

Original Article - Year 2011 - Volume 26 -
Clinical and dermal thickness assessment 1 year after hyaluronic acid filler treatment
Avaliação clínica e da espessura cutânea um ano após preenchimento de ácido hialurônico
ABSTRACT
Introduction: Few studies have evaluated the durability of tissue filler products in an objective and non-invasive manner. The aim of this study was to evaluate the durability of a formulation of hyaluronic acid (Derma Hyal®) for filling nasolabial folds by comparing perceptions of clinical effect and soft tissue thickness as measured by ultrasound. Methods: Ten women were treated. Clinical and ultrasound evaluation were performed after 1, 3, 6, 9, and 12 months. Results: The average thickness of the nasolabial folds as measured by ultrasound was 0.38 ± 0.14 mm at pretreatment, 0.69 ± 0.19 mm after 1 month, 0.65 ± 0.17 mm after 3 months, 0.61 ± 0.22 mm after 6 months, 0.57 ± 0.23 mm after 9 months, and 0.55 ± 0.14 mm after 12 months. Data analyzed using Friedman's test were not statistically significant. Regarding satisfaction, at 6 months, 3 patients considered the result unsatisfactory; 5, barely satisfactory; and 2, satisfactory. Only 1 patient considered the result satisfactory at 9 months. After 12 months, 1 patient considered the result barely satisfactory and the other patients, unsatisfactory. All patients stated that they would undergo the procedure again. Conclusions: Ultrasound proved to be an objective and non-invasive method of evaluating filling durability, provided that it is correlated with clinical parameters. An increase in skin thickness was demonstrated up to 12 months after injection of the product into the nasolabial fold, followed by a progressive decrease after this period. Although differences among the values as measured by ultrasound were observed over time, these were not statistically significant. Patients perceived a clinical effect up to 9 months after the product was applied.
Keywords: Hyaluronic acid. Ultrasonography. Face.
RESUMO
Introdução: Poucos estudos avaliam a durabilidade dos produtos de preenchimento facial de forma objetiva e não-invasiva. O objetivo deste estudo foi avaliar a durabilidade de formulação de ácido hialurônico (Derma Hyal®) no preenchimento de sulco nasogeniano, comparando percepção do efeito clínico e espessura de partes moles medida por ultrassonografia. Método: Dez mulheres foram tratadas. Avaliação foi realizada após um, três, seis, nove e doze meses, clinicamente e por ultrassonografia. Resultados: A média da espessura à ultrassonografia foi 0,38 ± 0,14 no pré, 0,69 ± 0,19 após 1 mês, 0,65 ± 0,17 após 3 meses, 0,61 ± 0,22 após 6 meses, 0,57 ± 0,23 após 9 meses e 0,55 ± 0,14 após 12 meses. Os dados analisados pelo teste Friedman não se mostraram estatisticamente significativos. Em relação à satisfação, aos 6 meses, 3 pacientes consideraram o resultado insatisfatório, 5, pouco satisfatório e 2, satisfatório. Apenas uma paciente considerou o resultado satisfatório aos 9 meses. Aos 12 meses, uma considerou pouco satisfatório e as demais, insatisfatório. Todas responderam que realizariam novamente o procedimento. Conclusões: A ultrassonografia demonstrou ser método objetivo e não-invasivo na avaliação da durabilidade de preenchimento, desde que avaliado com correlação clínica. Demonstrou-se aumento da espessura cutânea até 12 meses após injeção em sulco nasogeniano (SNG), com decréscimo progressivo. Embora exista diferença dos valores da ultrassonografia ao longo do tempo, essas não foram estatisticamente significantes. Clinicamente, o efeito percebido pelas pacientes se deu até o nono mês após aplicação do produto avaliado.
Palavras-chave: Ácido hialurônico. Ultrassonografia. Face.
Hyaluronic acid is a glycosaminoglycan polysaccharide found in the extracellular matrix of the skin, the connective tissue, and the vitreous humor. It is responsible for hydration, lubrication, and stabilization of these structures1,2. It represents an alternative in the treatment of facial aging and has been used for more than a decade to fill soft tissue to correct depressions, wrinkles, and folds. Owing to histological studies, its biological behavior is well known, and it is absorbed gradually over a period of months3-6. Its potential complications are uncommon and self-limiting and include inflammatory reactions, small hematomas and/or ecchymosis, abscesses at the site of application, tissue necrosis (by intravenous injection or compression of the adjacent vascular network), persistent edema, and granulomas1,6. Persistent edema and granulomas can result from allergy to the material or from an immune response to protein components present in preparations of hyaluronic acid and can be treated with local injection of hyaluronidase2,7.
Currently there are a large number of commercially available products, but only a few long-term scientific studies have described the actual durability of each product or made comparisons among them8-10.
Ultrasound, used in several studies to measure skin thickness in healthy individuals and those with sequelae of burns, has proven to be an advantageous method because it can be used on any part of the body, it does not use radiation, it is inexpensive, and there is high reproducibility and low variability between observers10-14.
At our institution, ultrasound of soft tissues was performed using an ATL Philips HD 5000 (Milwaukee, WI, USA) with a 14-Mhz small parts probe.
The nasolabial folds of 2 groups of patients were filled with 2 formulations of hyaluronic acid from the same manufacturer, with different molecular weights. After 6 months, the 800,000 daltons group showed less permanence of the product than did the 1,550,000 daltons group. The results confirmed that ultrasound is an efficient, objective, and non-invasive method of evaluating facial fillings and convinced the product manufacturer to replace the lower molecular weight formula with the new formulation15.
The aim of this study was to evaluate the durability of the current formulation of hyaluronic acid (Derma Hyal®) 12 months after the filling of nasolabial folds by comparing the perceptions of clinical effect and soft tissue thickness as measured by ultrasound.
METHODS
We selected 10 patients aged 38 to 55 (mean = 49.1 years) who complained of accentuation of the nasolabial folds caused by facial aging. We excluded patients with previous local ambulatory or surgical treatment, uncontrolled systemic disease, or allergy to any component of the product used. All patients signed an informed consent form approved by the institution.
A volume of 1 ml of hyaluronic acid at 3% (Derma Hyal®; Oft Vision, São Paulo, Brazil) with a molecular weight of 1,550,000 daltons was injected by deep intradermal injection in each nasolabial fold under aseptic technique.
Ultrasound of soft tissue was performed before the procedure and after 1, 3, 6, 9, and 12 months. Photographs were taken, and a subjective assessment of satisfaction was made. The examination was always performed by the same radiologist, with the patient's head deflected at 45°. The dermal-epidermal thickness was surveyed at a standardized point on the nasolabial fold 1.0 cm from the insertion of the nasal wing. At each evaluation, 3 measurements were taken from each side of the patient's face; the final value was the average of the 3 measurements.
Patients rated their satisfaction with the procedure as follows: 0 = unsatisfactory (no change from pretreatment), 1 = barely satisfactory (little improvement), 2 = satisfactory, 3 = very satisfactory (Figures 1 and 2).
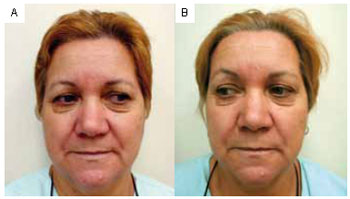
Figure 1 - A: Pretreatment. Frontal view. B: Six months posttreatment.
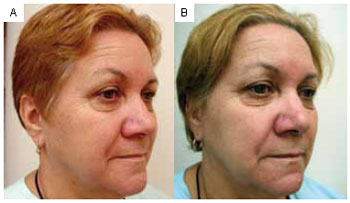
Figure 2 - A: Pretreatment. Right oblique profile. B: Six months posttreatment.
RESULTS
Among the 10 patients treated, only 1 (10%) presented with unilateral hematoma and 5 (50%) with slight edema after the procedure that completely resolved conservatively within 10 days.
The data were analyzed using Friedman's nonparametric test in order to compare the ultrasound results at different time points. The significance level was 0.05 (5%), and confidence intervals were 95%.
The average thickness of the 20 nasolabial folds analyzed was 0.38 ± 0.14 mm at pretreatment, 0.69 ± 0.19 mm after 1 month, 0.65 ± 0.17 mm after 3 months, 0.61 ± 0.22 mm after 6 months, 0.57 ± 0.23 mm after 9 months, and 0.55 ± 0.14 mm after 12 months (Figure 3).
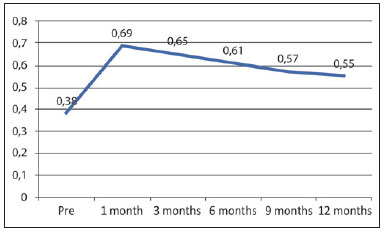
Figure 3 - Average dermal thickness (in millimeters) of the nasolabial fold over time as measured by ultrasound.
Among the 10 patients, 3 evaluated the results after 6 months as unsatisfactory; 5, as barely satisfactory; and 2, as satisfactory. Only 1 patient considered the treatment satisfactory after 9 months.
After 12 months, 1 patient considered the result barely satisfactory, and the others considered it unsatisfactory or saw no difference compared to pretreatment. All stated that they would undergo the procedure again (Figure 4).
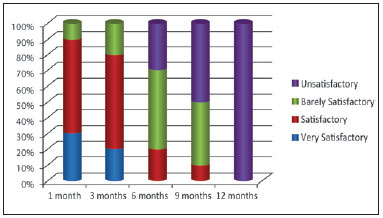
Figure 4 - Evaluation of patient satisfaction over time.
DISCUSSION
The dermal aging process is divided into intrinsic or "chronological" aging, starting at the age of about 30 years, and extrinsic aging or "photoaging." Intrinsic aging, which causes atrophy of the dermal components, is characterized by skin with a thin, fragile, inelastic, flaccid, and dry appearance that has an impaired ability to repair itself and has a predisposition to benign neoplasms. Extrinsic aging, mostly caused by ultraviolet radiation, involves superimposed alterations that lead to skin dryness and roughness; telangiectasias; rhytids of greater depth; and skin tumors, including malignant neoplasms16.
Wrinkles can be categorized as orthostatic, dynamic, static, and gravitational, the latter being caused by tissue descending from adhesion points, as in the case of nasolabial folds16.
An increasing number of non-invasive or minimally invasive ambulatory procedures have been used for the treatment and mitigation of various types of facial rhytids. Hyaluronic acid is an absorbable substance that is often used in clinical practice to fill wrinkles and folds3-6, 8-10. There are several products on the market, and they vary in terms of the durability of the clinical effect. There are no scientific comparisons of these products8.
Other treatment options for filling out wrinkles, folds, and facial depressions include polylactic acid, which is expensive and requires various applications, and fat grafting. The main advantage of fat grafting is the use of autologous material and the possibility of using more material as compared to other filling products17. Fat grafting has the following disadvantages: the procedure must be performed in a surgical environment; the results are unpredictable and vary with the degree of absorption; and the procedure is associated with possible complications such as infection, embolism, cyst formation, calcification, ossification, pseudotumors, amaurosis, fat necrosis, skin necrosis, hypercorrections, and asymmetries. In addition, the patient's body weight is a major limiting factor18,19.
This study used ultrasound to evaluate the durability of Derma Hyal®, hyaluronic acid at a concentration of 3%. In doing so, we could demonstrate the permanence of the product in the deep dermis over 12 months using an objective and non-invasive method.
Ultrasound has previously been used to evaluate the efficacy, safety, and durability of polylactic acid in HIV-positive patients with facial lipoatrophy20.
Despite the increased thickness of the soft parts observed using ultrasound after 6 months of treatment, this finding did not correlate with the patients' perceptions of clinical effect. The scores given by the patients reflected their discontent and corresponded to the subjective perceptions of the medical team, although these were not officially recorded in this study. This discontent may be explained by patients' high expectations in relation to the results of facial rejuvenation treatments. Better rejuvenation results are obtained with various other treatments, including botulinum toxin, skin care, and replenishment of facial volume. The plastic surgeon's knowledge in this area is essential for enhancing the results of facial surgeries.
CONCLUSIONS
Ultrasound proved to be an objective and non-invasive method of evaluating filling durability, provided that it is correlated with clinical parameters. An increase in skin thickness was apparent up to 12 months after injection of the product into the nasolabial fold, with a progressive decrease. Although there were differences among the values measured at different time points, these were not statistically significant. Patients perceived a clinical effect up to 9 months after the product was applied.
REFERENCES
1. Alster TS, West TB. Human-derived and new synthetic injectable materials for soft-tissue augmentation: current status and role in cosmetic surgery. Plast Reconst Surg. 2000;105(7):2515-25.
2. Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005;31(8 Pt 1):893-7.
3. Piacquadio D, Jarcho M, Goltz R. Evaluation of hylan b gel as a soft-tissue augmention implant material. J Am Acad Dermatol. 1997;36(4):544-9.
4. Duranti F, Salti G, Bovani B, Calandra M, Rosati ML. Injectable hyaluronic acid gel for soft tissue augmentation. A clinical and histological study. Dermatol Surg. 1998;24(12):1317-25.
5. Olenius M. The first clinical study using a new biodegradable implant for the treatment of lips, wrinkles, and folds. Aesthetic Plast Surg. 1998;22(2):97-101.
6. Lowe NJ, Maxwell A, Lowe P, Duick MG, Shah K. Hyaluronic acid skin fillers: adverse reactions and skin testing. J Am Acad Dermatol. 2001;6(45):930-3.
7. Cox SE. Clinical experience with filler complications. Dermatol Surg. 2009;35(Suppl 2):1661-6.
8. Alster TS, West TB. New options for soft tissue augmentation. Skin Aging 1998;32-6.
9. Larsen NE, Pollak CT, Reiner K, Leshchiner E, Balazs EA. Hylan gel biomaterial: dermal and immunologic compatibility. J Biomed Mar Res. 1993;27(9):1129-34.
10. Alexander H, Miller DL. Determining skin thickness with pulsed ultrasound. J Invest Dermatol. 1979;72(1):17-9.
11. Tan CY, Statham B, Marks R, Payne PA. Skin thickness measurement by pulsed ultrasound: its reproducibility, validation and variability. Br J Dermatol. 1982;106(6):657-67.
12. Kirsch JM, Hanson ME, Gibson JR. The determination of skin thickness using conventional diagnostic ultrasound equipment. Clin Exp Dermatol. 1984;9(3):280-5.
13. Van den Kerckhove E, Staes F, Flour M, Stappaerts K, Boeckx W. Reproducibility of repeated measurements on post-burn scars with Dermascan C. Skin Res Technol. 2003;9(1):81-4.
14. Hambleton J, Shakespeare PG, Pratt BJ. The progress of hypertrophic scars monitored by ultrasound measurements of thickness. Burns. 1992;18(4):301-7.
15. Salles AG, Remigio AFN, Saito OC, Camargo CP, Zacchi VB, Saito PL, et al. Avaliação da durabilidade de preenchimento de ácido hialurônico com ultra-som facial. Arq Cat Med. 2009;38(supl 1):281-3.
16. Borelli S, Pellegrino DG, Mapeli AB. O envelhecimento da face: como e por quê envelhecemos. In: Melega JM, Baroudi R, eds. Cirurgia Plástica Fundamentos e Arte: cirurgia estética. Rio de Janeiro:Medsi;2003. p.9-15.
17. Coleman SR. Structural fat grafting. 1ª ed. St. Louis:QMP;2004.
18. Khawaja HA, Hernández-Pérez E. Fat transfer review: controversies, complications, their prevention, and treatment. Intern J Cosm Surg Aesth Dermatol. 2002;4(2):131-8.
19. Lam SM, Glasgold RA, Glasgold MJ. Limitations, complications, and long-term sequelae of fat transfer. Facial Plast Surg Clin North Am. 2008;16(4):391-9.
20. Valantin MA, Aubron-Olivier C, Ghosn J, Laglenne E, Pauchard M, Schoen H, et al. Polylactic acid implants (New-Fill) to correct facial lipoatrophy in HIV-infected patients: results of the open-label study VEGA. AIDS. 2003;17(17):2471-7.
1. PhD in Plastic Surgery; Coordinator of the Cosmiatry Group of the Plastic Surgery Department at the Medical College of the University of São Paulo (FMUSP), São Paulo, SP, Brazil.
2. Full Member of the Brazilian Society of Plastic Surgery (BSPS), Medical Researcher in the Plastic Surgery Discipline, FMUSP, São Paulo, SP, Brazil.
3. Specialist Member of the BSPS, Medical Researcher in the Plastic Surgery Discipline, FMUSP, São Paulo, SP, Brazil.
4. PhD in Radiology, Assistant in the Radiology Institute at FMUSP Clinical Hospital, São Paulo, SP, Brazil.
5. Full Professor of the Plastic Surgery Discipline at FMUSP; Head of Plastic Surgery Service at FMUSP Clinical Hospital, São Paulo, SP, Brazil.
Corresponding author:
Alessandra Grassi Salles
Rua Joaquim Floriano, 466 - cj. 2102 6 - Itaim
São Paulo, SP, Brazil - CEP 04534-002
E-mail: agsalles@uol.com.br
Submitted to SGP (Sistema de Gestão de Publicações/ Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica / Brazilian Journal of Plastic Surgery).
Received: November 26, 2010
Accepted: February 5, 2011
Work was performed at the Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo - (Clinical Hospital of the Medical College of the University of São Paulo - FMUSP), São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter