

Ideas and Innovation - Year 2023 - Volume 38 -
Alternative model for training microsuture techniques in bovine pericardium patches
Modelo alternativo para o treinamento de técnicas de microssuturas em "patch" de pericárdio bovino
ABSTRACT
Introduction: Microsurgical techniques are characterized by the application of maneuvers and sutures to millimetric structures with the aid of magnifying lenses. These are complex techniques, used in various medical specialties, which require great skill and training before applying them to humans. The objective is to develop a lowcost and high-fidelity model for training microsurgery techniques using a fragment of bovine pericardium patch.
Method: Remaining segments of a bovine pericardium plate, previously used in vascular repairs, are used to create this model. The material is cut into two symmetrical parts, and its ends are fixed to the surgical drapes with the aid of clamps. The upper edge of each part is then sutured to the lower edge with 8-0 Prolene thread so that each one forms a tubular structure. Subsequently, the free tubular ends undergo dissection of the adventitial layer and are sutured together, mimicking an end-to-end vascular anastomosis.
Results: With the model, the same inconveniences/ difficulties present in human vascular sutures are simulated, such as delamination of layers, excess of the adventitial layer, and risk of inadvertent suturing of the posterior wall, proving its usefulness in the acquisition of basic microsurgical skills, without need to manipulate human or animal tissues. Practice in this model can take place within the surgical center itself and uses materials that would otherwise be discarded.
Conclusion: The use of bovine pericardium to create millimetric sutures mimics human vascular tissue and is a low-cost procedure that allows the training of microsurgical skills.
Keywords: Microsurgery; Sutures; Suture Techniques. Teaching; Plastic surgery procedures.
RESUMO
Introdução: As técnicas microcirúrgicas caracterizam-se pela aplicação de manobras e suturas em estruturas milimétricas com o auxílio de lentes de aumento. São técnicas complexas, utilizadas em diversas especialidades médicas, que demandam grande habilidade e treinamento antes da aplicação em humanos. O objetivo é desenvolver um modelo de baixo custo e alta fidelidade, para o treinamento de técnicas de microcirurgia, utilizando um fragmento de patch de pericárdio bovino.
Método: São utilizados para a confecção deste modelo segmentos remanescentes de uma placa de pericárdio bovino, previamente utilizado em reparos vasculares. O material é recortado em duas partes simétricas e suas extremidades fixadas aos campos cirúrgicos, com auxílio de clamps. A borda superior de cada uma das partes é, então, suturada à borda inferior com fio de Prolene 8-0, de maneira que cada uma forme uma estrutura tubular. Posteriormente, as extremidades tubulares livres passam pela dissecção da camada adventícia e são suturadas entre si, mimetizando uma anastomose vascular término-terminal.
Resultados: Com o modelo, simulam-se os mesmos inconvenientes/ dificuldades presentes nas suturas vasculares humanas, como a delaminação de camadas, excesso da camada adventícia e risco de sutura inadvertida da parede posterior, provando sua utilidade na aquisição de habilidades microcirúrgicas básicas, sem necessidade de manipulação de tecidos humanos ou animais. A prática neste modelo pode ocorrer dentro do próprio centro cirúrgico e emprega materiais que seriam descartados.
Conclusão: A utilização do pericárdio bovino para confecção de suturas milimétricas mimetiza o tecido vascular humano e é um procedimento de baixo custo, que possibilita o treinamento de habilidades microcirúrgicas.
Palavras-chave: Microcirurgia; Suturas; Técnicas de Sutura; Ensino; Procedimentos de cirurgia plástica.
INTRODUCTION
Microsurgical procedures are characterized by the application of a set of techniques and surgical maneuvers on millimetric anatomical structures carried out with the aid of optical magnifying lenses 1 . Its principles can be applied to various medical specialties, such as in the areas of reconstructive plastic surgery, oncology, urology, head and neck, and transplants 2 . General surgeons can also apply its principles to biliodigestive or bile duct anastomoses or to uncomplicated vascular reconstructions 3 . It is a complex technique that requires great manual skill and continuous surgical training before application in humans 2 .
In the literature, there are several training models described involving different materials, both of animal and non-animal origin. Each model has particularities, advantages, and disadvantages. In the training of initial basic techniques, most descriptions use non-animal models, such as latex 4 , 5 . When improving microscopic surgical skills, animal models are preferred, such as mice, pigs, birds and dogs, depending on the availability of each institution 6 - 9 .
Practices using synthetic materials that mimic organic tissue for the development of basic skills, followed by the use of models with tissues of animal origin, rationalize animal use and reduce costs in the initial stages of learning 10 . The experimental models developed for basic training in microsutures must enable the teaching of experimental surgery safely, allowing the handling of instruments for microsurgery and the surgical microscope 4 .
In many countries, there are no training centers or facilities that have adequate technical resources for learning in this area. In this context, the biggest obstacle to achieving adequate training becomes the necessary investment. Therefore, the development of alternative experimental models for the practice of microsurgical techniques is essential. With the diversity of models, each center can choose or adapt the one that best fits their routine and their possibilities in relation to costs 11 .
OBJECTIVE
The objective of this work is to propose a training model for microsurgical sutures based on the use of remaining, discarded segments of bovine pericardium patch, a material used in vascular repairs in humans.
METHOD
This work was submitted and analyzed by the Animal Use Ethics Committee of the Agricultural Sciences Sector of the Universidade Federal do Paraná (CEUA-SCA-UFPR) and deemed exempt from approval due to proof of the origin of the biological material sold, manipulated by the biomedical industry, certified by the National Health Surveillance Agency (ANVISA).
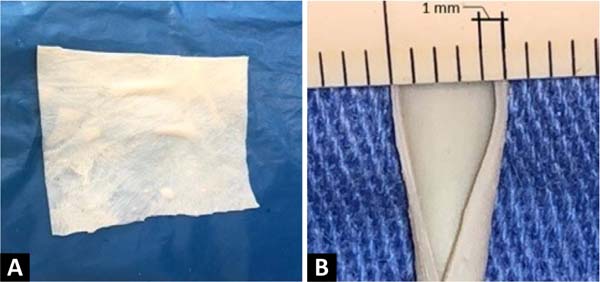
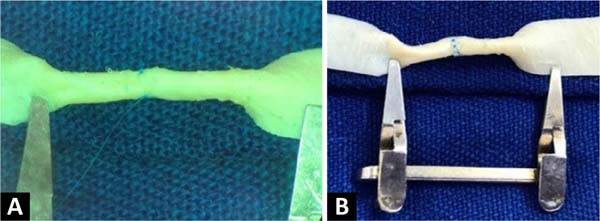
The creation of the microsurgical suture training model based on a fragment of bovine pericardium patch ( Figure 1 ) uses a surgical microscope, microclamp, scissors, straight and curved tweezers, microsurgical needle holder, as well as threads and materials of synthesis. The material used in this model is the remains of the open bovine pericardium plate, although not completely used, in carrying out vessel repairs in vascular surgeries in humans, which would be discarded.
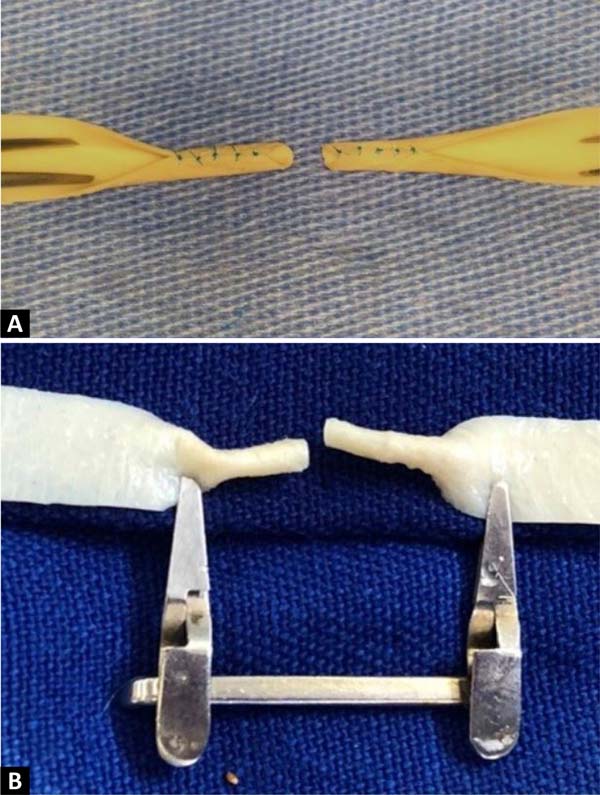
The procedure begins by cutting out a small segment of the bovine pericardium measuring 60mm x 6.8mm, fixing one of its edges to the surgical field using a clamp, and positioning the whole under the lenses of the surgical microscope. An incision is made in the middle of the plate, dividing it into two identical segments ( Figure 2 ). Two tubes are then made at each end, with the help of separate simple stitches, with Prolene®8-0 applied to join the upper and lower edges of each segment.
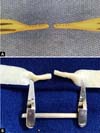
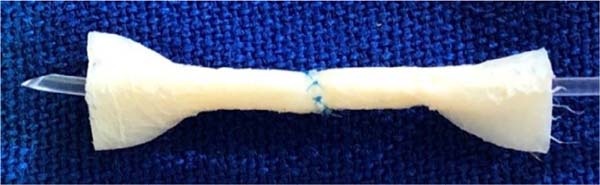
At the end of this step, two free circular ends are obtained, which mimic the sectioned ends of a 2.0 mm diameter vessel ( Figure 3 ). The outer edges of both segments of this section were prepared in the same way that vascular structures are prepared for microsuture, removing the excess adventitial layer to prevent the entry of excess tissue into the future vascular lumen during training in creating the anastomosis ( Figure 4 ).
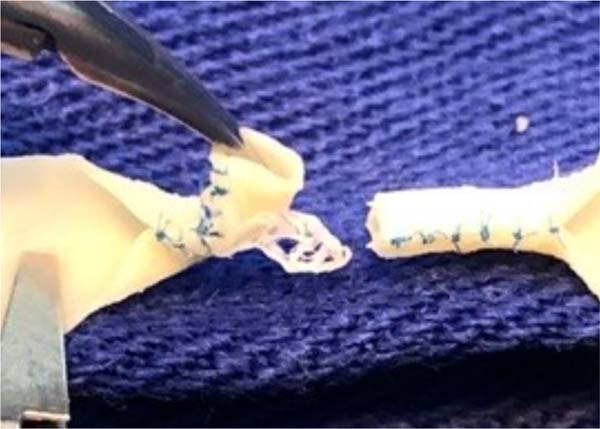
At this point, the model is ready to create an end-to-end microvascular anastomosis of the two ends that mimics the lumen of a small vessel. The vascular anastomosis can be created with the aid of an 8-0 Prolene®thread, creating two anchoring points opposite at 180° and followed by simple stitches separated between the two initial anchoring points ( Figure 5 ).
After suturing the anastomosis, it is possible to test the patency of the vessel by introducing a number 20F vascular catheter inside it. Transposition without resistance demonstrates the absence of accidental suturing of the posterior wall ( Figure 6 ).
RESULTS
With the alternative experimental model described, it was possible to train microsuture and microanastomose techniques using bovine pericardium patch plates. The model is reproducible in an experimental laboratory that has apparatus and instruments for microsurgery. This is an economically viable model, as it used leftover biological material widely used in surgeries and vascular repairs, which would be discarded after the original surgical procedure. In addition to the economic advantage, due to the use of fragments of biological material, the model did not involve the use of experimental animals.
It was observed that bovine pericardium tissue presents the same drawbacks present in vascular sutures in humans, such as the risk of layer delamination, excess adventitial layer and the risk of suturing the posterior wall. With direct visualization of all layers of the sutured tissue, dissection of the excess adventitial layer, and passage of a catheter to test its patency, it was possible to overcome these inconveniences.
DISCUSSION
Due to the great complexity, initial training in microsurgical procedures must first be carried out in an environment that does not involve patients. The microsurgical skills training routine must be defined in order first to become familiar with the instruments, properly handle the surgical threads, and master dissection, suturing and tissue anastomosis techniques. The practice of these skills under adequate supervision, with a well-established routine and with the use of materials that would otherwise be discarded, makes it possible to obtain the bases for the initial training of academics, residents, and surgeons, as it allows the immediate application of theoretical knowledge in an experimental model. and rationalizes animal use, reducing costs 6 , 12 .
Several experimental models for the practice of microsurgical techniques have already been described in various animals, such as pigs, dogs, chickens, and rats 6 , 8 , 9 . However, the strict institutional laws imposed by ethics committees on animal use can hinder the broad use of animals in the learning process. Artificial materials such as gloves or silicone tubes are also found as alternative models in the literature 4 , 13 , 14 . However, these models may have the disadvantage of not perfectly mimicking the vascular tissue, reducing the level of difficulty in learning microanastomoses techniques 12 .
Training these techniques can be long and exhausting. In this way, by scaling the technical difficulty, starting with synthetic materials, evolving to the pericardial patch model, and, later, animal models, training can become stimulating since the practical evolution of the training itself is perceived 8 .
The experimental model proposed here makes it possible to create anastomotic ends mimicking vessels of different calibers, depending on the size of the plate that is initially cut. In the described procedure, 8-0 Prolene thread was used for a structure with a diameter of 2.0 mm, obtained from a cut plate with a free end of 6.8 mm. It is recommended to use a 9-0 thread if the diameter of the anastomotic ends is less than 2mm.
One of the advantages of using surplus bovine pericardium patch material is precisely its ability to mimic the concentric layers of vascular tissue. When applying microsutures to this material, there is a risk of delamination or detachment of the intimal layer when the suture partially transfixes the wall thickness of the material. Care must be taken to cover the entire thickness of the bovine pericardium wall when applying sutures during end-to-end anastomosis training, and care must also be taken to keep the posterior wall free and not suture it together with the points of the previous anastomosis line so that the anastomosis is kept patent ( Figure 6 ).
Before performing a microvascular anastomosis, the adventitia and periadventitial tissues are routinely removed from the small arteries. This is one of the most important parts of vascular anastomoses, as it allows a clear definition of where the vessel ends and a more precise creation of the suture. Furthermore, excess tissue from the adventitial layer, if close to the cut ends of the vessel, can become interposed in the anastomosis and, as it is a highly thrombogenic tissue, can lead to anastomosis failure. Non-animal microsurgical models are unlikely to be able to mimic this important step 15 . This also provides training in removing the adventitial layer, a standard procedure performed before a microvascular anastomosis ( Figure 3 ).
CONCLUSION
The use of bovine pericardium plates as a training model for sutures and microsurgical end-to-end anastomoses allows the development of microsurgical skills in a realistic way since the manipulation of this material mimics the layers of a blood vessel and presents technical difficulties similar to those human tissues.
The proposed model uses good quality material, which would otherwise be discarded, which makes the implementation cost low when compared to models that use simulators or animals. Furthermore, it avoids the inconveniences of animal use, such as maintenance of breeding and storage facilities, food costs, analgesia, and disposal, among others. Therefore, the use of surplus bovine pericardium that would otherwise be discarded is an appropriate model of microsurgical training in the initial phase of learning, as it has properties very similar to human tissues and does not require animal use.
REFERENCES
1. Pessoa SGP, Riquet GF. Fundamentos básicos de microcirurgia vascular: estudo experimental. Ceará Med.1982;4(1):10-6.
2. Martins PNA, Montero EF. Basic microsurgery training. Comments and proposal. Acta Cir Bras. 2007;22(1):79-81.
3. Di Cataldo A, Li Destri G, Trombatore G, Papillo B, Racalbuto A, Puleo S. Usefulness of microsurgery in the training of the general surgeon. Microsurgery. 1998;18(8):446-8.
4. Dias IS, Pessoa SGP, Benevides AN, Macêdo JE. Treinamento inicial em microcirurgia. Rev Bras Cir Plást. 2010;25(4):595-9.
5. Pessoa BBGP, Pessoa SGP. Treinamento em microanastomoses utilizando tubos de látex. Acta Cir Bras. 2002;17(2):143-6.
6. Marcondes CA, Pessoa SGP, Pessoa BBGP, Dias IS, Guimarães MGM. Padronização técnica no treinamento em microcirurgia do serviço de cirurgia plástica e microcirurgia reconstrutiva do hospital universitário Walter Cantídio da Universidade Federal do Ceará (HUWC/UFC). Rev Bras Cir Plást. 2010;29(2):283-8.
7. Kinshoku MR, Rodriguez CAL, Fidalgo RDS, Duran CCG, Leme PLS, Duarte IDS. Uso racional de modelos animais para pesquisa e ensino de microcirurgia. Rev Col Bras Cir. 2012;39(5):414-7.
8. Maluf Junior I, Silva ABD, Groth AK, Lopes MAC, Kurogi AS, Freitas RS, et al. Modelo experimental alternativo para treinamento em microcirurgia. Rev Col Bras Cir. 2014;41(1):72-4.
9. Pessoa BBGP, Pessoa SGP. O retalho hipogástrico cutâneo no cão: modelo para o aprendizado experimental de microcirurgia. Acta Cir Bras. 2002;17(3):198-202.
10. Grober ED, Hamstra SJ, Wanzel KR, Reznick RK, Matsumoto ED, Sidhu RS, et al. The educational impact of bench model fidelity on the acquisition of technical skill: the use of clinically relevant outcome measures. Ann Surg. 2004;240(2):374-81.
11. Isolan GR, Santis-Isolan PMB, Dobrowolski S, Cioato MG, Meyer FS, Antunes ACM, et al. Considerações técnicas no treinamento de anastomoses microvasculares em laboratório de microcirurgia. J Bras Neurocir. 2010;21(1):8-17.
12. Webster R, Ely PB. Treinamento em microcirurgia vascular: é economicamente viável? Acta Cir Bras. 2002;17(3):194-7.
13. Hoşnuter M, Tosun Z, Savaci N. A nonanimal model for microsurgical training with adventitial stripping. Plast Reconstr Surg. 2000;106(4):958-9.
14. Lima DA, Galvão MSL, Cardoso MM, Leal PRDA. Rotina de treinamento laboratorial em microcirurgia do Instituto Nacional do Câncer. Rev Bras Cir Plást. 2012;27(1):141-9.
15. Lohman R, Siemionow M, Lister G. Advantages of sharp adventitial dissection for microvascular anastomoses. Ann Plast Surg. 1998;40(6):577-85.
1. Universidade Federal de São Paulo, São Paulo,
SP, Brazil
2. Universidade Federal do Paraná, Curitiba, PR,
Brazil
Corresponding author: Caroline Cunico Rua Botucatu, 740, 2º andar, Vila Clementino, São Paulo, SP, Brazil, Zip Code: 04023-062, E-mail: cunico.caroline@gmail.com
Article received: March 16, 2023.
Article accepted: June 13, 2023.
Conflicts of interest: none.









 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter