

Case Report - Year 2023 - Volume 38 -
Milky secretion after late breast reconstruction with latissimus dorsi musculocutaneous flap with implant
Produção de secreção leitosa após reconstrução mamária tardia com retalho musculocutâneo de latíssimo do dorso com implante
ABSTRACT
We present a rare case of a 28-year-old patient with invasive ductal carcinoma who underwent neoadjuvant chemotherapy, modified radical mastectomy, radiotherapy, and late breast reconstruction with latissimus dorsi flap associated with the implant. One year later, she underwent reconstruction of the nipple-areola complex over the cutaneous island of the latissimus dorsi flap with a total skin graft from the thigh and graft from the caudal portion of the contralateral papilla. She became pregnant six months after the reconstruction of the nipple-areola complex and, unexpectedly, presented milk production by the reconstructed breast.
Keywords: Surgical flaps; Breast neoplasms; Breast feeding; Mammaplasty; Breast implants
RESUMO
Apresentamos um caso raro de uma paciente de 28 anos com carcinoma ductal invasivo submetida a quimioterapia neoadjuvante, mastectomia radical modificada, radioterapia e reconstrução mamária tardia com retalho de latíssimo do dorso associado a implante. Um ano depois, foi submetida a reconstrução do complexo areolopapilar sobre a ilha cutânea do retalho do grande dorsal com enxerto de pele total da coxa e enxerto da porção caudal da papila contralateral. Ela engravidou seis meses após a reconstrução do complexo areolopapilar e, inesperadamente, apresentou produção de leite pela mama reconstruída.
Palavras-chave: Retalhos cirúrgicos; Neoplasias da mama; Aleitamento materno; Mamoplastia; Implantes de mama
INTRODUCTION
Patients undergoing breast surgery are at a greater risk of being unable to breastfeed. Milk production is not expected when all breast tissues are removed – as is the case with mastectomies. This brief report describes a rare case of a woman who could breastfeed following radical mastectomy, latissimus dorsi flap, and implant reconstruction.
Milk production and secretion depend on the mammary gland tissue being present and functioning properly.1 Patients undergoing radical mastectomy have the milk-producing tissue removed, rendering breastfeeding impossible.2
We present a case of a breast cancer patient who underwent late breast reconstruction with a latissimus dorsi flap (LDF) and implant following modified radical mastectomy (MRM). The patient subsequently became pregnant and presented milky secretion via the reconstructed nipple-areola complex (NAC). The COVID-19 pandemic isolation precluded laboratory analysis of the secretion, and it was impossible to confirm that it was milk.
CASE REPORT
A 28-year-old woman, nulliparous, non-smoker, and free of comorbidities, came to us for late right breast reconstruction. At 25, she was diagnosed with invasive ductal carcinoma in the right breast, Grade 3, cT3N2aM0, CS-IIIA (Immunohistochemistry: Ki67+; HER2 score 3+; RE-; RP-).
She received four cycles of neoadjuvant chemotherapy with adriamycin and cyclophosphamide over two months. Four additional cycles of paclitaxel and trastuzumab consolidation were performed. Following chemotherapy, she underwent MRM, axillary lymphadenectomy, and immediate breast reconstruction with a tissue expander. The anatomopathological examination revealed grade II invasive carcinoma with clear margins, no angiolymphatic invasion, carcinoma foci in situ, and no papillary infiltration. Five lymph nodes were examined, and all were found to be normal. There was no mention of tumor size; only final staging was mentioned (ypT1bN0M0).
Soon after the expansion was completed, the patient received maintenance chemotherapy with trastuzumab (completing 18 doses) and adjuvant radiotherapy. She received external radiotherapy with a 6MV linear accelerator using a 3D technique. The adjuvant dose prescribed was 50.4 Gy in the right breast and supraclavicular fossa (fractions of 180cGy). The treatment was halted on the final day due to radiodermatitis after administering a total dose of 48.6Gy. According to the report, she evolved as her radiodermatitis worsened; the expander extruded and was, therefore, removed.
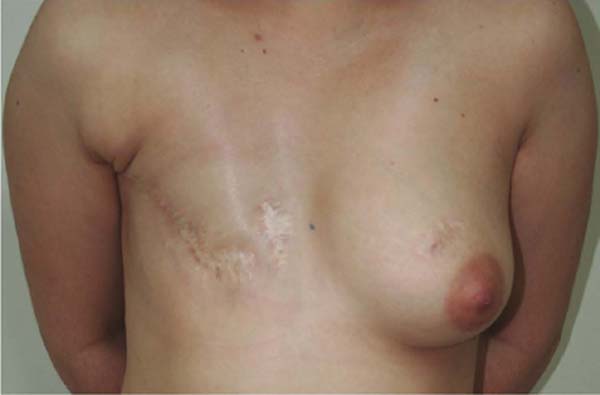
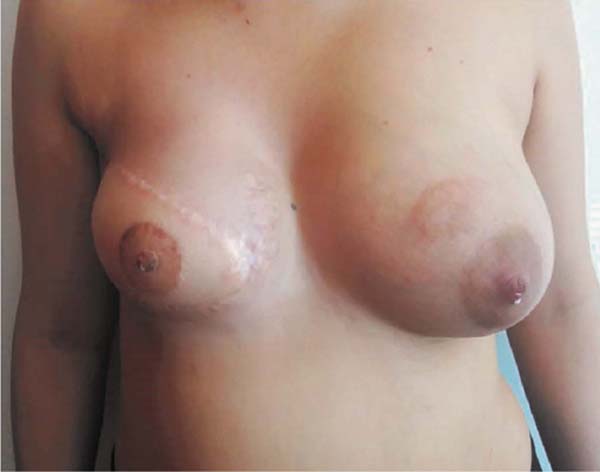
She contacted us 18 months after undergoing a mastectomy for late breast reconstruction (Figure 1). An LDF procedure was performed by placing a 320ml textured implant (Silimed™, Natural, extra-high projection).
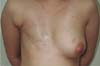
She was satisfied with the outcome one year after surgery and did not desire breast symmetrization procedures (Figure 2). Following that, NAC reconstruction was performed. The areola was reconstructed using a total skin graft from the medial thigh and the papilla was reconstructed using a graft from the contralateral papilla’s caudal portion. The patient lived in another city and did not return for follow-up after removing the sutures.
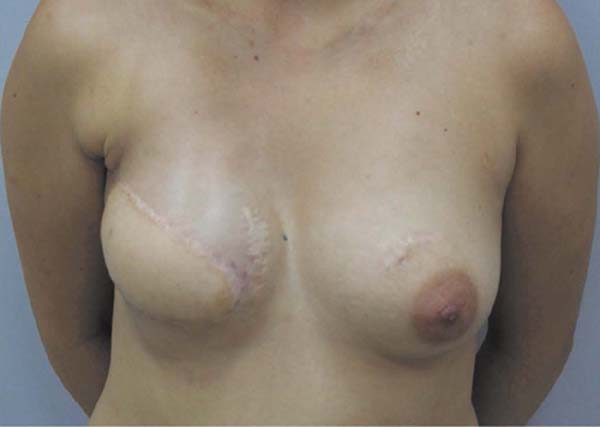
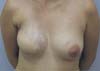
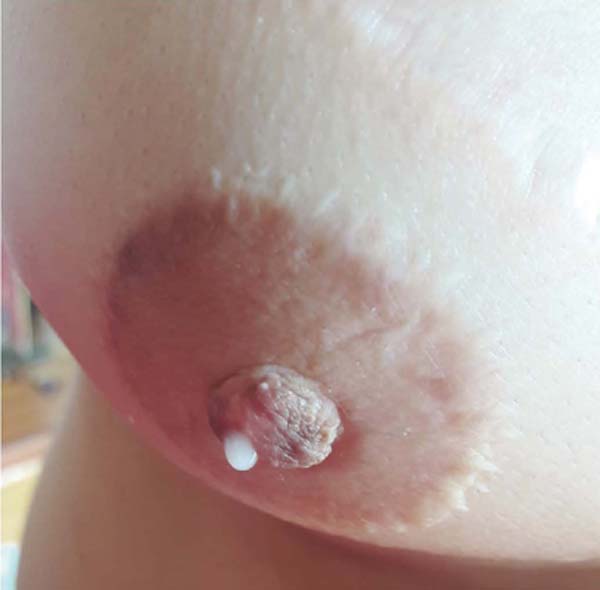
She contacted us six months later to inform us that she had become pregnant. Following the child’s birth in February 2020, she contacted us again to report that she was breastfeeding normally on the left breast despite the caudal papilla being removed and that the right breast produced milky secretion in small amounts (Figures 3 and 4, Video 1).
Due to the imposed social isolation caused by the COVID-19 pandemic, the patient could not return for imaging tests. The patient reported breastfeeding continuously for five months until July 2020.
DISCUSSION
Breastfeeding is inversely proportional to the amount of tissue removed following breast reduction.2,3 When all breast tissue is removed, milk production is not expected.2
Madeira et al.4 described a patient with Poland Syndrome who underwent LDF and implant reconstruction. Five years later, she became pregnant and successfully breastfed.4 However, that patient may have exhibited rudimentary remnants of breast tissue connected to the NAC.4 Conversely, our patient underwent a radical mastectomy, radiotherapy, and NAC reconstruction over the LDF’s cutaneous island.
Sakai and Sakai5 described a nipple-sharing technique that consists of harvesting the outer layer of the donor nipple and rolling this tissue into a spiral shape before grafting it. This technique is expected to make breastfeeding viable, as it is supposed to preserve the donor’s anatomy. In our case, however, the caudal portion of the papilla was utilized, reducing the likelihood of breastfeeding. Nevertheless, some ducts may have been left uncut, allowing milk ejection.
The infant’s suction of the NAC stimulates its nerve endings to secrete prolactin and oxytocin, thereby inducing milk production and ejection.1 One might hypothesize that the contralateral papilla fragment grafted to the reconstructed breast produced milky secretion in response to hormonal stimuli, as it is the only structure striving from breast tissue. This, however, contradicts the current understanding of lactation physiology. Due to the rarity of this situation, surgeons should be open to alternative theories, including construction morphology, bioelectricity, and the morphogenetic field.6,7
CONCLUSION
MRM, irradiation, total breast reconstruction using LDF in conjunction with the implant, and reconstruction of the NAC over the cutaneous island of the LDF all make the production of a milky secretion unlikely. Additional research is necessary to elucidate this unusual occurrence.
1. Universidade Federal de São Paulo, São Paulo, S P, Brazil.
2. Universidade do Vale do Sapucaí, Pouso Alegre, MG, Brazil.
Corresponding author: Daniela Francescato Veiga Av. Prefeito Tuany Toledo, 470, Fatima, Pouso Alegre, MG, Brazil Zip code: 37550-000 E-mail: danielafveiga@gmail.com






 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter