ABSTRACT
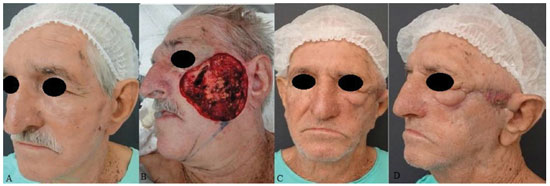
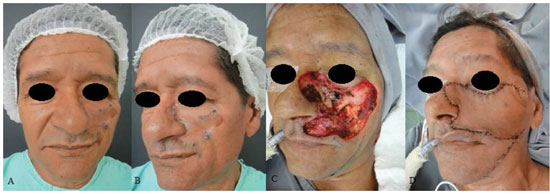
INTRODUCTION: The need for large amounts of skin and subcutaneous tissue makes it complex to repair extensive defects of the middle third of the face. Aiming to reduce morbidity and attain good aesthetic-functional outcomes, Ariyan and McGrath and, subsequently, Behan et al. proposed reconstruction techniques that use transverse and submandibular cervical flap, respectively. Such flaps have fair amounts of tissue and are associated with low morbidity in the donor area. The present authors propose and describe a variant of the Behan flap for use in the reconstruction of large defects of the middle third of the face.
METHODS: We conducted a retrospective study of 8 cases of reconstruction with transverse submandibular cervical flaps for defects of the middle third of the face, conducted between June 2011 and December 2013. The following parameters were analyzed: possible results, and early and late complications.
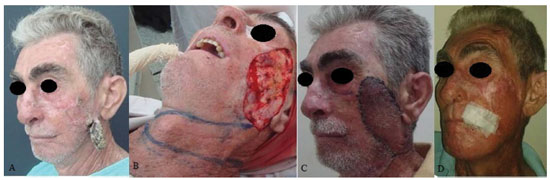
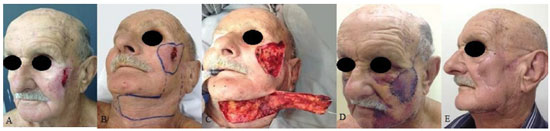
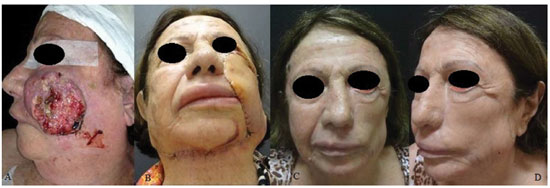
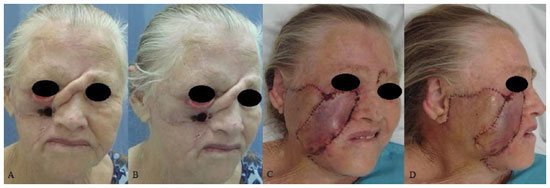
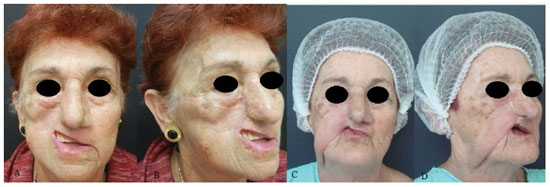
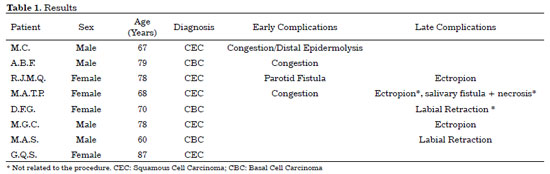
RESULTS: Eight patients with a mean age of 73.5 years were included. All of the patients presented aesthetic-functional satisfactory results. Among the early complications, vascular congestion occurred in 3 patients in the first week with spontaneous resolution, and salivary fistula occurred after tumor resection in 1 patient. With regard to late complications, scar retraction was observed, manifested by either by ectropion (2 patients) or labial retraction (1 patient).
CONCLUSION: Randomized transverse submandibular cervical flaps with axial pedicles may be considered as another option for reconstruction of defects of the middle third of the face.
Keywords: Surgical flaps; Head and neck cancer; Reconstruction; Plastic surgery; Elderly; Reconstructive surgical procedures.