

Original Article - Year 2011 - Volume 26 -
Use of adhesion sutures in body-contouring surgery
Pontos de adesão na cirurgia do contorno corporal
ABSTRACT
Introduction: Seroma, hematoma, and scars are potential postoperative complications of abdominoplasty. Aspiration drainage and use of adhesion sutures are strategies for preventing such complications. The aim of this study was to assess the effectiveness of adhesion sutures for preventing or reducing the formation of seroma, hematoma, and scars after body- contouring surgery. Methods: One hundred and fifteen patients (group 2, January 2006- June 2010) who underwent body-contouring surgery with the use of adhesion sutures were retrospectively compared with 39 patients (group 1, January 2004-December 2005) who underwent the same surgery with aspiration drainage. The patients were clinically assessed at 7 days; 2 weeks; and 1, 2, 3, and 6 months postoperatively. Results: Group 1 had 9 (23%) cases of seroma formation, 1 (2.5%) case of hematoma formation, and 20 (55%) cases of scarring, whereas no complications were reported in group 2. The clinical assessment revealed statistical differences in the incidences of seroma formation and scarring between the groups. Conclusion: The use of adhesion sutures is an efficient method for preventing complications such as seroma, hematoma, and scar formation after body-contouring surgery.
Keywords: Abdomen. Seroma/prevention & control. Plastic surgery/methods.
RESUMO
Introdução: Seromas, hematomas e alterações cicatriciais são complicações potenciais no pós-operatório das abdominoplastias. Drenagem aspirativa e pontos de adesão são estratégias utilizadas na prevenção destas complicações. A proposta deste estudo é avaliar a eficácia dos pontos de adesão na prevenção ou redução da formação de seroma, hematoma e de complicações cicatriciais em pacientes submetidos a cirurgia plástica do contorno corporal. Método: Cento e quinze pacientes (grupo 2: janeiro de 2006 - junho de 2010) submetidos a cirurgia plástica do contorno corporal com o uso dos pontos de adesão foram comparados, retrospectivamente, com um grupo de 39 pacientes (grupo 1: janeiro de 2004 - dezembro de 2005) submetidos a mesma cirurgia com o uso de drenagem aspirativa. Avaliação clínica dos pacientes foi realizada com 7 dias, 2 semanas, um, dois, três e seis meses de pós-operatório. Resultados: No grupo 1, houve 9 (23%) casos de seroma, 1 (2,5%) de hematoma e 20 (55%) de complicações cicatriciais, enquanto que, no grupo 2, nenhum caso de seroma ou hematoma foi reportado. A avaliação clínica revelou diferenças estatísticas entre o uso dos pontos de adesão e a drenagem aspirativa, no que se refere à incidência de seroma e de complicações cicatriciais. Conclusão: O uso dos pontos de adesão é um método eficiente na prevenção de seromas, hematomas e complicações cicatriciais nas cirurgias do contorno corporal.
Palavras-chave: Abdome. Seroma/prevenção & controle. Cirurgia plástica/métodos.
The 130-year history of abdominoplasty, published by Avelar1, Hakme2, and Baroudi3, among others, includes records of numerous technical procedures, given the diverse morphological changes the abdomen goes through during life. At present, abdominoplasty can be divided into 4 basic procedures: (1) low transversal pubic abdominoplasty4, (2) xypho-pubic abdominoplasty with pubic transversal compensation and inverted T scar5, (3) expanded and not expanded miniabdominoplasty, with elongated incision type Pffanestiel6,7, and (4) reverse abdominoplasty8. Moreover, Gonzalez-Ulloa9, in 1960, described circular abdominoplasty. Over the years, abdominoplasty has undergone continuous technical refinements, improving procedural implementation, reducing the number of complications, and achieving better long-term results. These refinements have always allowed for reproducibility, as has consistently been observed in the specialized literature.
The rate of complications after abdominoplasty varies widely, between 4% and 80%10. One of the most frequent complications in the postoperative period is the formation of seroma, with an incidence ranging between 10% and 15%. In the recent decades, Penrose® or vacuum drains have been classically used for seroma prevention. However, several reports still describe the occurrence of this complication during the postoperative period of abdominal surgeries11-13.
Baroudi and Ferreira14,15, in 1996 and 1998, published their findings on the application of adhesion sutures in the course of abdominoplasty and in other dissected areas to eliminate dead space and the possibility of hematoma and seroma formation.
The aim of this study was to present our experience related to the effectiveness of adhesion sutures for preventing the formation of seroma, hematoma, and scars after body-contouring surgery, especially circular and isolated abdominoplasties.
METHODS
From January 2004 to June 2010, 154 abdominoplasties, with or without other surgical procedures, were performed. The patient age ranged from 34 years to 65 years, with an average of 45 years; the patients comprised 151 women and 3 men. All the patients underwent anamnesis, detailed physical examination, laboratory tests, and photographic recording. Surgery was contraindicated in patients with unabated comorbidities, a body mass index higher than 35, and considerable history of smoking. All the patients were informed about the procedures, problems, complications, and expected results; received printed information regarding the proposed procedure; and signed informed consent and liability forms.
Surgical selection was determined by the degree of body-contour impairment16. Patients with anterior abdominal wall impairment (predominantly infraumbilical) underwent expanded or not expanded miniabdominoplasty, those with infraumbilical and supraumbilical dermal-adipose impairment underwent low transversal pubic abdominoplasty, those patients with excess dermal-adipose tissue (usually secondary to intense weight loss) underwent xypho-pubic abdominoplasty (also called "fleur-de-lis" or inverted T abdominoplasty), and patients with dermal-adipose panniculus excess extending to the flanks and buttocks underwent circular torsoplasty.
The patients who underwent abdominoplasty with aspiration drainage between 2004 and 2005 were categorized as group 1. Drainage was performed with a Portovac #4.8 suction device, which was maintained until the drained volume was less than 30 ml in 24 h. In the patients who underwent abdominoplasty and bodyplasty between 2006 and 2010 (group 2), adhesion sutures were applied without drainage.
Surgical Technique
The surgeries were performed under general or epidural anesthesia with sedation. In all the patients, lower-limb compression was applied by means of elastic stockings and intermittent pneumatic massage; low-molecular-weight heparin (40 mg/week) was selectively used according to the individual risk rating.
During miniabdominoplasty and abdominoplasty, after excess-skin dissection and resection, the rectus muscles were plicated, when indicated. The dermal-adipose flap was repositioned by applying isolated adhesion sutures (2-0 absorbable sutures) in rows and columns from the upper limit of the dissected area along the xiphoid process, with a maximum distance of 4 cm between sutures, until the incision edges were approximated. The sutures were applied to transfix the muscular fascia of the abdominal wall and the lamellar adipose layer, with or without the superficial fascia. Externally, the navel was positioned on the skin. At each suture line, the correct position of the flap was assessed, until the junction with the skin incision bottom edge. The number of sutures ranged from 40 to 45. The incision edges at the dermal-adipose level were sutured with isolated 3-0 absorbable sutures and the cuticular layer was sutured with continuous 4-0 nonabsorbable sutures (Figures 1 and 2).

Figure 1 - Miniabdominoplasty. A: marking; B: placement of adhesion sutures; C: projection of the adhesion sutures in the dermal-adipose flap.
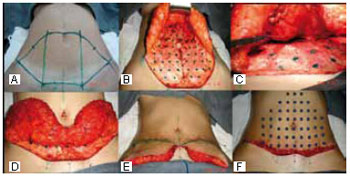
Figure 2 - Intraoperative view of low transversal pubic minoplasty evidencing the use of adhesion sutures.
During xypho-pubic abdominoplasty, after dermal-adipose flap resection, consisting of horizontal and vertical segments (Figures 3A and 3B), the rectus abdominis was plicated with isolated 0 nonabsorbable sutures and continuous 2-0 nonabsorbable sutures were applied for reinforcement. Then, the dermal-adipose flap was repositioned by applying adhesion sutures, securing the lamellar layer to the muscular aponeurosis, to pull the flap edges toward the midline (Figures 3C-3F).
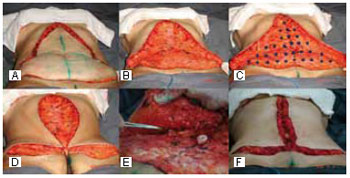
Figure 3 - Intraoperative view of "fleur-de-lis" abdominoplasty. A and B: dissection of the dermal-adipose flap; C: placement of adhesion sutures; D and E: repositioning of the flap edges with traction toward the abdominal midline; F: the repositioned flap secured with adhesion sutures.
For corpoplasty (also called circular abdominoplasty), general anesthesia with endotracheal intubation was preferred, with the patient in the horizontal prone position. After the appropriate resection (Figures 4A-4C), the open area between the gluteal muscular aponeurosis and the lamellar layer of the dermal-adipose flap was approximated with isolated (2-0 absorbable) adhesion sutures at 4-cm intervals (Figures 4D and 4E). The incision edges including the superficial fascia were sutured with single 3-0 absorbable sutures. The skin was sutured with isolated 3-0 absorbable sutures, and the cuticular layer was sutured with continuous 4-0 nonabsorbable sutures (Figure 4F). After this first surgical phase, skin excess in the flanks was isolated and covered, and the patient was positioned in the horizontal prone position. Abdominoplasty was then performed according to the routine described, and subsequently, the lateral excesses were resected and sutured bilaterally, resulting in a circular scar. At the end of the surgery, the scars were immobilized with porous tape and the patient was placed in a gown with a molding belt.
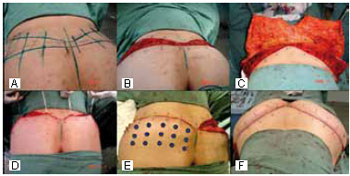
Figure 4 - Transoperative view of circular abdominoplasty, with the patient in the horizontal prone position. A-C: dissection of the dermal-adipose flap (C, cranial view); D and E: repositioning of the flap and securing with adhesion sutures; F: final aspect of the flap (cranial view).
Postoperative Care
The antibiotic therapy initiated during the surgery (1-g cephalosporin every 3 h) was maintained postoperatively for 1 week at a dosage of 500 mg twice daily. When indicated, low-molecular-weight heparin (40 mg/day) was administered until the seventh postoperative day.
Intermittent pneumatic massage, used during the surgical procedures, was maintained for 24 h, and the use of elastic stockings was advised until the patient resumed active strolling.
Postoperatively, the patients were assessed at 7 days; 2 weeks; and 1, 2, 3, and 6 months. Data on the presence of hematoma, seroma, and unsightly and poorly positioned scars as well as the patient rating and surgical team satisfaction were recorded.
Statistical Analysis
Student's t-test was used to assess the differences between the groups. Statistical analysis was performed with GraphPad Prism software (GraphPad Software, Inc., San Diego, CA, USA).
RESULTS
Group 1 comprised 39 patients, with only 1 male patient. One (2.5%) patient underwent miniabdominoplasty, 10 (25%) patients underwent low transversal abdominoplasty, and 2 (5%) patients underwent "fleur-de-lis" abdominoplasty; 16 (41%) surgeries were associated with liposuction of the flanks and back and 10 (25%) were associated with mammaplasty.
No anesthetic complication was recorded; on the 14th postoperative day, 1 (2.5%) patient developed deep venous thrombosis and remained hospitalized for 10 days with a good outcome.
The patients then underwent aspiration drainage for 2-8 days, with an average of 5 days. Of the 39 patients, 9 (23%) developed seroma after drain removal. Seven (77%) of the 9 patients underwent transcutaneous puncture and 2 (22%) underwent reoperation for serous bag removal. Four (44%) of the patients with seroma had undergone isolated abdominoplasty, 3 (33%) had undergone associated liposuction of the flanks and back, and 2 (22%) had undergone associated mammaplasty. One (2.5%) patient had a small hematoma in the hypogastric area but showed spontaneous recovery, without the need for intervention. Twelve (30%) patients developed pubic scar dehiscence: in 10 (83%), healing occurred by secondary intention, and in 2 (16%), resuturing was performed, given the extent of the dehiscence. Six (15%) patients had hypertrophic scars and 2 (5%) patients had keloid scars. Misplaced scars were observed in 16 (41%) patients, causing dissatisfaction to both the patients and the surgical team. Of these, 10 (62%) underwent scar retouching, but 6 (38%) preferred not to undergo the new procedure to correct the scar. No patient had skin flap or umbilicus pedicle necrosis (Table 1 and Figure 5).
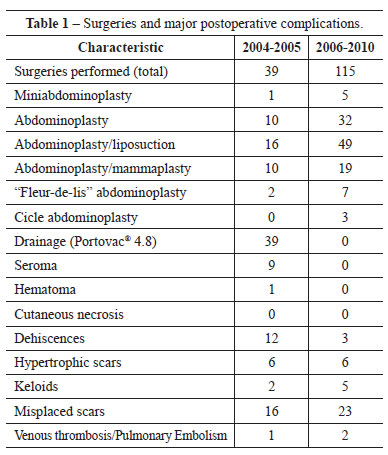
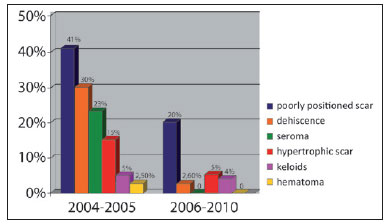
Figure 5 - Comparison of the incidences (%) of major complications between groups 1 and 2 (p<0.0001).
Group 2 comprised 115 patients, of which 113 (98%) were female patients. Five (4%) patients underwent miniabdominoplasty, 32 (27%) patients underwent abdominoplasty, 49 (42%) patients underwent abdominoplasty associated with liposuction of the flanks and back, and 19 (16%) patients underwent abdominoplasty combined with mammaplasty. Seven (6%) patients underwent "fleur-de-lis" abdominoplasty and 3 (2.6%) patients underwent corpoplasty. Of the 115 patients, 2 (1.7%) developed pulmonary thromboembolism (PTE) postoperatively: one of these patients had undergone abdominoplasty and the other had undergone abdominoplasty associated with liposuction. Both the patients required intensive care, with evolution without consequences.
Seroma and hematoma were not clinically observed in any of the patients to justify percutaneous or open drainage. Further, none of the patients had skin flap or umbilicus pedicle necrosis. Three (2.6%) patients showed suture dehiscence: one developed dehiscence in the flanks after circular abdominoplasty, which required resuturing, and the remaining developed dehiscence in the vertical compensation for the "inverted T" in the midline of the pubis, which healed by secondary intention. After 3 months, the latter 2 patients underwent local scar retouching. Hypertrophic scars formed in 6 (5%) patients and keloid scars developed in 5 (4%) patients. The scar position in the pubis was satisfactory in 80% of the patients with adhesion sutures. Among the 23 patients with badly positioned scars, 7 (39%) had their scars surgically retouched (Table 1 and Figure 5).
DISCUSSION
Hematoma and seroma are the most common postoperative complications of abdominoplasty. The proposed pathogenesis is based on lymphatic and vascular lesions, the development of dead space secondary to dermal-adipose flap detachment, and the shear force between the flap and the muscle-aponeurotic complex17. Bozola and Psillakis16, in 1988, related the incidence of seroma with the extent of surgery and indicated that larger surgeries have an increased predisposition for the development of this complication. Similarly, Matarasso18, in 1991, correlated the incidence of seroma with the type of surgery and suggested limited detachment and the use of suction drains. In 1999, Vastine et al.19 retrospectively analyzed 90 patients who underwent abdominoplasty; they noted the occurrence of complications in 80% of the patients with obesity at the time of the surgery, and the most frequent complications were seroma and scar dehiscence. Among the patients in the normal weight range, the complication rate was 30%. Nurkim et al.20, in 2002, reported the occurrence of seroma in 57% of their patients who underwent abdominoplasty without drainage, 29% of those who underwent the same surgery with chest tube drainage, and 4% of those who underwent vacuum drainage.
Baroudi and Ferreira14, in 1998, described the use of adhesion sutures with absorbable sutures between the dermal-adipose flap and the muscular aponeurosis to decrease dead space after the detachment in abdominoplasty. Daltrey et al.,21 in a randomized study involving 108 patients who underwent breast reconstruction with the latissimus dorsi, noted a considerable reduction in the incidence and volume of postoperative seroma. Moura and Bezerra22, in 2008, obtained similar results by retrospectively assessing 20 patients who underwent breast reconstruction with TRAM flap. Nahas et al.23 ultrasonographically assessed 21 patients who underwent abdominoplasty with adhesion sutures and noted the presence of seromas, with an average volume of 8.2 ml, at 2 weeks after the surgery.
In the present study, 23% of the group 1 patients had seroma after drain removal whereas none of the group 2 patients developed seroma, confirming the concepts of Baroudi and Ferreira14,15. With adhesion sutures, the dead space generated by dermal-adipose flap detachment can be reduced while reducing the flap's ability to slide along the aponeurotic plane, thus excluding the 2 major predisposing factors involved in the postoperative development of seroma and hematoma. Strict homeostasis coupled with care to avoid excessive manipulation of the flap and combined surgeries are major approaches, which should be included with adhesion sutures for prophylaxis against seroma formation.
Fixing of the dermal-adipose flap to the aponeurotic plane with adhesion sutures considerably reduces the forces exerted by the flap over the pubic scar and thus decreases the prevalence of dehiscence and badly positioned and extended scars relative to that after the classically performed surgeries. Traction is also an important predisposing factor in hypertrophic scar development. By reducing the tension exerted by the flap over the pubic scar with adhesion sutures, it was possible to observe a lower incidence of hypertrophic scars. The incidences of keloids in surgeries with or without the use of adhesion sutures showed no meaningful statistical differences.
The hospitalization period of patients who undergo body-contouring surgery, previously determined by the volume collected by suction drains, has been shortened, on average, by 24 h with the use of adhesion sutures, enabling early return to normal activities.
The use of adhesion sutures has, on average, increased the time for abdominoplasty by 30 min. Assuming that the benefits provided by adhesion sutures overcome the inconveniences caused by seroma, hematoma, and unsightly scar formation, such an increase in surgical time is justified.
The observation that several plastic surgeons do not use adhesion sutures yet or apply fewer adhesion sutures when they use them is often associated with the need for drainage. The present work reinforces the guidelines suggested by Baroudi and Ferreira, collaborating to augment the teaching and application of their technique.
CONCLUSION
The use of adhesion sutures in body-contouring surgery significantly reduces the incidence of complications such as seroma formation; hematoma formation; wound dehiscence; and misplaced, extended, hypertrophic scarring as well as enables patients to return to their normal activities early.
REFERENCES
1. Avelar JM. Abdominoplasty. Systematization of a technique without external umbilical scar. Aesthetic Plast Surg. 1978;2:141-51.
2. Hakme F. Lipectomia peri e supra-umbilical nas abdominoplastias. Rev Bras Cir. 1979;69:27.
3. Baroudi R. Abdominoplasty: historical review and personal references. Newsletter. Lipoplasty Society North America. 1993;10(3):8.
4. Monard P. La resection esthetique du ventre em tablier avec transplantation de lómbelique. In: Pauchet V, ed. La practique chirurgicale illusttree. vol V. Paris:Gastón Doin;1930.
5. Dufourmentel C, Mouly R. Chirurgie Plastique. Collection medico-chirurgicale. Paris:Flamarion;1959.
6. Glicenstein J. Difficulties of the surgical treatment of abdominal dermodystrophies. Ann Surg Plast. 1975;20(2):147-55.
7. Wilkinson TS, Swarzt BE. Individual modifications in the body contour surgery: the "limited" abdominoplasty. Plast Reconstr Surg. 1986;77(5):779-84.
8. Rebello C, Franco T. Abdominoplasty through a submammary incision. Int Surg. 1977;62(9):462-3.
9. Gonzalez-Ulloa M. Belt lipectomy. Br J Plast Surg. 1960;13:179-86.
10. Floros C, Davis PK. Complications and long-term results following abdominoplasty: a retrospective study. Br J Plast Surg. 1991;44(3):190-4.
11. Antonetti JW, Antonetti AR. Reducing seroma in outpatient abdominoplasty: analysis of 516 consecutive cases. Aesthetic Surg J. 2010;30(3):418-25.
12. Beer GM, Wallner H. Prevention of seroma after abdominoplasty. Aesthet Surg J. 2010;30(3):414-7.
13. Fang RC, Lin SJ, Mustoe TA. Abdominoplasty flap elevation in a more superficial plane: decreasing the need for drains. Plast Reconstr Surg. 2010;125(2):677-82.
14. Baroudi R, Ferreira CA. Seroma: how to avoid it and how to treat it. Aesthet Surg J. 1998;18(6):439-41.
15. Baroudi R, Ferreira CA. Contouring the hip and the abdomen. Clin Plast Surg. 1996;23(4):551-72.
16. Bozola AR, Psillakis JM. Abdominoplasty: a new concept and classification for treatment. Plast Reconstr Surg. 1988;82(6):983-93.
17. Andrades P, Prado A, Danilla S, Guerra C, Benitez S, Sepulveda S, et al. Progressive tension sutures in the prevention of postabdominoplasty seroma: a prospective, randomized, double-blind clinical trial. Plast Reconstr Surg. 2007;120(4):935-46.
18. Matarasso A. Abdominolipoplasty: a system of classification and treatment for combined abdominoplasty and suction-assisted lipectomy. Aesthetic Plast Surg. 1991;15(2):111-21.
19. Vastine VL, Morgan RF, Williams GS, Gampper TJ, Drake DB, Knox LK, et al. Wound complications of abdominoplasty in obese patients. Ann Plast Surg. 1999;42(1):34-9.
20. Nurkim MV, Mendonça LB, Martins PAM, Silva JLB, Martins PDE. Incidência de hematoma e seroma em abdominoplastia com e sem uso de drenos. Rev Soc Bras Plast. 2002;17(1):69-74.
21. Daltrey I, Thomson H, Hussien M, Krishna K, Rayter Z, Winters ZE. Randomized clinical trial of the effect of quilting latissimus dorsi flap donor site on seroma formation. Br J Surg. 2006;93(7):825-30.
22. Moura RMG, Bezerra FJF. Emprego dos pontos de adesão nas reconstruções mamárias com TRAM mais tela sintética. Rev Bras Cir Plast. 2008;23(3):153-7.
23. Nahas FX, Ferreira LM, Ghelfond C. Does quilting suture prevent seroma in abdominoplasty? Plast Reconstr Surg. 2007;119(3):1060-4.
1. Member of the Brazilian Society of Plastic Surgery (BSPS), PhD in Science from Universidade de São Paulo (USP); Faculty Member at Oswaldo Cruz Department of Plastic Surgery and the Brazilian Institute for Research and Education, São Paulo, SP, Brazil.
2. Full Member of the BSPS; Owner of the Plastic Surgery Clinic Dr. Fábio Rosa, São Paulo, SP, Brazil.
3. Full Member of the BSPS, Editor of the Journal of Plastic Surgery, São Paulo, SP, Brazil.
Corresponding author:
Ricardo Frota Boggio
Av. Brigadeiro Luiz Antônio, 3005, casa 5 - Jd. Paulista
São Paulo, SP, Brazil - CEP 01401-000
E-mail: ricardoboggio@ricardoboggio.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Paper received: January 7, 2011
Paper accepted: March 21, 2011
Work performed at the Serviço de Cirurgia Plástica Oswaldo Cruz, Instituto Brasileiro de Pesquisa e Ensino - (Oswaldo Cruz Department of Plastic Surgery, Brazilian Institute for Research and Education), São Paulo, SP, Brazil.
Article submitted for promotion to the status of full member of the BSPS.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter