| NAC necrosis |
0 |
| Suture dehiscence |
n = 1 (5%) |
| Hematoma |
n = 1 (5%) |
| Rippling |
n = 1 (5%) |
| Seroma |
0 |
| Capsular contracture |
0 |
| Total |
3 (15%) |
Table 2 - Postoperative complications.
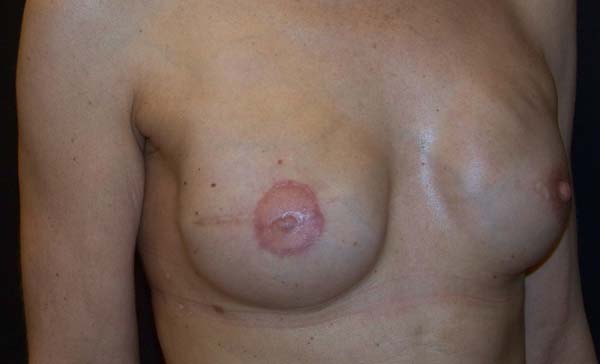
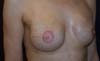
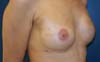
Figure 7 - Six-month follow-up of a 47-year-old patient who underwent
prophylactic left adenomastectomy after right mastectomy for
invasive ductal carcinoma, with visible rippling in the left
breast.
Figure 7 - Six-month follow-up of a 47-year-old patient who underwent
prophylactic left adenomastectomy after right mastectomy for
invasive ductal carcinoma, with visible rippling in the left
breast.
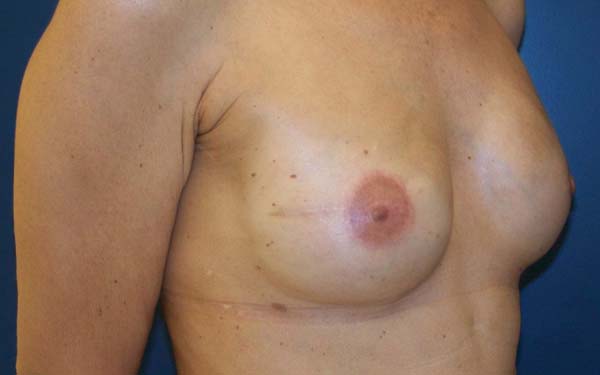
Figure 8 - Twelve-month follow-up of a 47-year-old patient who underwent
prophylactic left adenomastectomy after right mastectomy for
invasive ductal carcinoma. Note the improvement of rippling.
Figure 8 - Twelve-month follow-up of a 47-year-old patient who underwent
prophylactic left adenomastectomy after right mastectomy for
invasive ductal carcinoma. Note the improvement of rippling.
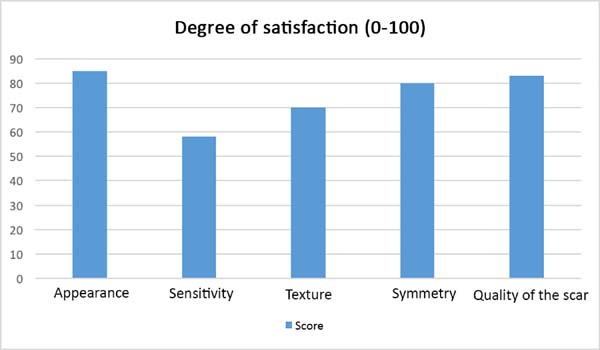
After the sixth month postoperatively, the patients answered the questionnaire
regarding their degree of satisfaction. The mean score for satisfaction with
breast appearance was 85 points. Sensitivity (58 points), texture (70 points),
symmetry (80 points), and scar quality (83 points) were also scored. The overall
mean was 75.2 points (Figure 9). Only one
patient (6%) responded “maybe” when asked whether she would opt for a different
surgical technique.
Figure 9 - Patient degree of satisfaction after the surgical procedure with
regard to the 5 parameters.
Figure 9 - Patient degree of satisfaction after the surgical procedure with
regard to the 5 parameters.
DISCUSSION
The benefits of immediate breast reconstruction have been proven with regard to
improved quality of life in mastectomy patients, especially among young
women14. Indeed, the level of
psychosocial stress caused by the feeling of mutilation is alleviated in
patients who undergo immediate reconstruction8.
The major advantage of immediate reconstruction with implants is that it is
performed in a single stage, which means less patient morbidity and reduced
costs. However, rigorous patient selection is mandatory to achieve a good
cosmetic result after surgery. The decision to perform immediate reconstruction
should be multifactorial and should consider important preoperative factors such
as the patient’s oncological status, presence of comorbidities, smoking, and
previous history of mammoplasty, neoadjuvant chemotherapy, or radiotherapy15.
Assessment of the mastectomy flap is another critical factor in the success of
the surgery. This assessment may be performed clinically by analyzing patterns
of ischemia or through studies of intraoperative imaging16. When intraoperative adjuvant radiotherapy is indicated,
two-stage reconstruction with an expander should be selected, since irradiation
of the implants is associated with increased complications17.
The success of the cosmetic result of immediate reconstructions with implants
also hinges on the evolution of mastectomy surgical techniques, in particular
skin-sparing and NAC-sparing mastectomies. The primary indications for this type
of approach include prophylactic mastectomy or early-stage tumors; however, the
spectrum of indications has been increasing. In the context of the appropriate
indication, immediate reconstruction has been shown to be associated with a low
rate of complications and good oncologic safety, even in patients with locally
advanced tumors who underwent neoadjuvant chemotherapy18.
The use of acellular dermal matrices in breast reconstruction has allowed
surgeons to obtain better cosmetic results as a result of less musculofascial
dissection, better control of the inframammary fold with projection of the lower
pole, and expansion of the submuscular pocket, which permits the safe use of
larger implants19.
Despite these benefits, the use of acellular dermal matrices in Brazil is still
restricted by the policies governed by Anvisa, which forbid the use of human
(cadaveric) biological material in other patients. Dermal matrices of animal
origin (porcine or bovine) are an alternative that yields similar results20, but their use is limited by their high
cost. In general, it is estimated that the use of matrices is 8 to 10 times more
expensive than that of meshes. Therefore, the use of a synthetic material that
mimics biological matrices is being advocated with the aim of obtaining optimal
aesthetic results.
In the present study, a partially absorbable, lightweight mesh made of synthetic
materials, namely Prolene (polypropylene) and Monocryl (poliglecaprone), was
used. The Monocryl portion is absorbed within 90-120 days and becomes
incorporated into the tissue. The use of synthetic meshes in plastic surgery is
not novel. In fact, many authors in Brazil have published studies on the
subject, with a large number of citations pertaining to methods of mastopexy
with mesh support21,22. The use of this synthetic material in
breast reconstruction is also not new and has been shown to be a very safe
technique with a low rate of complications, despite some technical differences
between the published articles23,24.
The rigorous selection of appropriate patients is required for the success of the
surgical technique. In the present study, 12 consecutive patients with a
postoperative follow-up of at least six months were selected. Although obesity,
smoking, and history of radiation may increase the risk of complications from
this procedure15, a multivariate analysis
of the patients’ characteristics as risk factors for complications was not
performed owing to the small size of the sample. However, in the study cohort,
the factors of the patients’ medical conditions, history of mammoplasty, and
smoking appeared to have minimally impacted the rate of complications, with the
viability of the mastectomy flap being the primary factor linked to the
occurrence of epidermolysis and suture dehiscence.
The patients who underwent bilateral prophylactic adenomastectomy had a strong
family history and BRCA1 gene mutation, and breast reconstruction in combination
with videolaparoscopic bilateral oophorectomy was indicated. In addition, the
association of an abdominal surgery did not appear to increase complications,
even with an increased duration of surgery.
All patients who underwent unilateral procedures with this technique had
undergone previous mastectomy and breast reconstruction with an expander or
flap. During the exchange of the expander for the definitive implant or in a
second stage, when indicated, these patients underwent contralateral
prophylactic adenomastectomy and reconstruction with the technique in question.
The patient who progressed to suture dehiscence and required reoperation was
included in this group. In cases of prophylactic surgery, given the absence of
local disease, it is essential to ensure that the mastectomy flap has an
adequate thickness to avoid ischemic complications.
The patients who underwent secondary reconstruction exhibited the same profile,
i.e., they had undergone conservative surgery and adjuvant radiotherapy and
exhibited significant asymmetry. However, one patient had undergone
reconstruction with implants at the time and progressed to capsular contracture
in addition to asymmetry (Figures 10 to
13).
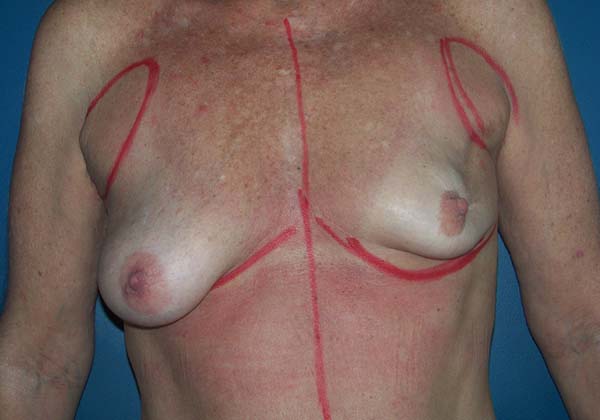
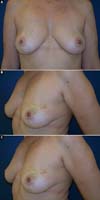
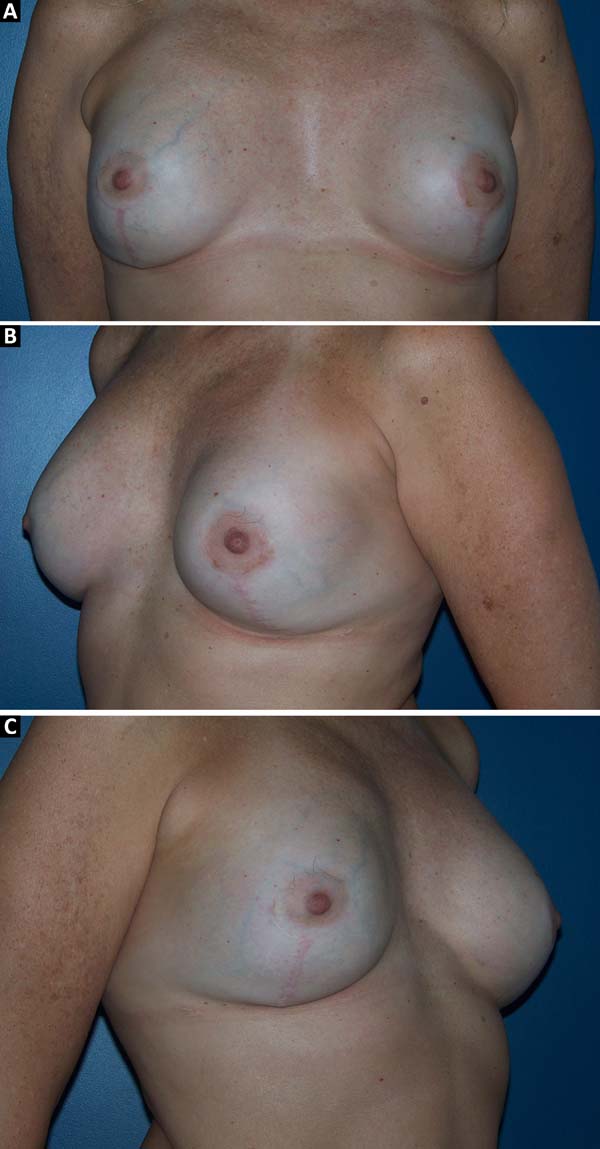
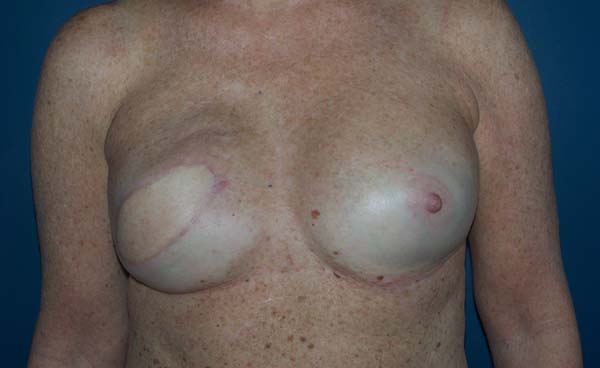
Figure 10 - Preoperative view of a 52-year-old patient who underwent
conservative surgery for invasive ductal carcinoma in the left
breast 10 years ago with significant asymmetry.
Figure 10 - Preoperative view of a 52-year-old patient who underwent
conservative surgery for invasive ductal carcinoma in the left
breast 10 years ago with significant asymmetry.
Figure 11 - Postoperative view (6 months) of a 52-year-old patient after
bilateral breast reconstruction with implant, mesh, and fat
graft.
Figure 11 - Postoperative view (6 months) of a 52-year-old patient after
bilateral breast reconstruction with implant, mesh, and fat
graft.
Figure 12 - Preoperative view of a 47-year-old patient who underwent
conservative surgery for ductal in situ carcinoma
in the left breast 8 years ago with significant asymmetry and
capsular contracture.
Figure 12 - Preoperative view of a 47-year-old patient who underwent
conservative surgery for ductal in situ carcinoma
in the left breast 8 years ago with significant asymmetry and
capsular contracture.
Figure 13 - Postoperative view (6 months) of a 47-year-old patient after
bilateral breast reconstruction with implant, mesh, and fat
graft.
Figure 13 - Postoperative view (6 months) of a 47-year-old patient after
bilateral breast reconstruction with implant, mesh, and fat
graft.
Another factor that is essential to reduce complications is the location of the
incisions. This should be discussed and planned based on the mastologist’s
experience, tumor location (in the case of cancer), and presence or absence of
previous scars. In the present study, there were incisions along the submammary
fold, comma-shaped inferior incisions (Figure 14), periareolar incisions with lateral extension (Figure 15), and classical mammoplasty
incisions.
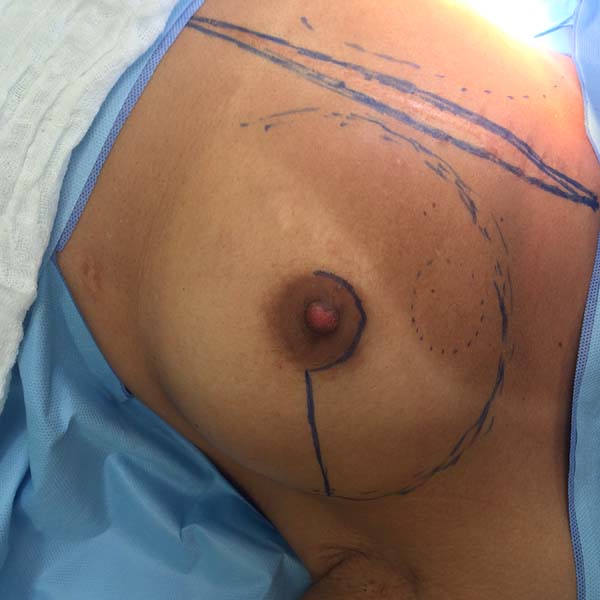
Figura 14 - Comma-shaped incision.
Figura 14 - Comma-shaped incision.
Figura 15 - Superior periareolar incision with lateral extension.
Figura 15 - Superior periareolar incision with lateral extension.
Incisions that involve the areola appear to be more associated with ischemic
complications of the NAC25. Thus,
patients at higher risk of NAC necrosis after mastectomy, such as smokers or
patients with previous mammoplasty, may undergo areola autonomization in an
outpatient setting26 to improve local
vascularization. However, no patient in the present study underwent this
procedure. It is very important that the mastectomy flap is well vascularized,
especially when incisions are made to elevate the NAC.
One of the main complaints of patients in the postoperative period is pain, which
is primarily caused by the major musculofascial dissection. Therefore, the aim
of using intramuscular bupivacaine was to control pain and reduce the need for
opioids postoperatively. Although the results of using liposomal bupivacaine are
promising27, its use is still not
permitted in Brazil. Recent studies analyzing immediate breast reconstruction
with implants covered by dermal matrices in the prepectoral position also aimed
to reduce postoperative pain as well as complications such as hyper-animation
deformity, but they remain very controversial28.
Despite the small size of the study sample, the rate of complications of
reconstruction surgery with implants and mesh was similar to that observed in
other large series29,30. The degree of satisfaction of the
patients was measured using a visual analog scale comprising 5 parameters. While
the best method for assessing the patients’ degree of satisfaction in terms of
quality of life after breast reconstruction is a validated and translated
questionnaire, such as the Breast-Q31, it
is a long questionnaire that patients have difficulty completing in the private
clinic setting. Therefore, it was not used in the present study.
The patients were primarily dissatisfied with the sensitivity; however, because
the questionnaire was administered at six months postoperatively, these patients
would likely report some degree of improvement in a later follow-up. Although
the parameter “texture” had a good satisfaction score in the questionnaire, some
patients reported feeling the surface of the mesh, especially in thin flaps.
However, this complaint was not frequent after the period of absorption of the
PDS thread, which is used in the fixation of the mesh to the muscle wall, and of
the Monocryl portion of the mesh. The patients’ degree of satisfaction with the
cosmetic result also scored well, which validates the continued application of
the technique (Figures 16 to 19).
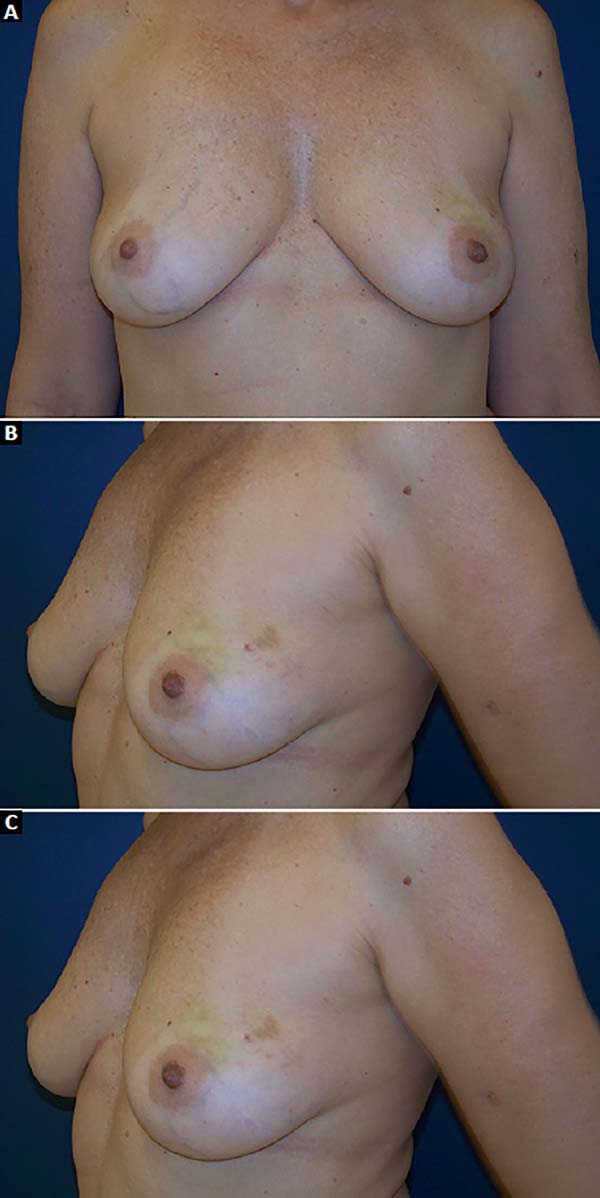
Figure 16 - Preoperative view of a 47-year-old patient with a history of
ductal carcinoma in situ in the left breast.
A: Frontal view; B: Left oblique view;
C: Right oblique view.
Figure 16 - Preoperative view of a 47-year-old patient with a history of
ductal carcinoma in situ in the left breast.
A: Frontal view; B: Left oblique view;
C: Right oblique view.
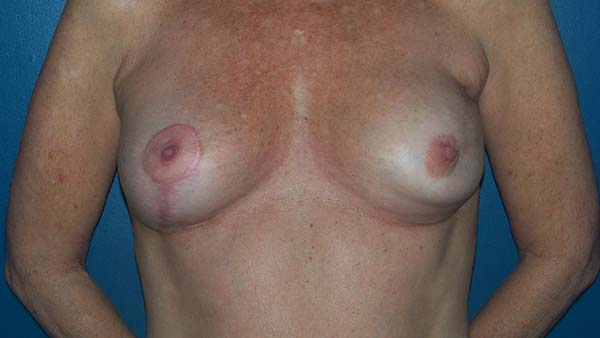
Figure 17 - Postoperative view (6 months) of a 47-year-old patient who
underwent bilateral breast reconstruction with implant and mesh.
A: Frontal view; B: Left oblique view;
C: Right oblique view.
Figure 17 - Postoperative view (6 months) of a 47-year-old patient who
underwent bilateral breast reconstruction with implant and mesh.
A: Frontal view; B: Left oblique view;
C: Right oblique view.
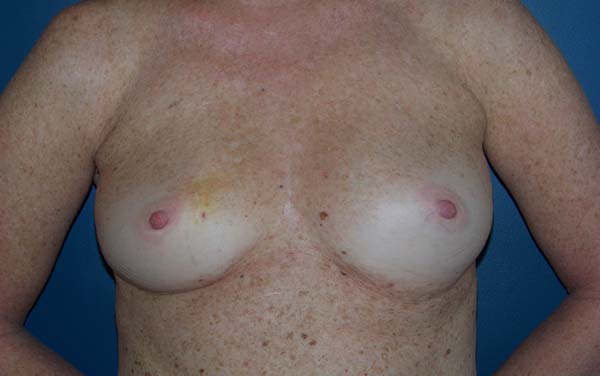
Figure 18 - Preoperative view of a 53-year-old patient with a history of
invasive ductal carcinoma in the right de breast.
Figure 18 - Preoperative view of a 53-year-old patient with a history of
invasive ductal carcinoma in the right de breast.
Figure 19 - Postoperative view (6 months) of a 53-year-old patient with a
history of invasive ductal carcinoma in the right breast who
underwent reconstruction with latissimus dorsi muscle flap and
implant in the right breast and prophylactic adenomastectomy and
reconstruction with implant and mesh in the left breast.
Figure 19 - Postoperative view (6 months) of a 53-year-old patient with a
history of invasive ductal carcinoma in the right breast who
underwent reconstruction with latissimus dorsi muscle flap and
implant in the right breast and prophylactic adenomastectomy and
reconstruction with implant and mesh in the left breast.
Although this was a retrospective study with a small number of cases, the results
demonstrated that the proposed reconstruction technique with implants and mixed
mesh has a lower rate of complications compared to other techniques of immediate
reconstruction with implants with total muscle coverage or the use of dermal
matrices.
Studies on the placement of implants with total muscle coverage indicate
complication rates of up to 40%, including mainly implant malposition, asymmetry
of the submammary fold, and capsular contracture30. Therefore, the limitations of this technique are based on the
small size of the submuscular pocket, which prevents the placement of larger
implants and hinders the creation of a natural breast and a defined submammary
fold. The use of a mesh helps to enlarge the pocket and allows for better
control of the implant positioning, as well as greater expansion of the lower
pole of the breast.
A prospective, randomized, and controlled study is necessary to compare the use
of mesh with that of acellular dermal matrices, including an assessment of
cost-effectiveness. Matrix-related complications are usually associated with
infection and seroma, with rates ranging between 6% and 29%32,33. In
the present study, the complications were minor and occurred at a rate of
15%.
CONCLUSION
The proposed breast reconstruction technique using implants and synthetic mesh
was found to be associated with a low rate of complications, a high degree of
patient satisfaction with the cosmetic result, and a lower cost relative to the
use of acellular dermal matrices. However, rigorous patient selection, careful
incision planning, and a well-vascularized flap after mastectomy are extremely
critical factors for the success of the surgical procedure.
COLLABORATIONS
|
DGL
|
Analysis and/or interpretation of data; statistical analyses; final
approval of the manuscript; conception and design of the study;
completion of surgeries and/or experiments; writing the manuscript
or critical review of its contents.
|
REFERENCES
1. Halsted WS. I. The Results of Radical Operations for the Cure of
Carcinoma of the Breast. Ann Surg. 1907;46(1):1-19. PMID:
17861990
2. Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER,
et al. Twenty-year follow-up of a randomized trial comparing total mastectomy,
lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast
cancer. N Engl J Med. 2002;347(16):1233-41. PMID: 12393820 DOI: http://dx.doi.org/10.1056/NEJMoa022152
3. Al-Ghazal SK, Fallowfield L, Blamey RW. Comparison of psychological
aspects and patient satisfaction following breast conserving surgery, simple
mastectomy and breast reconstruction. Eur J Cancer. 2000;36(15):1938-43. DOI:
http://dx.doi.org/10.1016/S0959-8049(00)00197-0
4. Taylor GI. The angiosomes of the body and their supply to perforator
flaps. Clin Plast Surg. 2003;30(3):331-42. DOI: http://dx.doi.org/10.1016/S0094-1298(03)00034-8
5. Reuben BC, Manwaring J, Neumayer LA. Recent trends and predictors in
immediate breast reconstruction after mastectomy in the United States. Am J
Surg. 2009;198(2):237-43. DOI: http://dx.doi.org/10.1016/j.amjsurg.2008.11.034
6. Alderman AK, Wei Y, Birkmeyer JD. Use of breast reconstruction after
mastectomy following the Women's Health and Cancer Rights Act. JAMA.
2006;295(4):387-8. PMID: 16434628
7. Brasil. Governo Federal. Ministério da Saúde. Lei Nº 9.797, de 6 de
maio de 1999. Alterada pela Lei Federal 12.802, de 24/04/2013. Dispõe sobre a
obrigatoriedade da cirurgia plástica reparadora da mama pela rede de unidades
integrantes do Sistema Único de Saúde - SUS nos casos de mutilação decorrentes
de tratamento de câncer. Brasília: Governo Federal; 2013.
8. Zhong T, Hu J, Bagher S, Vo A, O'Neill AC, Butler K, et al. A
Comparison of Psychological Response, Body Image, Sexuality, and Quality of Life
between Immediate and Delayed Autologous Tissue Breast Reconstruction: A
Prospective Long-Term Outcome Study. Plast Reconstr Surg. 2016;138(4):772-80.
PMID: 27673514 DOI: http://dx.doi.org/10.1097/PRS.0000000000002536
9. Albornoz CR, Bach PB, Mehrara BJ, Disa JJ, Pusic AL, McCarthy CM, et
al. A paradigm shift in U.S. Breast reconstruction: increasing implant rates.
Plast Reconstr Surg. 2013;131(1):15-23. PMID: 23271515 DOI: http://dx.doi.org/10.1097/PRS.0b013e3182729cde
10. Munhoz AM, Aldrighi CM, Montag E, Arruda EG, Aldrighi JM, Gemperli
R, et al. Clinical outcomes following nipple-areola-sparing mastectomy with
immediate implant-based breast reconstruction: a 12-year experience with an
analysis of patient and breast-related factors for complications. Breast Cancer
Res Treat. 2013;140(3):545-55. PMID: 23897416 DOI: http://dx.doi.org/10.1007/s10549-013-2634-7
11. Macadam SA, Lennox PA. Acellular dermal matrices: Use in
reconstructive and aesthetic breast surgery. Can J Plast Surg. 2012;20(2):75-89.
DOI: http://dx.doi.org/10.1177/229255031202000201
12. Maxwell GP, Gabriel A. Use of the acellular dermal matrix in
revisionary aesthetic breast surgery. Aesthet Surg J. 2009;29(6):485-93. DOI:
http://dx.doi.org/10.1016/j.asj.2009.09.007
13. Shulman AG, Amid PK, Lichtenstein IL. The safety of mesh repair for
primary inguinal hernias: results of 3,019 operations from five diverse surgical
sources. Am Surg. 1992;58(4):255-7. PMID: 1586085
14. Dauplat J, Kwiatkowski F, Rouanet P, Delay E, Clough K, Verhaeghe
JL, et al.; STIC-RMI working group. Quality of life after mastectomy with or
without immediate breast reconstruction. Br J Surg. 2017;104(9):1197-206. DOI:
http://dx.doi.org/10.1002/bjs.10537
15. Tang R, Coopey SB, Colwell AS, Specht MC, Gadd MA, Kansal K, et al.
Nipple-Sparing Mastectomy in Irradiated Breasts: Selecting Patients to Minimize
Complications. Ann Surg Oncol. 2015;22(10):3331-7. DOI: http://dx.doi.org/10.1245/s10434-015-4669-y
16. Gurtner GC, Jones GE, Neligan PC, Newman MI, Phillips BT, Sacks JM,
et al. Intraoperative laser angiography using the SPY system: review of the
literature and recommendations for use. Ann Surg Innov Res. 2013;7(1):1. DOI:
http://dx.doi.org/10.1186/1750-1164-7-1
17. Kronowitz SJ, Robb GL. Radiation therapy and breast reconstruction:
a critical review of the literature. Plast Reconstr Surg. 2009;124(2):395-408.
PMID: 19644254 DOI: http://dx.doi.org/10.1097/PRS.0b013e3181aee987
18. Coopey SB, Tang R, Lei L, Freer PE, Kansal K, Colwell AS, et al.
Increasing eligibility for nipple-sparing mastectomy. Ann Surg Oncol.
2013;20(10):3218-22. DOI: http://dx.doi.org/10.1245/s10434-013-3152-x
19. Vardanian AJ, Clayton JL, Roostaeian J, Shirvanian V, Da Lio A, Lipa
JE, et al. Comparison of implant-based immediate breast reconstruction with and
without acellular dermal matrix. Plast Reconstr Surg. 2011;128(5):403e-410e.
PMID: 22030500
20. Glasberg SB, Light D. AlloDerm and Strattice in breast
reconstruction: a comparison and techniques for optimizing outcomes. Plast
Reconstr Surg. 2012;129(6):1223-33. PMID: 22327891 DOI: http://dx.doi.org/10.1097/PRS.0b013e31824ec429
21. Sampaio Góes JC. Periareolar mammaplasty: double-skin technique with
application of mesh support. Clin Plast Surg. 2002;29(3):349-64. DOI: http://dx.doi.org/10.1016/S0094-1298(02)00005-6
22. Bozola AR. Mamoplastia pós-cirurgia bariátrica usando suporte
protético complementar de contenção glandular. Rev Bras Cir Plást.
2016;31(3):299-307.
23. Tessler O, Reish RG, Maman DY, Smith BL, Austen WG Jr. Beyond
biologics: absorbable mesh as a low-cost, low-complication sling for
implant-based breast reconstruction. Plast Reconstr Surg. 2014;133(2):90e-9e.
PMID: 24469217 DOI: http://dx.doi.org/10.1097/01.prs.0000437253.55457.63
24. Pukancsik D, Kelemen P, Gulyás G, Újhelyi M, Kovács E, Éles K, et
al. Clinical experiences with the use of ULTRAPRO® mesh in single-stage
direct-to-implant immediate postmastectomy breast reconstruction in 102
patients: A retrospective cohort study. Eur J Surg Oncol. 2017;43(7):1244-51.
DOI: http://dx.doi.org/10.1016/j.ejso.2017.01.236
25. Rawlani V, Fiuk J, Johnson SA, Buck DW 2nd, Hirsch E, Hansen N, et
al. The effect of incision choice on outcomes of nipple-sparing mastectomy
reconstruction. Can J Plast Surg. 2011;19(4):129-33. DOI: http://dx.doi.org/10.1177/229255031101900410
26. Jensen JA, Lin JH, Kapoor N, Giuliano AE. Surgical delay of the
nipple-areolar complex: a powerful technique to maximize nipple viability
following nipple-sparing mastectomy. Ann Surg Oncol. 2012;19(10):3171-6. DOI:
http://dx.doi.org/10.1245/s10434-012-2528-7
27. Leiman D, Barlow M, Carpin K, Piña EM, Casso D, et al. Medial and
lateral pectoral nerve block with liposomal bupivacaine for the management of
postsurgical pain after submuscular breast augmentation. Plast Reconstr Surg
Glob Open. 2015;2(12):e282.
28. Sigalove S, Maxwell GP, Sigalove NM, Storm-Dickerson TL, Pope N,
Rice J, et al. Prepectoral Implant-Based Breast Reconstruction: Rationale,
Indications, and Preliminary Results. Plast Reconstr Surg. 2017;139(2):287-94.
DOI: http://dx.doi.org/10.1097/PRS.0000000000002950
29. Baldelli I, Cardoni G, Franchelli S, Fregatti P, Friedman D, Pesce
M, et al. Implant-Based Breast Reconstruction Using a Polyester Mesh
(Surgimesh-PET): A Retrospective Single-Center Study. Plast Reconstr Surg.
2016;137(6):931e-9e. PMID: 27219260 DOI: http://dx.doi.org/10.1097/PRS.0000000000002180
30. Meyer Ganz O, Tobalem M, Perneger T, Lam T, Modarressi A, Elias B,
et al. Risks and benefits of using an absorbable mesh in one-stage immediate
breast reconstruction: a comparative study. Plast Reconstr Surg.
2015;135(3):498e-507e. DOI: http://dx.doi.org/10.1097/PRS.0000000000001027
31. Pusic AL, Klassen AF, Scott AM, Klok JA, Cordeiro PG, Cano SJ.
Development of a new patient-reported outcome measure for breast surgery: the
BREAST-Q. Plast Reconstr Surg. 2009;124(2):345-53. PMID: 19644246 DOI: http://dx.doi.org/10.1097/PRS.0b013e3181aee807
32. Chun YS, Verma K, Rosen H, Lipsitz S, Morris D, Kenney P, et al.
Implant-based breast reconstruction using acellular dermal matrix and the risk
of postoperative complications. Plast Reconstr Surg. 2010;125(2):429-36. PMID:
20124828 DOI: http://dx.doi.org/10.1097/PRS.0b013e3181c82d90
33. Lanier ST, Wang ED, Chen JJ, Arora BP, Katz SM, Gelfand MA, et al.
The effect of acellular dermal matrix use on complication rates in tissue
expander/implant breast reconstruction. Ann Plast Surg. 2010;64(5):674-8. PMID:
20395795
1. Sociedade Brasileira de Cirurgia Plástica, São
Paulo, SP, Brazil.
2. Américas Centro de Oncologia Integrado, Rio de
Janeiro, RJ, Brazil.
3. Hospital Federal de Ipanema, Rio de Janeiro,
RJ, Brazil.
*Corresponding author: Daniel Gouvea Leal, Rua
Redentor, 26 - Rio de Janeiro, RJ, Brazil. Zip Code 22421-030. E-mail:
danileal.rlk@terra.com.br
Article received: June 5, 2017.
Article accepted: August 7, 2017.
Conflicts of interest: none.