

Original Article - Year 2026 - Volume 41Issue 1
Histopathological Evaluation of Inflammatory Response and Capsular Formation with Smooth, Textured, and Polyurethane Implants in Rats
Avaliação histopatológica da resposta inflamatória e formação capsular com implantes lisos, texturizados e de poliuretano em ratos
ABSTRACT
Introduction Silicone breast prostheses, designed in the 1960s, have undergone several modifications to minimize the inflammatory response and capsular contracture. Biomaterial research enhances the understanding of the interaction between implants and tissues, providing insights into innovation, repair, regeneration, and capsular contracture.
Objective Materials and
Methods Ten female Wistar rats, approximately 3 months old and weighing from 200 to 250 grams, were divided into 2 groups. Each rat received three implants, one of each type. A pathologist blinded to the origin of the samples performed the histopathological analysis. There was no statistically significant difference (p > 0.05) in acute inflammation between the implants. For chronic inflammation, textured and polyurethane implants differed significantly (p < 0.05) from smooth ones. Foreign body reactions were more common in polyurethane implants compared with smooth and textured implants (p < 0.05). There was no statistical difference regarding granulation tissue and fibrosis (p > 0.05).
Conclusion Compared with smooth and textured implants, polyurethane-coated implants increased the occurrence of foreign body reactions, led to more intense chronic inflammation, and resulted in a greater amount of granulation tissue.
Keywords: biocompatible materials; breast implantation; clinical trials as topic; implant capsular contracture; inflammation
RESUMO
Introdução As próteses mamárias de silicone, idealizadas na década de 1960, passaram por diversas modificações para minimizar a resposta inflamatória e a contratura capsular. Pesquisas em biomateriais possibilitam melhor compreensão da interação entre o implante e o tecido, especificada por inovação, reparo, regeneração e contratura capsular.
Objetivo Avaliar a resposta biológica de diferentes implantes: liso, texturizado e revestido de poliuretano, identificar complicações associadas e comparar a formação capsular pós-operatória em animais submetidos aos implantes.
Materiais e Métodos Foram utilizados 10 ratos Wistar, fêmeas, com aproximadamente 3 meses e peso entre 200 e 250 gramas, divididos em 2 grupos. Cada rato recebeu três implantes, um de cada tipo. Foram realizadas análise histopatológica, por um patologista cego à origem das amostras. Não houve diferença estatística relevante (p>0,05) na inflamação aguda entre os implantes. Para inflamação crônica, os implantes texturizados e poliuretano diferiram significativamente (p<0,05) dos lisos. A ocorrência de corpo estranho foimaior em implantes de poliuretano em comparação com lisos e texturizados (p<0,05). Não houve diferença estatística para tecido de granulação e fibrose (p>0,05).
Conclusão Em comparação com os implantes lisos e texturizados, os implantes revestidos por poliuretano causaram maior ocorrência de corpo estranho, maior inflamação crônica, com maior presença de tecido de granulação.
Palavras-chave: contratura capsular em implantes; ensaio clínico; implante mamário; inflamação; materiais biocompatíveis
Introduction
Silicone breast implants, developed by Cronin and Gerow in the 1960s, have undergone numerous modifications to minimize adverse effects and improve outcomes.1 Capsular contractureisthemost commonsideeffect of breast implants, and the complication most avoided by plastic surgeons.2 It is a foreign body reaction with scar tissue formation around the breast implant. Histologically, capsular contracture consists of a multilayered structure composed of fibroblasts, fibrocytes, myofibroblasts, and histiocytes surrounded by acellular tissue rich in collagen fibers.2
The materials and textures used in implants are strongly related to the inflammatory response and the degree of capsular contracture. In the Baker classification (1981), contracture grades range from I to IV depending on symptoms and findings at the physical examination of the breasts.3
Studies on capsular contracture indicated different inflammatory responses to smooth and textured implants. However, analyses on polyurethane-coated implants remain scarce.4
An attempt to reduce capsular contracture led to the development of textured surface prostheses; however, the intensity of the inflammatory reaction around the prosthesis did not change significantly.4 Implants covered with polyurethane foam, introduced in the 1970s, emerged as an alternative to minimize inflammation and contracture induced by smooth and textured implants.5
The Scales’ criteria (1953) define an ideal implant as inert, not causing adverse reactions or carcinogenesis, withstanding mechanical forces, and maintaining tissue integrity.5
It is essential to understand the histopathological interaction between the tissue and the implant coating to assess the risk of chronic inflammation or immunological events, which can directly influence the safety and durability of the material. The selection of the coating type is a key factor in the body’s biological response, potentially impacting the formation of the fibrous capsule around the implant and the occurrence of complications, such as capsular contracture.6
Ongoing studies are crucial in understanding the histopathological interaction between the tissue and the implant coating, to assess the risk of chronic inflammation or reaction, and to identify materials and surfaces that minimize capsular contractures and inflammatory conditions.7
Objective
Main objective: To perform a histological assessment of the inflammatory response and capsular contracture induced by smooth, textured, and polyurethane-coated implants in the backs of Wistar rats.
Materials and Methods
Experimental animals: Ten female Wistar rats, approximately 3 months old and weighing between 200 and 250 g.
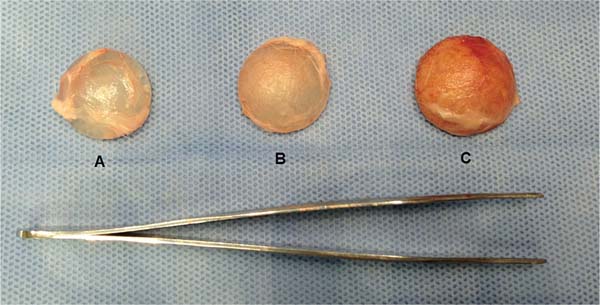
Study design: The study compared the inflammatory response and capsular formation of three types of silicone implants, namely, smooth (A), textured (B), and polyurethane-coated (C). We divided the animals into 2 groups (n = 5), and each one received one implant of each type. Histopathological analysis occurred after four months.
Experimentation material: We used the following discshaped silicone implants supplied by Silimed (Silimed Indústria de Implantes Ltda):
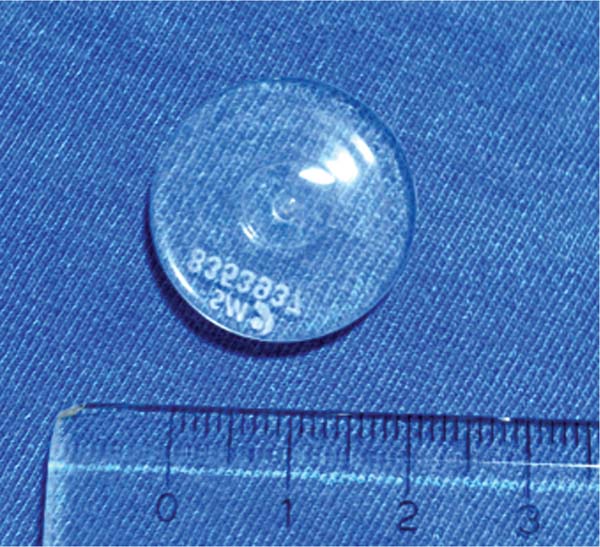
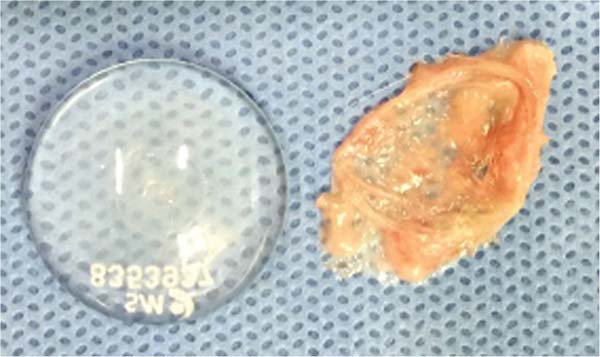
Smooth implant: 2.3 cm diameter x 0.4 cm thickness, 3.18 cm3 (►Fig. 1).
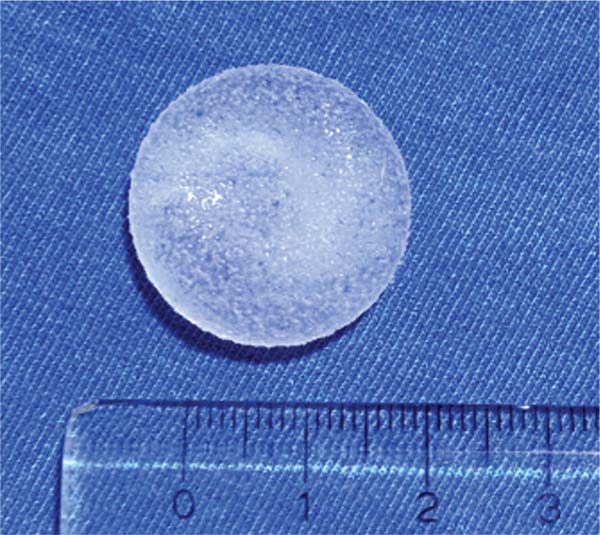
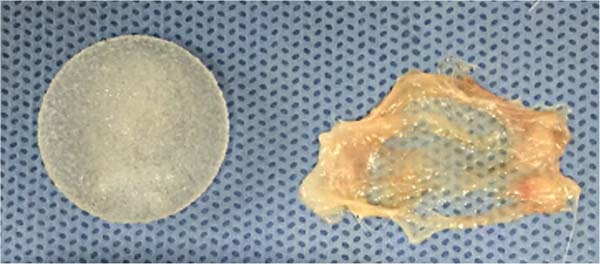
Textured implant: 2.4 cm diameter x 0.7 cm thickness, 3.61 cm3 (►Fig. 2).
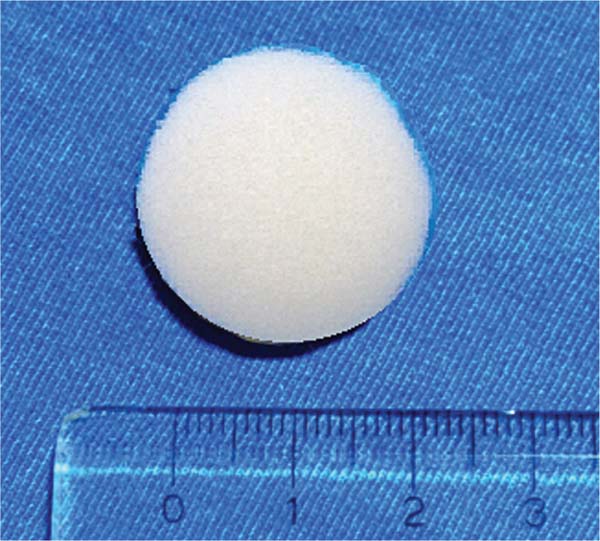
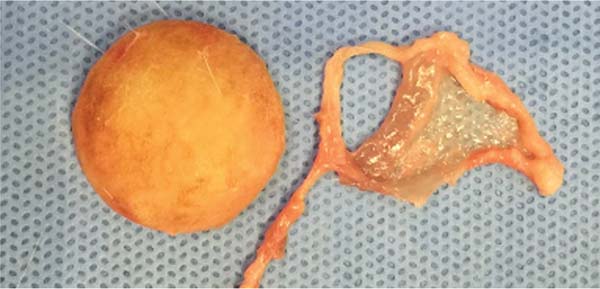
Polyurethane-coated implant: 2.5 cm diameter x 0.9 cm thickness, 4.0 cm3 (►Fig. 3)
All implants underwent sterilization with ethylene oxide.
Surgical procedure
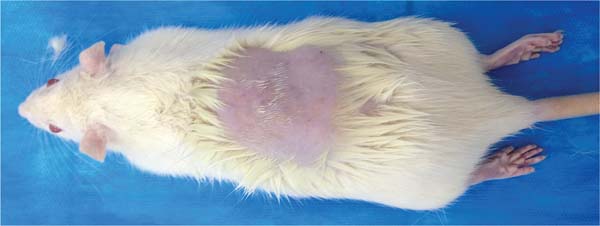
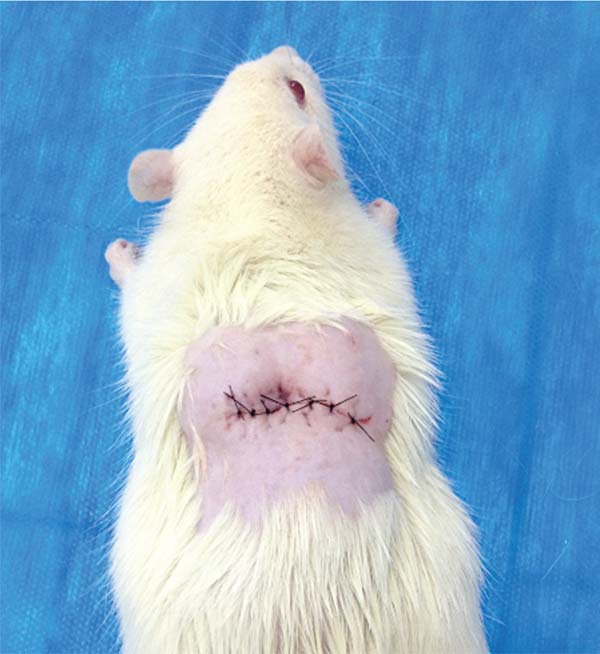
Anesthesia: Anesthesia consisted of 10% ketamine (40 mg/kg) and 2% xylazine (5 mg/kg) via the intramuscular route. The duration of anesthesia was approximately 3 hours. After anesthesia, we proceeded to the trichotomy of the dorsal region of each rat (►Fig. 4).
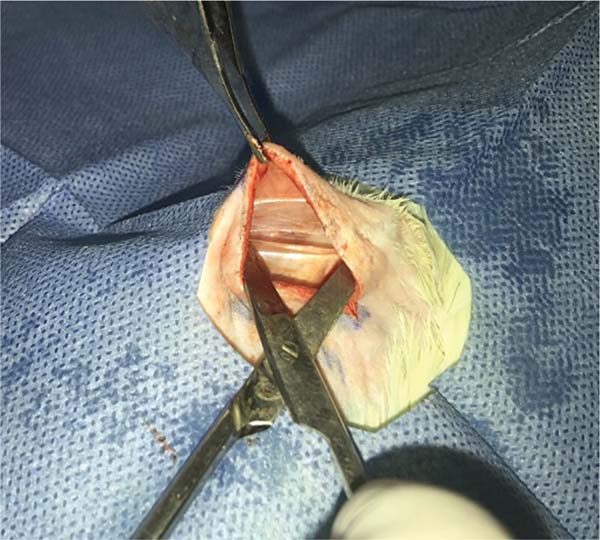
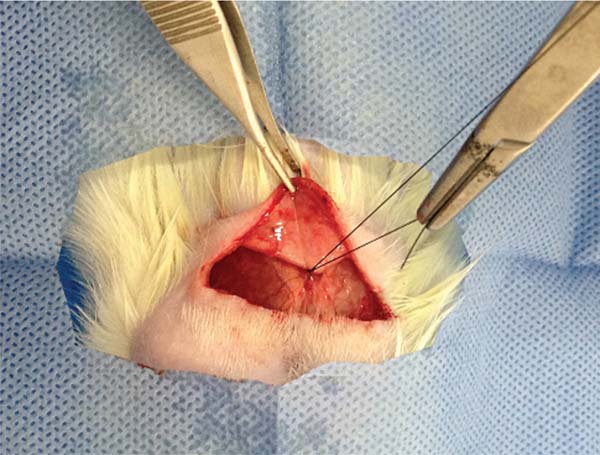
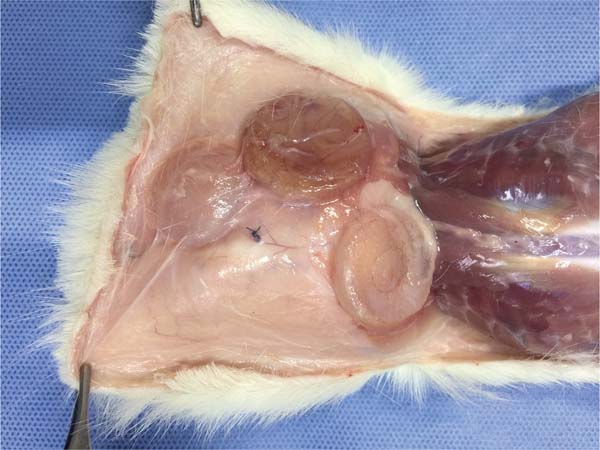
Implant insertion: We made a 3-cm transverse incision in the back of each rat, creating three subcutaneous pockets for smooth, textured, and polyurethane-coated implants (►Figs. 5-6).
After implant insertion, we performed a simple suture between the subcutaneous pockets to prevent the implants from overlapping (►Fig. 7).
Lastly, we performed skin closure with simple 4-0 nylon sutures (►Fig. 8).
Implant removal: After 4 months, the animals underwent another surgery with the same anesthetic protocol and euthanasia with a lethal dose of ketamine and xylazine.
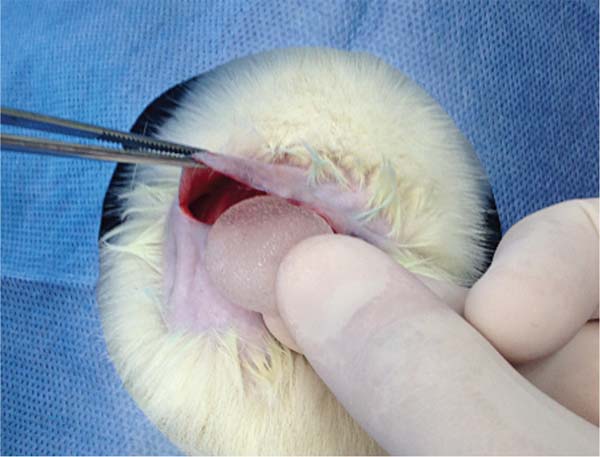
Transverse and horizontal incisions on the back allowed implant visualization (►Fig. 9). The implants remained in their respective subcutaneous pockets. We dissected each implant separately to obtain the capsule surrounding it. The surgical margins of the histological specimens constitute the ridges of the implants, and resection occurred from the dermis to the muscle plane (►Fig. 10).
Histopathological analysis
We removed the membranes covering the implants (►Figs. 11-13) and fixed them in 10% formalin for future histopathological analysis. Each sample received a code corresponding to the rat number and the type of implant (e.g., 1A: first rat, smooth implant; 1B: first rat, textured implant; 1C: first rat, polyurethane-coated implant).
Complications: Two animals exhibited an intense reaction and extruded both the smooth and textured implants; as such, we excluded them from the study. The polyurethanecoated implants remained, allowing for analysis of the remaining membrane.
Results
We converted the histopathological assessment results into numerical values for statistical analysis of the variables (►Table 1).
| 95%CI | ||||||
|---|---|---|---|---|---|---|
| Variable | Comparison | Mean difference | Standard error | p | Lower | Upper |
| Refractile material | Polyurethane-coated x smooth | 0.600 | 0.207 | 0.022 | 0.081 | 1.119 |
| Textured x smooth | 0.000 | 0.219 | 1.000 | -0.547 | 0.547 | |
| Textured x polyurethane-coated | -0.600 | 0.207 | 0.022 | -1.119 | -0.081 | |
| Lymphocytes | Polyurethane-coated x smooth | 0.300 | 0.188 | 0.266 | -0.170 | 0.770 |
| Textured x smooth | -0.500 | 0.198 | 0.048 | -0.995 | -0.005 | |
| Textured x polyurethane-coated | -0.800 | 0.188 | 0.001 | 1.270 | -0.330 | |
| Neutrophils | Polyurethane-coated x smooth | -0.350 | 0.325 | 0.537 | -1.163 | 0.463 |
| Textured x smooth | -0.625 | 0.342 | 0.183 | -1.482 | 0.232 | |
| Textured x polyurethane-coated | -0.275 | 0.325 | 0.678 | -1.088 | 0.538 | |
| Macrophages | Polyurethane-coated x smooth | -0.025 | 0.224 | 0.993 | -0.587 | 0.537 |
| Textured x smooth | -0.750 | 0.237 | 0.011 | -1.342 | -0.158 | |
| Textured x polyurethane-coated | -0.725 | 0.224 | 0.010 | -1.287 | -0.163 | |
| Giant cells | Polyurethane-coated x smooth | 1.250 | 0.305 | 0.001 | 0.487 | 2.013 |
| Textured x smooth | 0.000 | 0.321 | 1.000 | -0.804 | 0.804 | |
| Textured x polyurethane-coated | -1.250 | 0.305 | 0.001 | -2.013 | -0.487 | |
| Granulation tissue | Polyurethane-coated x smooth | -0.125 | 0.293 | 0.905 | -0.857 | 0.607 |
| Textured x smooth | -0.750 | 0.308 | 0.058 | -1.522 | 0.022 | |
| Textured x polyurethane-coated | -0.625 | 0.293 | 0.105 | -1.357 | 0.107 | |
| Fibrosis | Polyurethane-coated x smooth | 0.125 | 0.131 | 0.612 | -0.203 | 0.453 |
| Textured x smooth | 0.250 | 0.138 | 0.188 | -0.095 | 0.595 | |
| Textured x polyurethane-coated | 0.125 | 0.131 | 0.612 | -0.203 | 0.453 | |
Note: Values of p in bold indicate statistical significance.
We compared each type of implant (smooth, textured, and polyurethane-coated) with each variable (refractive material, lymphocytes, neutrophils, macrophages, giant cells, granulation tissue, and fibrosis). The variables refractile material, lymphocytes, macrophages, and giant cells showed a significant difference (p < 0.05) in comparison with each implant type (smooth, textured, and polyurethane-coated). However, there were no statistically significant differences (p > 0.05) between the variables neutrophils, granulation tissue, and fibrosis regarding each implant.
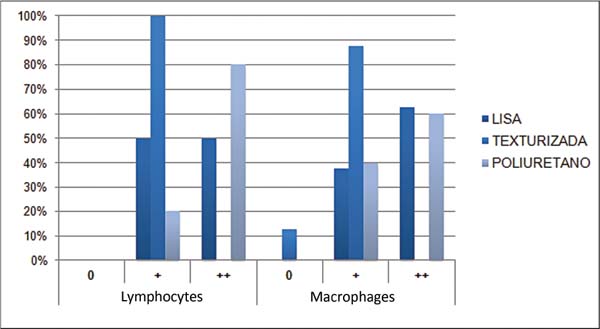
We determined the development of acute inflammation according to the presence of neutrophils. The histopathological analysis revealed acute inflammation in 37.5% of the rats with smooth implants, 12.5% with textured implants, and 40% with polyurethane-coated implants.
Table 2 reveals no statistically significant difference (p > 0.05) between implant types.
| 95%CI | ||||||
|---|---|---|---|---|---|---|
| Variable | Comparison | Mean difference | Standard error | p | Lower | Upper |
| Neutrophils | Polyurethane-coated x smooth | -0.350 | 0.325 | 0.537 | -1.163 | 0.463 |
| Textured x smooth | -0.625 | 0.342 | 0.183 | -1.482 | 0.232 | |
| Textured x polyurethane-coated | -0.275 | 0.325 | 0.678 | -1.088 | 0.538 | |
Note: Values of p in bold indicate statistical significance.
We defined chronic inflammation as the presence of lymphocytes and macrophages (►Fig. 14). Cell quantification was performed using crosses (þ), according to which zero (0) indicated the absence of cells and 2 crosses (þþ) designated a moderate presence. No implant type exhibited an intense cell presence (þþþ).
Table 3 shows that there was no statistically significant difference (p > 0.05) between smooth and polyurethane implants. However, there was a statistically significant difference (p < 0.05) when comparing textured with smooth and polyurethane-coated implants.
| 95%CI | ||||||
|---|---|---|---|---|---|---|
| Variable | Comparison | Mean difference | Standard error | p | Lower | Upper |
| Lymphocytes | Polyurethane-coated x smooth | 0.300 | 0.188 | 0.266 | -0.170 | 0.770 |
| Textured x Smooth | -0.500 | 0.198 | 0.048 | -0.995 | -0.005 | |
| Textured x polyurethane-coated | -0.800 | 0.188 | 0.001 | 1.270 | -0.330 | |
| Macrophages | Polyurethane-coated x smooth | -0.025 | 0.224 | 0.993 | -0.587 | 0.537 |
| Textured x smooth | -0.750 | 0.237 | 0.011 | -1.342 | -0.158 | |
| Textured x polyurethane-coated | -0.725 | 0.224 | 0.010 | -1.287 | -0.163 | |
Note: Values of p in bold indicate statistical significance.
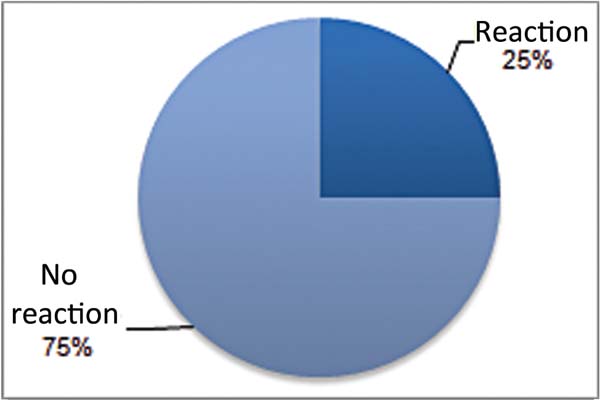
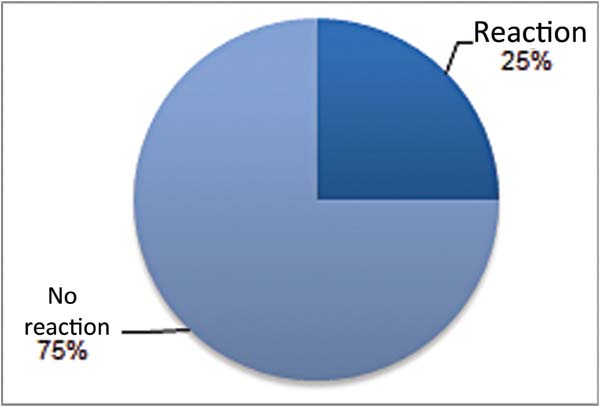
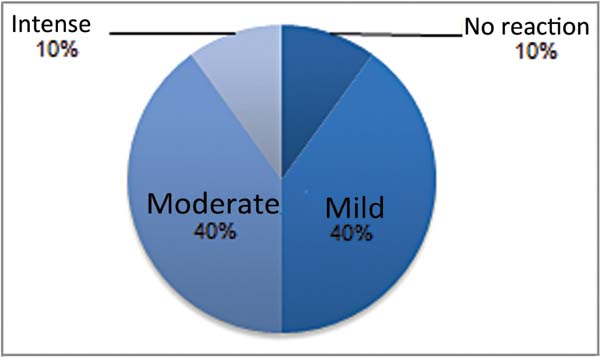
Foreign body reactions were defined by the presence of giant cells. Only 25.0% of rats with smooth implants showed a foreign body reaction (►Fig. 15). The same occurred with textured implants, as 25.0% of rats presented a foreign body reaction (►Fig. 16). Regarding polyurethane-coated implants, foreign body reactions were absent at 10.0% of rats, mild in 40.0%, moderate in 40.0%, and intense in 10.0% (►Fig. 17).
Table 4 reveals that there was no statistically significant difference (p > 0.05) when comparing smooth and textured implants. The comparison between smooth versus polyurethane-coated implants and textured versus polyurethane-coated implants found no statistically significant difference (p < 0.05).
| 95%CI | ||||||
|---|---|---|---|---|---|---|
| Variable | Comparison | Mean difference | Standard error | p | Lower | Upper |
| Foreign body reaction | Polyurethane-coated x smooth | 1.250 | 0.305 | 0.001 | 0.487 | 2.013 |
| Textured x smooth | 0.000 | 0.321 | 1.000 | -0.804 | 0.804 | |
| Textured x polyurethane-coated | -1.250 | 0.305 | 0.001 | -2.013 | -0.487 | |
Note: Values of p in bold indicate statistical significance.
Fibrosis was categorized as mild, moderate, or intense. Smooth implants predominantly presented with moderate fibrosis (87.5%), while textured implants showed 87.5% moderate fibrosis and 12.5% intense fibrosis. Polyurethanecoated implants demonstrated 100% moderate fibrosis. Granulation tissue underwent the same categorization. In smooth implants, moderate and intense forms predominated (37.5% each). In textured implants, most forms were mild (62.5%).
Table 5 indicates no statistically significant differences (p > 0.05) when analyzing the presence of granulation tissue and fibrosis in the three implants studied.
| 95%CI | ||||||
|---|---|---|---|---|---|---|
| Variable | Comparison | Mean difference | Standard error | p | Lower | Upper |
| Granulation tissue | Polyurethane-coated x smooth | -0.125 | 0.293 | 0.905 | -0.857 | 0.607 |
| Textured x smooth | -0.750 | 0.308 | 0.058 | -1.522 | 0.022 | |
| Textured x polyurethane-coated | -0.625 | 0.293 | 0.105 | -1.357 | 0.107 | |
| Fibrosis | Polyurethane-coated x smooth | 0.125 | 0.131 | 0.612 | -0.203 | 0.453 |
| Textured x smooth | 0.250 | 0.138 | 0.188 | -0.095 | 0.595 | |
| Textured x polyurethane-coated | 0.125 | 0.131 | 0.612 | -0.203 | 0.453 | |
Note: Values of p in bold indicate statistical significance.
The refractile material corresponded to the presence of implant residue. Histopathological analysis concluded that only the samples from the polyurethane-coated implants presented implant residue, corresponding to 50% of the samples in group C. This finding demonstrates that the polyurethane coating triggers an intense inflammatory response, which leads to capsule formation around the implant to integrate with the coating.
Table 6 shows that there was no statistically significant difference (p > 0.05) when comparing smooth and textured implants. Comparison of smooth versus polyurethane-coated implants and textured versus polyurethane-coated implants revealed a statisticallysignificantdifference (p < 0.05).
| 95%CI | ||||||
|---|---|---|---|---|---|---|
| Variable | Comparison | Mean difference | Standard error | p | Lower | Upper |
| Refractile material | Polyurethane-coated x smooth | 0.600 | 0.207 | 0.022 | 0.081 | 1.119 |
| Textured x smooth | 0.000 | 0.219 | 1.000 | -0.547 | 0.547 | |
| Textured x polyurethane-coated | -0.600 | 0.207 | 0.022 | -1.119 | -0.081 | |
Note: Values of p in bold indicate statistical significance.
Discussion
The choice of the experimental animal, the Wistar rat, was based on its ease of acquisition and handling, adaptation to the artificial environment, and the possibility of placing three implants in the same animal. Its dimensions do not interfere with the analysis of macroscopic and microscopic characteristics.8
Several researchers have used rodents in experiments with silicone implants.9 A recent study reported that the tissue reaction caused by silicone breast implants leads to the formation of a periprosthetic capsule, which is similar in experimental animals and in humans.10
In this study, polyurethane-coated implants exhibited an intense acute inflammatory reaction. A large number of giant cells partially engulfed the implant material, indicating a foreign body inflammatory process characteristic of this material, consistent with several similar studies.9 This acute inflammatory reaction was observed late, characterizing a chronic inflammation with the presence of lymphocytes and macrophages.
The presence of refractile material (implant residue) only in samples from polyurethane-coated implants indicates a greater inflammatory response compared with other implants (i.e., smooth and textured), which did not present refractile material in the histopathological analysis. It is worth noting that the polyurethane coating integrates with the membrane surrounding the implant, resulting in a more intense inflammatory response.11,12
Therefore, compared with smooth and textured implants, the polyurethane-coated implant induced greater foreign body reactions, acute and chronic inflammation, and granulation tissue formation.
Conclusion
In the present study, after comparing the inflammatory reaction caused by the three types of implants (smooth, textured, and polyurethane-coated), it was concluded that:
a) The polyurethane-coated implant caused more acute inflammation when compared with smooth and textured implants;
b) Chronic inflammation was greater in polyurethane-coated implants and lower in textured implants.
c) Polyurethane-coated implants elicited a foreign body reaction in 90% of the rats, while smooth and textured implants did it in only 25% of the rats.
d) The fibrosis caused by smooth, textured, and polyurethane-coated implants was virtually the same in all cases. Granulation tissue around the polyurethane-coated implants was in moderate quantity in 80% of rats, indicating a greater inflammatory reaction.
e) The polyurethane-coated implants were the only ones that presented difficulty in removing the membrane that surrounded them. This fact was proven by histopathological analysis as they were the only samples with refractile material, that is, remnants of the polyurethane coating.
References
1. Monteiro Ll, Mangiavacchi W, Machado DG. A evolução das próteses mamárias e os métodos de incisão utilizados em procedimentos de mamoplastia de aumento. Rev Bras Cir Plást 2022;37 (01):125-131. Doi: 10.5935/2177-1235.2022RBCP0022
2. Bozola AR. Passado, presente e futuro utilizando implantes mamários de silicone no Brasil, um relato de 45 anos. Rev Bras Cir Plást 2020;35:505-513. Doi: 10.5935/2177-1235.2020RBCP 0086
3. Oliveira KCD, Pereira RMR, Salgado IV, Baptista EVDP, Arantes GC, Luna IC. Opções a capsulotomia e capsulectomia no tratamento da contratura capsular: existem alternativas medicamentosas ao tratamento cirúrgico? Revisão de literatura. Rev Bras Cir Plást 2015;30(01):123-128. Doi: 10.5935/2177-1235.2015RBCP 0127
4. Sperli A, Bersou A Junior, Freitas JOG, Michalany N. Complicações com próteses mamárias. Rev Bras Cir Plást 2000;15(03):33-46
5. Valencia-Lazcano AA, Román-Doval R, De La Cruz-Burelo E, Millán-Casarrubias EJ, Rodríguez-Ortega A. Enhancing surface properties of breast implants by using electrospun silk fibroin. J Biomed Mater Res B Appl Biomater 2018;106(05):1655-1661. Doi: 10.1002/jbm.b.33973
6. Kumar V, Abbas AK, Fausto N, Aster JC, editors. Robbins & Cotran Patologia - bases patológicas das doenças. 8th ed. Rio de Janeiro: Elsevier; 2004
7. Lee Y, Song SE, Yoon ES, Bae JW, Jung SP. Extensive silicone lymphadenopathy after breast implant insertion mimicking malignant lymphadenopathy. Ann Surg Treat Res 2017;93(06): 331-335. Doi: 10.4174/astr.2017.93.6.331
8. Fagundes DJ, Taha MO. Modelo animal de doença: critérios de escolha e espécies de animais de uso corrente. Acta Cir Bras 2004; 19(01):59-65. Doi: 10.1590/S0102-86502004000100010
9. Nichter LS, Hardesty RA, Anigian GM. IDEAL IMPLANT Structured Breast Implants: Core Study Results at 6 Years. Plast Reconstr Surg 2018;142(01):66-75. Doi: 10.1097/PRS.0000000000004460
10. França DCC, Castro AL, Soubhia AMP, Rosa E, Aguiar SMHCA. Avaliação da biocompatibilidade de implantes de silicone gel em subcutâneo de ratos - estudo histomorfométrico. Rev Fac Odontol (Univ Passo Fundo) 2011;16(01):69-74. Doi: 10.5335/rfo.v16i1.868
11. Daniel MJB, Daniel LAGB, Daniel VAGB, Graça Neto L. Aumento do volume do implante mamário por passagem de material orgânico para seu interior. Rev Bras Cir Plást 2023;38(01):e0185. Doi: 10.5935/2177-1235.2023RBCP0185-PT
12. Miró AL. Polyurethane-coated silicone breast implants: evaluation of 14 years experience. Rev Bras Cir Plást 2009;24:296- 303
1. School of Medical and Health Sciences, Pontifícia Universidade Católica de São
Paulo, Sorocaba, SP, Brazil
Address for correspondence Luciana Canabarro, School of Medical and Health Sciences, Pontifícia Universidade Católica de São Paulo, Sorocaba, SP, Brazil (e-mail: kanabarro@gmail.com ,lcandrade@pucsp.br).
Artigo submetido: 21/03/2025.
Artigo aceito: 26/08/2025.
Conflict of Interests
The authors have no conflict of interests to declare.













 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter