INTRODUCTION
The repair of structural defects with endogenous tissues, undoubtedly a skill of
plastic surgeons, is limited in many situations and has stimulated the
production of supportive biomaterials, with numerous synthetic materials
developed and used on a large scale for applications in various fields of
reconstructive surgery.
As part of an evolution of this process, biological prostheses were developed,
originating from acellularized natural tissues1, providing biodegradable three-dimensional
support for the recipient’s cellular growth and requiring sophisticated
degradation kinetics over time2. Basically represented by the extracellular membrane (ECM)
resulting from the acellularization process, these membranes develop an active
biological role at the implantation site, in theory favoring tissue remodeling
rather than the formation of scar fibrosis or chronic inflammation3, concepts pursued in the field
of regenerative medicine.
Progressively degraded by metalloproteinases4 - especially collagenase - acellular membranes must
support a complex balance between resistance to degradation and promotion of
cell growth from the receptor bed, with dynamic reciprocity favoring tissue
neoformation and adequate collagen deposition until the repair site has healed
adequately. Thus, in addition to the basic aspect of biocompatibility,
evaluating the degradation time of the three-dimensional support is also
essential, as its very early occurrence can lead to failure of the repair,
especially in those that require greater mechanical resistance, such as in the
reconstruction of the abdominal wall5.
In this sense, in addition to the differences in relation to their allogeneic or
xenogenic origin, as well as their tissue biological nature - dermis, intestinal
mucosa, pericardium, etc. - aspects related to the preparation and reticulation
processes are described as important factors in the biological behavior of ECMs.
Studies demonstrate that reticulation increases the durability of implanted
biomaterials, thus providing a greater capacity to provide adequate support for
remodeling processes with endogenous collagen in abdominal hernia
repairs7.
Numerous publications use histological analyses as the main tool for evaluating
these biological processes in different bioprostheses. However, the vast
majority are in animal experimentation8,9, with
observations in humans restricted to complicated cases of reoperations in the
presence of infections and implant removal.10,11
OBJECTIVE
The objective of this publication is to report the histological findings observed
in biopsies of acellular bovine pericardial membranes implanted in abdominal
wall repair.
METHOD
From a series of 40 abdominal wall repairs associated with implantation of
acellular bovine pericardial membrane, 3 patients underwent surgical revision,
namely 2 cases, secondary to incisional hernias, for correction of hypertrophic
scar at 13 months (Figure 1) and 22 months
postoperatively, and 1 case, secondary to post-resection reconstruction of wall
endometrioma, reviewed at 23 months postoperatively to explore possible
recurrence. In all cases, the postoperative evolution was without any
complications, with clinical and radiological examinations not identifying
problems related to the implanted areas, with successful repairs, with revisions
being carried out for indications not related to implants. The patients were
duly informed, through a form of consent, that biopsies would be taken in the
implant area at the time of eventual surgical revision.
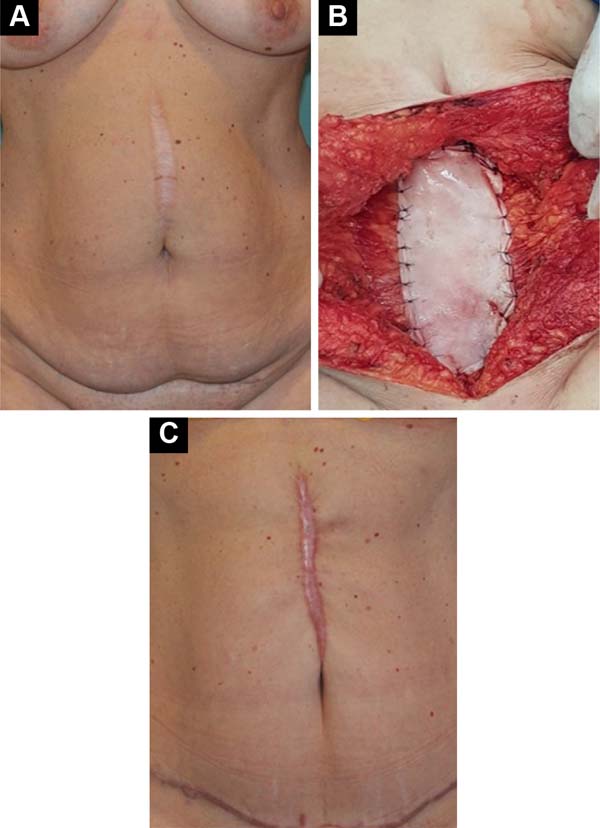
Figure 1 - A: Patient with supraumbilical incisional hernia,
with indication for repair associated with abdominal wall
reinforcement with acellular bovine pericardium membrane.
B: Intraoperative view of the correction performed,
with the membrane in a suprafascial position after direct
approximation of the muscles. C: 13 months
postoperatively, showing hypertrophic supraumbilical and suprapubic
scars compromising the aesthetic result. During the surgical review
of this scar, the implanted area was observed, confirming the
absence of recurrence of the hernia or other changes, and 3 biopsies
were then obtained for histological analysis.
Figure 1 - A: Patient with supraumbilical incisional hernia,
with indication for repair associated with abdominal wall
reinforcement with acellular bovine pericardium membrane.
B: Intraoperative view of the correction performed,
with the membrane in a suprafascial position after direct
approximation of the muscles. C: 13 months
postoperatively, showing hypertrophic supraumbilical and suprapubic
scars compromising the aesthetic result. During the surgical review
of this scar, the implanted area was observed, confirming the
absence of recurrence of the hernia or other changes, and 3 biopsies
were then obtained for histological analysis.
In the areas corresponding to previous implants in a pre-aponeurotic situation,
made by the same surgeon and identified photographically, 3 samples were taken
at different points in the implanted region, removing samples from the muscular
aponeurosis in its entire thickness. After fixation and inclusion in paraffin,
serial sections of 5µm thickness were made, with 60 slides being stained for
each patient with Hematoxylin - Eosin, Gomori’s Trichrome, and Picrosirius Red
for the different analyses.
Morphometric analysis
The slides were examined with a Nikon SI E200 Trinocular optical microscope
for the usual stains and with polarized light for Picrosirius Red, and the
images were digitized with a Digilab™ jkc camera at 8MB resolution. In
addition to the basic aspects regarding the biocompatibility of the material
and characteristics of tissue neoformation, aspects of
absorption/degradation of the implants and the process of cellularization
and collagen deposition in the recipient bed were also analyzed, quantified
by computerized analysis using the ImageJ software, specific for this
purpose12.
Using the histological image of the “in natura” acellular
pericardium as a standard (Figure 2),
the percentages of residual membrane present in the different periods were
quantified on the HE-stained slides. The acellular pericardium still present
in the different samples was identified and delimited manually by two
independent examiners, with the corresponding percentage calculated
automatically by the software.
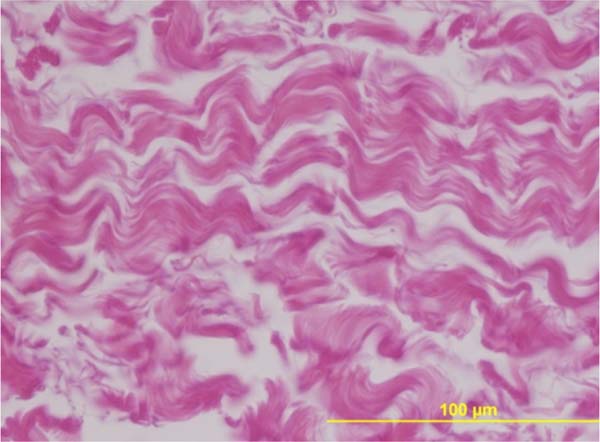
Figure 2 - Standard histological appearance of the bovine pericardial
membrane “in natura” after acellularization process, confirmed
by the total absence of cell nuclei. This pattern, digitally
memorized in pixels and colored by ImageJ software tools, was
used to identify and quantify residual fragments of the
membranes implanted in different postoperative periods. HE
staining/100xx magnification.
Figure 2 - Standard histological appearance of the bovine pericardial
membrane “in natura” after acellularization process, confirmed
by the total absence of cell nuclei. This pattern, digitally
memorized in pixels and colored by ImageJ software tools, was
used to identify and quantify residual fragments of the
membranes implanted in different postoperative periods. HE
staining/100xx magnification.
On slides stained with Picrosirius Red - specific for collagen fibers - the
quantification of tissue fractal dimension was additionally carried out by
digital analysis13, also
using the ImageJ software, representing tissue fragmentation by a specific
automatic method called “Box-Count /Binary - Outline”.
Statistical analysis
The values obtained in the quantification of the percentages of residual
membrane had their means statistically compared by analysis of variance
(ANOVA) and the unpaired Student’s T test, also used to analyze the values
obtained in the quantification of the fractal dimension. An alpha error of
5% was allowed, with p-values less than or equal to 0.05 being considered
significant.
RESULTS
Histological analyses clearly demonstrated the biocompatibility of the material,
with all samples showing tissue neoformation replacing the implanted membranes,
with significant deposition of collagen and cellularized tissue with a normal
appearance. No important local reactions were observed, with some rare isolated
focal points being identified showing macrophages in a mild inflammatory
process. In all periods analyzed, it was possible to identify the presence of
fragments of acellular tissue corresponding to the original membrane (Figure 3).
Using ImageJ software, residual fragments of the implanted membrane were
quantified at 40%±7% at 13 months, at 20%±6% at 22 months, and 17%±6% at 23
months postoperatively. This quantification, analyzed by the unpaired t-test,
was statistically significant between the 13-month and later samples, with no
statistical difference between 22 and 23 months (Graph 1).
Graph 1 - Box-plot representation of the quantification and statistics by
analysis of variance of the percentages of residual fragments of
acellular pericardial membrane in the different periods. Residual
membranes were histologically identified at 13, 22, and 23 months
postoperatively, and their respective percentages were calculated
using the specific ImageJ software tool. There was a statistically
significant difference between the 13-month samples compared to 22
and 23 months (p<0.0001) and no difference in
the comparison between 22 and 23 months.
Graph 1 - Box-plot representation of the quantification and statistics by
analysis of variance of the percentages of residual fragments of
acellular pericardial membrane in the different periods. Residual
membranes were histologically identified at 13, 22, and 23 months
postoperatively, and their respective percentages were calculated
using the specific ImageJ software tool. There was a statistically
significant difference between the 13-month samples compared to 22
and 23 months (p<0.0001) and no difference in
the comparison between 22 and 23 months.
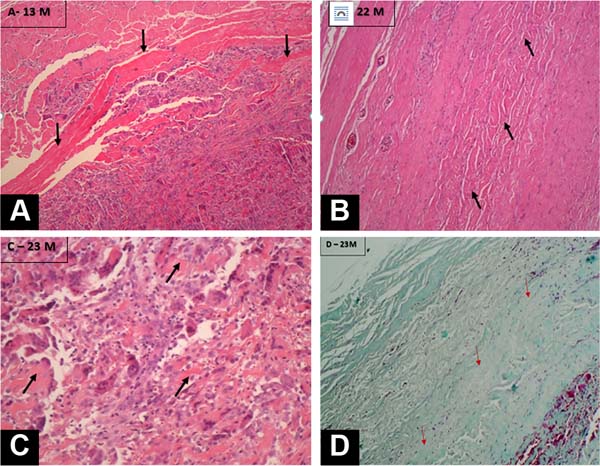
Figure 3 - Histological sections of biopsies from the areas corresponding to
implants of acellular bovine pericardial membranes in the
postoperative periods of A, 13 months; B, 22 months; C and D, 23
months. In all periods, newly formed cellularized tissue is observed
replacing the implanted membranes, demonstrating a good quality
repair process and the absence of inflammatory processes or “foreign
body” type reactions. In all samples, fragments of residual
acellularized tissue from the implanted membrane were observed
(black arrows), quantified at approximately 40% at 13 months, 20% at
22 months, and 17% at 23 months postoperatively. In D, 23 months
postoperatively, neocollagen deposition is observed with a good
pattern of scar repair and normal cellularized tissue replacing the
implant (red arrows). A, B, and C Hematoxylin-Eosin staining. In D
Gomori Trichrome. 40xx increase in B and D; 100xx increase in A and
C.
Figure 3 - Histological sections of biopsies from the areas corresponding to
implants of acellular bovine pericardial membranes in the
postoperative periods of A, 13 months; B, 22 months; C and D, 23
months. In all periods, newly formed cellularized tissue is observed
replacing the implanted membranes, demonstrating a good quality
repair process and the absence of inflammatory processes or “foreign
body” type reactions. In all samples, fragments of residual
acellularized tissue from the implanted membrane were observed
(black arrows), quantified at approximately 40% at 13 months, 20% at
22 months, and 17% at 23 months postoperatively. In D, 23 months
postoperatively, neocollagen deposition is observed with a good
pattern of scar repair and normal cellularized tissue replacing the
implant (red arrows). A, B, and C Hematoxylin-Eosin staining. In D
Gomori Trichrome. 40xx increase in B and D; 100xx increase in A and
C.
Using Picrosirius staining with polarized light, the fractal dimension of the
slides was analyzed at different postoperative periods, also using an automatic
method in a specific tool in the ImageJ software, demonstrated in Figure 4.
The distribution of fractal dimension values for each subgroup, using the
Box-Plot graph, shows a clear separation of values between the subgroup with
the
shortest follow-up time (13 months) and the subgroups (together or separately)
with 22 and 23 months of follow-up. (Graph 2).
Graph 2 - Box-plot analysis of the fractal dimension of the histological
images of the acellular bovine pericardium in the different
postoperative periods, showing a clear separation of values for the
13-month samples and the subgroups (together or separately) of 22
and 23 months, indicating a difference statistically significant in
the progressive fragmentation of implants. ImageJ software (Make
Binary, Outline Method).
Graph 2 - Box-plot analysis of the fractal dimension of the histological
images of the acellular bovine pericardium in the different
postoperative periods, showing a clear separation of values for the
13-month samples and the subgroups (together or separately) of 22
and 23 months, indicating a difference statistically significant in
the progressive fragmentation of implants. ImageJ software (Make
Binary, Outline Method).
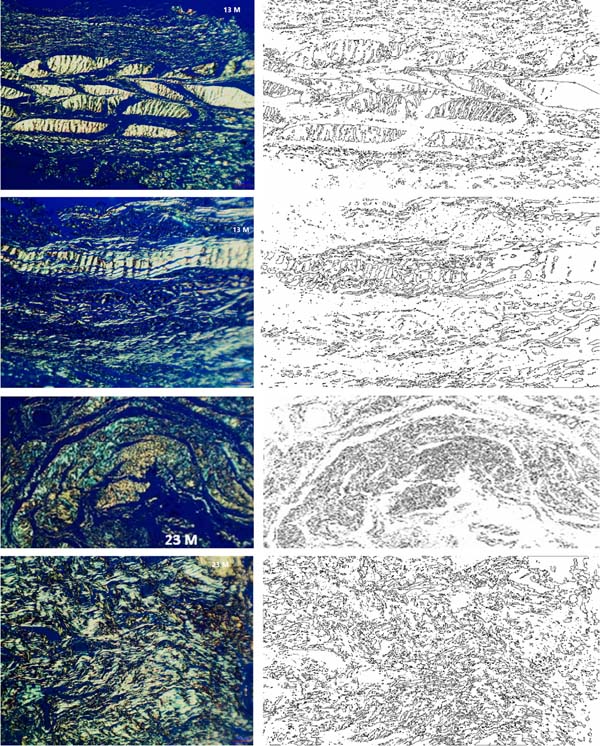
Figure 4 - Representation of the automated analysis of the fractal dimension
by ImageJ software. In the left column, digitized images of slides
stained in Picrosirius under polarized light vision, at 13 and 23
months postoperatively as indicated. On the right, the corresponding
computerized representation for analyzing the degree of structural
fragmentation defined as a fractal dimension. Automatic analyses
show a statistically significant increase in fractal dimension in
later cases, indicating a progressive occurrence of tissue
neoformation due to the cellularization process and collagen
deposition in the receptor bed, corroborating the findings of
membrane degradation kinetics.
Figure 4 - Representation of the automated analysis of the fractal dimension
by ImageJ software. In the left column, digitized images of slides
stained in Picrosirius under polarized light vision, at 13 and 23
months postoperatively as indicated. On the right, the corresponding
computerized representation for analyzing the degree of structural
fragmentation defined as a fractal dimension. Automatic analyses
show a statistically significant increase in fractal dimension in
later cases, indicating a progressive occurrence of tissue
neoformation due to the cellularization process and collagen
deposition in the receptor bed, corroborating the findings of
membrane degradation kinetics.
Analysis using the unpaired t-test showed a statistically significant difference
between 13 months versus 22 months (p=0.0058), between 13
months versus 23 months (p=0.0128), and between 13 months
versus the set of 22 and 23 months (p<0.0001), with an
increase in fractal dimension indicating the progressive occurrence of tissue
neoformation due to the cellularization process and collagen deposition in the
receptor bed. There was no statistically significant difference in the fractal
dimension comparing 22 months versus 23 months (p=0.3141).
The two morphometric evaluation methods adopted had concordant findings, with a
reduction in the percentage of residual implant demonstrating its progressive
absorption/degradation, concomitant with the occurrence of cellularization and
collagen deposition evidenced by the progressive increase in the fractal
dimension.
DISCUSSION
The exponential increase in the supply of acellular matrices of different origins
in recent years and the growth projections of this market14 prove the increasing adoption
of bioprostheses in different therapeutic options, as well as in tissue
engineering15, as
molds for stem cell cultivation16 and in the application of “drug delivery”17, with MECs embedded in
medicines with different purposes.
Its differential as an implant in various repair processes lies particularly in
its biocompatibility characteristics, the progressive degradation/absorption
of
the implants, and its concomitant replacement by tissue neoformation.
Furthermore, unlike synthetic implants, which can induce a polymer-dependent
inflammatory response with the formation of biofilms18,19,
acellular bioprostheses exert biological functions “in situ”,
favoring regenerative processes20,21, in addition
to allowing their application in contaminated and infected surgical
sites.22,23
Histological analyses based on experimental models constitute the main tool for
evaluating these biological processes, with hundreds of publications describing
various aspects of extracellular matrices such as tissue origin, thickness,
acellularization methods, reticulation, etc. - in an attempt to indicate the
best choices for the different repair processes. In the present study, it was
possible to histologically observe the main biological processes in humans under
normal conditions, an uncommon condition with aspects not yet described in the
literature for abdominal wall repairs.
In the implanted areas, it was possible to observe the incorporation of the
pericardial ECM into the recipient bed, with neovascularization and increasing
presence of cellularized neotissue and adequate collagen deposition in all
periods analyzed, with good quality repair and absence of inflammatory processes
or important signs of immune response. In addition to excellent
biocompatibility, this demonstrates that the material fulfilled its function
as
a biological scaffold, favoring the processes of cell adhesion, proliferation,
and differentiation, serving as a substrate for tissue repair, a fundamental
characteristic expected in biological structures composed of extracellular
matrices24.
Similar findings with acellular bioprostheses implanted in humans for breast
reconstructions have been reported in the literature, with human25 and porcine dermis26,27, describing the process of integration of ECMs as a
form of normal healing, with initial neovascularization followed by progressive
cellular repopulation of the matrix with cells of the receptor and absence of
foreign body type reactions.
With data also not yet found in the literature, it was possible to quantify the
degradation kinetics of the acellular bovine pericardium implanted in the
abdominal wall, analyzed by two complementary computerized methods. In all
biopsies from areas implanted in different periods, it was possible to identify
standard fragments of residual acellular pericardium, which were quantified as
a
percentage, complemented with the analysis of the fractal dimension of the
samples over time.
Both analyses indicated that the process of reabsorption and replacement by
neotissue is progressive, with a statistically significant difference, observing
that around 60% of the implant was reabsorbed after 13 months post-surgery and
around 80% after around two years, suggesting that the entire matrix should be
degraded in the long term.
Other publications also describe the degradation kinetics in percentages of
residual or absorbed ECM for porcine dermis and intestinal serosa, also with
morphometry computerized, by multispectral analysis of histological
images28 or with
matrices marked with Carbon-1429. The results show the presence of residual membrane for up
to 90 days for non-reticulated intestinal serous matrices, disappearing around
180 days and, for reticulated dermal matrices, much slower reabsorption, with
the presence of 80% of the implant in the first 4 weeks and 50 % still present
at around 6 months.
As described in the literature7,8,30, this aspect confirms the greater resistance to
degradation of the reticulated matrix used and may represent an advantage for
repairs in which greater long-term mechanical resistance is required, such as
in
the abdominal wall. The functionality of degradable materials depends on the
balance between the rate of degradation and the rate of tissue remodeling in
the
host bed, and it is necessary to understand not only the biological response
to
degradable biomaterials but also the expected mechanical properties of the
implant and replacement tissues over time for each therapeutic
application31.
These findings are compatible with several clinical and experimental studies
using different ECMs in abdominal wall repairs32,33,
also including bovine pericardium34, showing very satisfactory characteristics for their use
even in high-risk situations35. In a comparative analysis with the vast literature presented,
the results highlight the translational nature of the experimental models used
to evaluate and characterize acellular matrices and demonstrate the close
similarity of the pericardium used with those general characteristics and
therapeutic applications. However, numerous particular variables can affect
clinical results36-38, highlighting here for
discussion specific aspects of the receptor bed itself and the matrix used in
terms of acellularization, reticulation and its presentation in liquid
media.
The action of biomechanical forces acting in different locations can
differentially affect collagen distribution and tissue remodeling of biological
molds39, which is a
fundamental component to be considered when using ECMs in the abdominal
wall40. The results
obtained demonstrated good-quality tissue neoformation in all samples, attesting
to the functionality of the implant under different levels of mechanical stress
on the abdominal wall.
The pericardium used is fixed in glutaraldehyde - a technique used effectively
for decades in acellular matrices41 - and soaked post-fixation in 4% formaldehyde and is sold
in this way. In addition to glutaraldehyde promoting a reduction in connective
tissue antigenicity and stabilization against chemical and enzymatic degradation
in varying degrees of “reticulation” 42,43, this
association has well-described terminal sterilization effects44. This important factor can
also affect the structural properties of acellular matrices45. In addition to simpler
processing, maintenance in liquid media is described as advantageous for tissue
architecture, avoiding collapse and preserving matrix components that provide
mechanical and biochemical benefits after implantation46.
Although freeze-drying facilitates the manipulation and long-term preservation of
ECMs, factors can affect their performance both during their synthesis, with
disturbances of collagen fibers47, and at the time of their implantation, with rehydration
time being able to alter their biomechanical and physical properties
significantly. -chemicals48.
We can speculate that these factors also favored the behavior of the membrane
used, both due to its biocompatibility and its observed degradation
kinetics.
CONCLUSION
Histological analyses demonstrated similarity with all the biological
characteristics described in the literature for acellular tissue matrices, and
the process of integration and incorporation of ECMs could be observed in the
samples, with neovascularization followed by progressive cellular repopulation
of the matrix with receptor cells and collagen deposition with good healing
quality, demonstrated by the increase in fractal dimension. Also relevant in
humans, the degradation kinetics of the bovine pericardium matrix was quantified
at approximately 60% after 13 months and 80% after approximately two years,
suggesting that the entire matrix may be degraded over a longer period.
Under both aspects, the results attested to the functionality of the acellular
bovine pericardium under different levels of mechanical stress in abdominal wall
repairs in humans.
REFERENCES
1. Baumann DP, Butler CE. Bioprosthetic mesh in abdominal wall
reconstruction. Semin Plast Surg. 2012;26(1):18-24.
2. Panayi AC, Orgill DP. Current Use of Biological Scaffolds in Plastic
Surgery. Plast Reconstr Surg. 2019;143(1):209-20. DOI:
10.1097/PRS.0000000000005102
3. Brown BN, Badylak SF. Extracellular matrix as an inductive scaffold
for functional tissue reconstruction. Transl Res.
2014;163(4):268-85.
4. Sternlicht MD, Werb Z. How matrix metalloproteinases regulate cell
behavior. Annu Rev Cell Dev Biol. 2001;17:463-516.
5. Blatnik J, Jin J, Rosen M. Abdominal hernia repair with bridging
acellular dermal matrix--an expensive hernia sac. Am J Surg.
2008;196(1):47-50.
6. Costa A, Naranjo JD, Londono R, Badylak SF. Biologic Scaffolds. Cold
Spring Harb Perspect Med. 2017;7(9):a025676. DOI:
10.1101/cshperspect.a025676
7. Smart NJ, Bloor S. Durability of biologic implants for use in hernia
repair: a review. Surg Innov. 2012;19(3):221-9.
8. Liang HC, Chang Y, Hsu CK, Lee MH, Sung HW. Effects of crosslinking
degree of an acellular biological tissue on its tissue regeneration pattern.
Biomaterials. 2004;25(17):3541-52.
9. Mestak O, Spurkova Z, Benkova K, Vesely P, Hromadkova V, Miletin J,
et al. Comparison of Cross-linked and Non-Cross-linked Acellular Porcine Dermal
Scaffolds for Long-term Full-Thickness Hernia Repair in a Small Animal Model.
Eplasty. 2014;14:e22.
10. Wotton FT, Akoh JA. Rejection of Permacol mesh used in abdominal
wall repair: a case report. World J Gastroenterol.
2009;15(34):4331-3.
11. Cheung D, Brown L, Sampath R. Localized inferior orbital fibrosis
associated with porcine dermal collagen xenograft orbital floor implant.
Ophthalmic Plast Reconstr Surg. 2004;20(3):257-9.
12. Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years
of image analysis. Nat Methods. 2012;9(7):671-5.
13. Backes AR, Bruno OM. Técnicas de estimativa de dimensão fractal
aplicadas em imagens digitais. Relatórios Técnicos. São Carlos: Universidade
de
São Paulo; 2005. Disponível em: http://repositorio.icmc.usp.br//handle/RIICMC/6846
14. Acellular Matrix Treatment Market - Global Industry Analysis, Size,
Share, Growth, Trends and Forecast, 2021 - 2031. Disponível em: https://www.transparencymarketresearch.com/acellular-dermal-matrix-treatment-market.html
15. Knight RL, Wilcox HE, Korossis SA, Fisher J, Ingham E. The use of
acellular matrices for the tissue engineering of cardiac valves. Proc Inst Mech
Eng H. 2008;222(1):129-43. DOI: 10.1243/09544119JEIM230.
16. Iyyanki TS, Dunne LW, Zhang Q, Hubenak J, Turza KC, Butler CE.
Adipose-derived stem-cell-seeded non-cross-linked porcine acellular dermal
matrix increases cellular infiltration, vascular infiltration, and mechanical
strength of ventral hernia repairs. Tissue Eng Part A. 2015;21(3-4):475-85. DOI:
10.1089/ten.tea.2014.0235
17. Friess W. Collagen--biomaterial for drug delivery. Eur J Pharm
Biopharm. 1998;45(2):113-36.
18. Robinson TN, Clarke JH, Schoen J, Walsh MD. Major mesh-related
complications following hernia repair: events reported to the Food and Drug
Administration. Surg Endosc. 2005;19(12):1556-60.
19. Klosterhalfen B, Klinge U, Hermanns B, Schumpelick V. Pathology of
traditional surgical nets for hernia repair after long-term implantation in
humans. Chirurg. 2000;71(1):43-51. German.
20. Melman L, Jenkins ED, Hamilton NA, Bender LC, Brodt MD, Deeken CR,
et al. Early biocompatibility of crosslinked and non-crosslinked biologic meshes
in a porcine model of ventral hernia repair. Hernia. 2011;15(2):157-64. DOI:
10.1007/s10029-010-0770-0
21. Connor J, McQuillan D, Sandor M, Wan H, Lombardi J, Bachrach N, et
al. Retention of structural and biochemical integrity in a biological mesh
supports tissue remodeling in a primate abdominal wall model. Regen Med.
2009;4(2):185-95. DOI: 10.2217/17460751.4.2.185
22. Brennan EP, Reing J, Chew D, Myers-Irvin JM, Young EJ, Badylak SF.
Antibacterial activity within degradation products of biological scaffolds
composed of extracellular matrix. Tissue Eng.
2006;12(10):2949-55.
23. Harth KC, Broome AM, Jacobs MR, Blatnik JA, Zeinali F, Bajaksouzian
S, et al. Bacterial clearance of biologic grafts used in hernia repair: an
experimental study. Surg Endosc. 2011;25(7):2224-9.
24. Badylak SF, Freytes DO, Gilbert TW. Extracellular matrix as a
biological scaffold material: Structure and function. Acta Biomater.
2009;5(1):1-13.
25. Boháč M, Danišovič Ľ, Koller J, Dragúňová J, Varga I. What happens
to an acellular dermal matrix after implantation in the human body? A
histological and electron microscopic study. Eur J Histochem. 2018;62(1):2873.
DOI: 10.4081/ejh.2018.2873
26. Katerinaki E, Zanetto U, Sterne GD. Histological appearance of
Strattice tissue matrix used in breast reconstruction. J Plast Reconstr Aesthet
Surg. 2010;63(12):e840-1. DOI: 10.1016/j.bjps.2010.06.033
27. Salzberg CA, Dunavant C, Nocera N. Immediate breast reconstruction
using porcine acellular dermal matrix (Strattice™): long-term outcomes and
complications. J Plast Reconstr Aesthet Surg. 2013;66(3):323-8. DOI:
10.1016/j.bjps.2012.10.015
28. Costa A, Naranjo JD, Turner NJ, Swinehart IT, Kolich BD, Shaffiey
SA, et al. Mechanical strength vs. degradation of a biologically-derived
surgical mesh over time in a rodent full thickness abdominal wall defect.
Biomaterials. 2016;108:81-90.
29. Carey LE, Dearth CL, Johnson SA, Londono R, Medberry CJ, Daly KA, et
al. In vivo degradation of 14C-labeled porcine dermis biologic scaffold.
Biomaterials. 2014;35(29):8297-304. DOI:
10.1016/j.biomaterials.2014.06.015
30. de Castro Brás LE, Shurey S, Sibbons PD. Evaluation of crosslinked
and non-crosslinked biologic prostheses for abdominal hernia repair. Hernia.
2012;16(1):77-89. DOI: 10.1007/s10029-011-0859-0
31. Badylak S, Kokini K, Tullius B, Whitson B. Strength over time of a
resorbable bioscaffold for body wall repair in a dog model. J Surg Res.
2001;99(2):282-7. DOI: 10.1006/jsre.2001.6176
32. López Cano M, Armengol Carrasco M, Quiles Pérez MT, Arbós Vía MA.
Biological implants in abdominal wall hernia surgery. Cir Esp.
2013;91(4):217-23.
33. Lotan AM, Cohen D, Nahmany G, Heller L, Babai P, Freier-Dror Y, et
al. Histopathological Study of Meshed Versus Solid Sheet Acellular Dermal
Matrices in a Porcine Model. Ann Plast Surg. 2018;81(5):609-14.
34. Limpert JN, Desai AR, Kumpf AL, Fallucco MA, Aridge DL. Repair of
abdominal wall defects with bovine pericardium. Am J Surg.
2009;198(5):e60-5.
35. Shieh MK. Bovine Pericardium in Complex Abdominal Wall
Reconstruction in Patients with Obesity or Morbid Obesity. Bariatric Times.
2014;11(9):14-8.
36. Keane TJ, Londono R, Turner NJ, Badylak SF. Consequences of
ineffective decellularization of biologic scaffolds on the host response.
Biomaterials. 2012;33(6):1771-81.
37. Tierney CM, Haugh MG, Liedl J, Mulcahy F, Hayes B, O’Brien FJ. The
effects of collagen concentration and crosslink density on the biological,
structural and mechanical properties of collagen-GAG scaffolds for bone tissue
engineering. J Mech Behav Biomed Mater. 2009;2(2):202-9.
38. Badylak SF. Decellularized allogeneic and xenogeneic tissue as a
bioscaffold for regenerative medicine: factors that influence the host response.
Ann Biomed Eng. 2014;42(7):1517-27.
39. Cavallo JA, Roma AA, Jasielec MS, Ousley J, Creamer J, Pichert MD,
et al. Remodeling characteristics and collagen distribution in biological
scaffold materials explanted from human subjects after abdominal soft tissue
reconstruction: an analysis of scaffold remodeling characteristics by patient
risk factors and surgical site classifications. Ann Surg. 2015;261(2):405-15.
DOI: 10.1097/SLA.0000000000000471
40. Ventral Hernia Working Group; Breuing K, Butler CE, Ferzoco S, Franz
M, Hultman CS, Kilbridge JF, et al. Incisional ventral hernias: review of the
literature and recommendations regarding the grading and technique of repair.
Surgery. 2010;148(3):544-58. DOI: 10.1016/j.surg.2010.01.008
41. Reece IJ, van Noort R, Martin TR, Black MM. The physical properties
of bovine pericardium: a study of the effects of stretching during chemical
treatment in glutaraldehyde. Ann Thorac Surg. 1982;33(5):480-5.
42. Schmidt CE, Baier JM. Acellular vascular tissues: natural
biomaterials for tissue repair and tissue engineering. Biomaterials.
2000;21(22):2215-31. DOI: 10.1016/s0142-9612(00)00148-4
43. Jayakrishnan A, Jameela SR. Glutaraldehyde as a fixative in
bioprostheses and drug delivery matrices. Biomaterials.
1996;17(5):471-84.
44. Gorman SP, Scott EM, Russell AD. Antimicrobial activity, uses and
mechanism of action of glutaraldehyde. J Appl Bacteriol.
1980;48(2):161-90.
45. Freytes DO, Stoner RM, Badylak SF. Uniaxial and biaxial properties
of terminally sterilized porcine urinary bladder matrix scaffolds. J Biomed
Mater Res B Appl Biomater. 2008;84(2):408-14. DOI:
10.1002/jbm.b.30885
46. Faleris JA, Hernandez RM, Wetzel D, Dodds R, Greenspan DC. In-vivo
and in-vitro histological evaluation of two commercially available acellular
dermal matrices. Hernia. 2011;15(2):147-56. DOI:
10.1007/s10029-010-0749-x
47. Freytes DO, Tullius RS, Valentin JE, Stewart-Akers AM, Badylak SF.
Hydrated versus lyophilized forms of porcine extracellular matrix derived from
the urinary bladder. J Biomed Mater Res A. 2008;87(4):862-72. DOI:
10.1002/jbm.a.31821
48. Bottino MC, Jose MV, Thomas V, Dean DR, Janowski GM. Freeze-dried
acellular dermal matrix graft: effects of rehydration on physical, chemical,
and
mechanical properties. Dent Mater. 2009;25(9):1109-15. DOI:
10.1016/j.dental.2009.03.007
1. Hospital do Coração de São José do Rio Preto,
São José do Rio Preto, SP, Brazil
2. Instituto Frascino, Unidade de Publicação e
Pesquisa, São José do Rio Preto, SP, Brazil
3. Faculdade de Ciências Médicas da Santa Casa de
São Paulo, São Paulo, SP, Brazil
4. Faculdade de Medicina de São José do Rio Preto,
São José do Rio Preto, SP, Brazil
Corresponding author: Luiz Fernando Frascino Av.
Juscelino Kubitscheck de Oliveira, 3700, São José do Rio Preto, SP, Brazil, Zip
Code: 15093-225, E-mail: drfrascino@gmail.com
Article received: March 14, 2023.
Article accepted: December 5, 2023.
Conflicts of interest: none.