

Review Article - Year 2021 - Volume 36 - Issue 3
Genomic bases of non-syndromic basal cell carcinoma: literature review
Bases genômicas do carcinoma basocelular não sindrômico: revisão da literatura
ABSTRACT
Introduction: Non-melanoma skin neoplasms represent the most frequent type in both sexes globally, with basal cell carcinoma being the most prevalent, representing 75 to 80% of cases. In Brazil, the number of new cases expected for the triennium 2020-2022 will be 83,770 in men and 93,160 in women, corresponding to an estimated risk of 80.12 new cases for 100,000 men and 86.65 new cases for 100,000 women. This data demonstrates the great importance of genomic knowledge in the genesis of sporadic basal cell carcinoma.
Objective: To describe the main genes and molecular markers involved in the predisposition and pathogenesis of non-syndromic basal cell carcinoma.
Methods: Literature review in the main databases NCBI-GTR, ClinVar, ClinGen, MedGen, OMIM and GeneReviews, using as descriptors: "BCC" and "basal cell carcinoma". Inclusion criteria: Portuguese or EnGLIsh language, articles on sporadic BCC.
Results: Thirteen articles were selected for analysis. The analysis revealed a robust hedgehog pathway link in the genesis of sporadic basal cell carcinoma, with the main genes involved represented by PATCH1, PATCH2 and smoothened. The variants with the highest clinical significance were SMO-M2, PTCH1 and PTCH2-Δ22. The mutation most found was related to the action of UVB, being represented by the substitution of C>T or CC>TT at the pyrimidine site, both in PTCH and in SMO.
Conclusion: Extremely important to professionals working in the diagnosis and treatment of BCC, including plastic surgeons, as this way they can better conduct their cases, with more accurate diagnoses and prevention approaches based on the individual susceptibility of each patient, as well as targeted therapies and individualized with better success rates.
Keywords: Human genome; Basal cell carcinoma; Neoplasm of basal cells; Genes; Mutation.
RESUMO
Introdução: As neoplasias cutâneas não melanoma representam o tipo mais frequente em ambos os sexos no mundo, sendo o carcinoma basocelular o mais prevalente, representando de 75 a 80% dos casos. No Brasil, o número de casos novos esperados para o triênio 2020-2022, será de 83.770 em homens e 93.160 em mulheres, correspondendo a um risco estimado de 80,12 casos novos para 100 mil homens e de 86,65 casos novos para 100 mil mulheres. Este dado demonstra a grande importância do conhecimento genômico na gênese do carcinoma basocelular esporádico.
Objetivo: Descrever os principais genes e marcadores moleculares envolvidos na predisposição e na patogênese do carcinoma basocelular não sindrômico.
Métodos: Revisão da literatura nas principais bases de dados NCBI-GTR, ClinVar, ClinGen, MedGen, OMIM e GeneReviews , utilizando como descritores: "BCC" e "basal cell carcinoma ". Critérios de inclusão: língua portuguesa ou inglesa, artigos sobre CBC esporádico. Resultados: Foram selecionados treze artigos para análise. A análise revelou uma robusta ligação da via hedgehog na gênese do carcinoma basocelular esporádico, com os principais genes envolvidos representados por PATCH1, PATCH2 e smoothened . As variantes com maior significância clínica foram SMO-M2, PTCH1 e PTCH2-?22. A mutação mais encontrada fora a relacionada à ação do UVB, sendo representada pela substituição de C>T ou CC>TT no sítio das pirimidinas, tanto no PTCH, quanto no SMO.
Conclusão: Extremamente importante aos profissionais que atuam no diagnóstico e tratamento do CBC, dentre os quais os cirurgiões plásticos, pois assim poderão melhor conduzir seus casos, com diagnósticos mais precisos e condutas de prevenção baseadas na suscetibilidade individual de cada paciente, bem como terapêuticas direcionadas e individualizadas com melhores taxas de sucesso.
Palavras-chave: Genoma humano; Carcinoma basocelular; Neoplasia de células basais; Genes; Mutação
INTRODUCTION
Since ancient times, the greatest enigma represented by the origin of life has challenged philosophers and scientists and represented a great key for medicine in its purposes of understanding, treating, and curing ills. Many theories have been framed through the centuries; Charles Darwin, in his emeritus work “The Origin of Species” 1 in 1859, already lectured on the importance of knowledge about the constitution of organisms for evolution. In the following century, in the year 1920, Oparin and Haldane2 presented to the scientific community the theory of coacervates to explain the origin of life and how single-celled structures derived from organic compounds developed to foster beings and life as we know it at present times.
Conspicuous advances in knowledge about this motto occurred in the year 1990, with the official beginning of the Human Genome Project, which organized efforts of many countries in the search for the identification of all human genes and their sequences of base pairs3. This project, which ended in 2003, provided researchers with tools to understand thousands of diseases, including the syndromic diseases, as well as providing a myriad of possibilities of action for their treatment and prevention, and the understanding of how the replacement of a single nitrogen base could cause serious consequences to the organism.
Regimented in its results, it is important to save some data obtained in the first publication. The human genome contains 3.2 billion nucleotides; the average gene size is 3,000 bases, 50% of the discovered genes do not have their functions elucidated, only 2% of the genome encodes instructions for protein synthesis, 99.9% of the human genome sequence is exactly the same in all individuals, so that makes us unique represents 0.1% of our genome. Relevant information obtained was that half of the genome consists of repeated sequences that do not encode proteins and do not have known functions, but that help in understanding the structure and dynamics of chromosomes. It is suggested that these repetitions can reformulate the genome, rearranging it and thus creating new genes or modifying existing ones3.
Such knowledge framed the archetype to explain clinically observed facts in medical practice and the higher incidence of non-brain basal cell carcinoma (BCC) in patients with their own and family history of non-melanoma cutaneous neoplasia. Non-melanoma skin neoplasms represent the most frequent type in both sexes globally, with BCC being the most prevalent, representing 75 to 80% of cases4. In Brazil, the number of new cases expected for the triennium 2020-2022 will be 83,770 in men and 93,160 in women, corresponding to an estimated risk of 80.12 new cases for 100,000 men and 86.65 new cases for 100,000 women. The main risk factors are prolonged exposure to the sun in childhood and adolescence, exposure to the tanning chamber and family history of nonmelanoma skin neoplasia4.
Thus, because it represents great relevance to public health, expressed by the numbers mentioned above, it is necessary to understand the genomic bases that provide this greater predisposition to the development of BCC in individuals with family history, as well as the understanding of the action of ultraviolet radiation in genes, resulting in loss of suppressive function and, thus, propitiating the development of BCC and other non-melanoma skin neoplasms. Thus, this article portrays the current “state of the art” in the tangent to the knowledge of genes related to non-syndromic BCC, seeking to individualize preventive and therapeutic measures according to the genomic alterations found.
OBJECTIVE
Describe the main genes and molecular markers involved in the predisposition and pathogenesis of non-syndromic basal cell carcinoma.
METHODS
The design of the study is a review of the literature, carried out in the meantime from October to November 2020 in the main databases NCBI-GTR, ClinVar, ClinGen, MedGen, OMIM and GeneReviews, using as descriptors: “BCC” and”basal cell carcinoma”. Inclusion criteria: Portuguese or English, data only related to non-syndromic BCC. Exclusion criteria include languages other than those related to inclusion factors, repeated publications in different databases, publications older than ten years, data on synthetic BCC - e.g., Gorlin syndrome, nevoid basal cell carcinoma syndrome, xeroderma pigmentosum, etc.
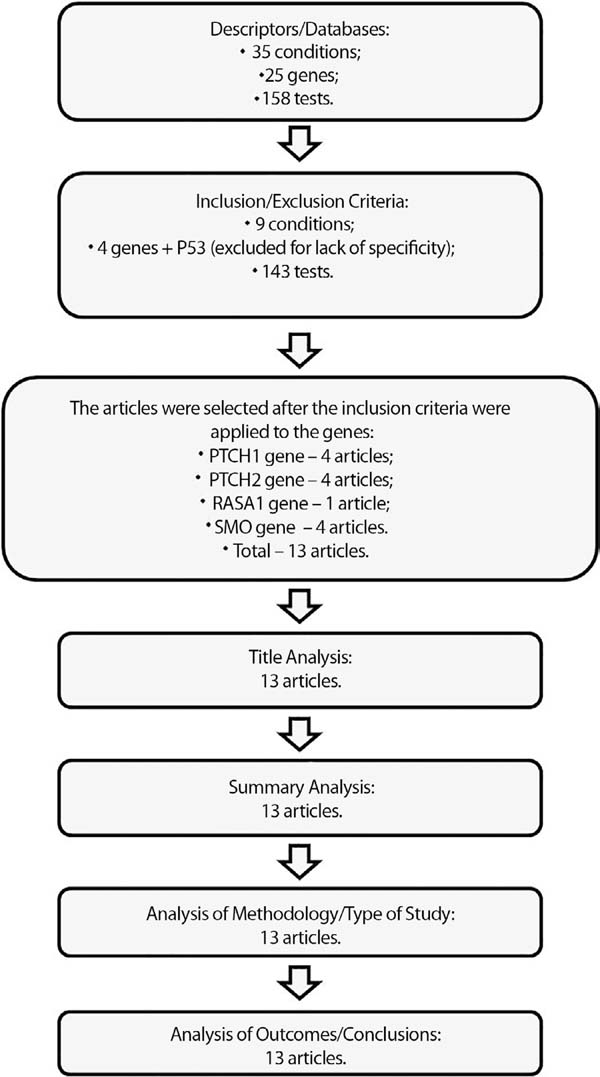
The results of the research were analyzed according to the flowchart (Figure 1) to adjust to inclusion/exclusion factors and subsequent analysis and tabulation of data. The articles that contemplated the determined methodology were selected for the study and discussion. Due to the heterogeneity of the databases and the results obtained, the results were separated according to the database into tests, conditions, genes and articles.
RESULTS
Preliminary, the search in the NCBI-GTR had returned thirty-five conditions, twenty-five genes and one hundred and fifty-eight tests for the descriptors used. After the first selection, there were nine conditions, five genes and one hundred and forty-three tests.
The articles were only researched after determining the genes, including those that presented as the motto the genes and the condition selected. A total of 13 articles were selected from this search because they met all inclusion criteria and, therefore, being part of this review.
Among the conditions presented, BCC1, BCC2, BCC3, BCC4, BCC5, BCC6, BCC7, BCC with follicular differentiation and BCC without specifications are listed.
On the tangent to the genes, PTCH1(patched), PTCH2(patched), RASA1 (RAS- MAPKinase), SMO (smoothened)and TP53 (Protein P53) returned. Therefore, performing the analysis of such data under inclusion criteria; there was only one condition left, the motto of this review - BCC1 that presented data on genes and tests, because the others do not have such information in the databases until this time, thus not contemplating the inclusion criteria.
Considering that the TP53 gene is not specific to BCC, being altered in various conditions and syndromes, besides being linked only to the BCC7 condition that was excluded because it did not circumscribe the criteria of choice, this gene was also excluded from the analyses of this review. Therefore, below are the summary tables with the articles (Chart 1), the tests (Charts 2, 3 and 4) and the selected genes (Charts 5, 6, 7 and 8).
| Articles included in the review | ||||
|---|---|---|---|---|
| Reference | Year publication |
Type of study |
Gene | Conclusion |
| Gailani MR, et al.5 | 1966 | Experimental (amostra tumoral) | PTCH1 | The study showed that most mutations resulted in “truncated” proteins. Scattered throughout the gene, 5 premature “Stop Codon” were found, 3 frameshift mutations. Changes that do not cause truncated proteins - 4 missense mutations and 1 deletion. Mutations typical of sun exposure were found by the action of UVB: C-T substitution at the pyrimidine site, including double base-mutation CC-TT. A C-G transversion mutation in an allele was found in a patient with no history of sun exposure. The PTCH1 gene has tumor suppressor action, a membrane protein with an intracellular portion, but without a mechanism still known. |
| Aszterbaum M, et al.6 | 1998 | Experimental (tumor sample and blood) | PTCH1 | The PTCH1 gene has somatic mutations in BCC. Encodes 12 transmembrane protein domains that are receptors of hedgehog ligands. They have an inhibiting function to signal such a pathway. In addition, they perform an inhibitory interaction to membrane SMO proteins (membrane G proteins). In BCC, there are three ways to its genesis: mutation in proto-oncogenes - SMO and SHH (expression increase) and/or mutation with inactivation of the PTCH1 tumor suppressor gene. Sporadic BCC requires a mutation in both alleles. The mutations found were: frameshift, premature stop codon, characteristic mutations of UVB action - C-T or CC-TT. Most mutations found had, as a consequence, truncated proteins, especially in the extracellular loop. |
| Zhang H, et al.7 | 2001 | Experimental (Lesion sample in patients under 30 years of age) | PTCH1 | As mutações encontradas foram: mudança simples de nucleotídeos, inserção de AT e deleção de 15-bp. Nucleotídeo alterados característicos de UVB C-T ou CC-TT tiveram grande prevalência no sítio da pirimidina. Na amostra somática encontradas mutações - nonsense, missense, deleção e inserção - todas com terminação prematura da proteína. Mutação do PTCH1 altera duas grandes proteínas extracelulares - loops e 12 domínios transmembranas de ligação da via hedgehog. Indivíduos jovens com mesmas alterações que indivíduos idosos, apresentam alterações em suas capacidades de reparo de DNA - p53, além de mutações no PTCH1. |
| Maglic D, et al.8 | 2018 | Experimental (tumor sample and blood) | PTCH1 | The main change caused by mutations in BCC development occurs in the hedgehog pathway, in its regulators - PTCH1 receptor, SMO (coupled G protein). Mutations instill the repressive function of PTCH1, releasing THE and promoting the growth factors of intranuclear GLI. BCCs that do not respond to MOS inhibitors escape such mutation at the drug binding site in THE OR with potentiation of GLI signaling. Hippo signaling is also related to the BCC. The hippo pathway outside the hedgehog acts through the YAP, but not the TAZ, to initiate the progression of the BCC, acting in JNK-JUN (phosphorylation) signaling; there is no impact on wnt or hedgehog. YAP works on wound healing, rapid recovery of basal cells and epidermis regeneration. There’s an increase in nuclear YAP in the BCC. YAP-TEAD-AP1 interaction is required for the initiation and maintenance of BCC. This pathway may be the cause of resistance to SMO inhibitors. |
| Smyth I, et al.9 | 1999 | Experimental (tumor sample and blood) | PTCH2 | Tumor suppressor gene homologous to PTCH1 located on chromosome 1. Presence of splice mutation at the donor site of the gene in BCC in exon 20. Not found in the other germ cells, only in the tumor sample. It has been shown that the inactivation of a PTCH2 allele is related to medulloblastoma and that a splice mutation is present in BCC since it is involved in tumor genesis. |
| Zaphiropoulos PG, et al.10 | 1999 | Experimental (tumor sample and blood) | PTCH2 | PTCH2 is 57% identical to PTCH1, with significant variation in transmembrane domains 6 and 7. It presents alternative Splice in exons 9 and 10 responsible for the extracellular loop and the transmembrane domains 2 and 3 of the PTCH1 protein structure. PTCH2, similar to PTCH1, acts as a tumor suppressor gene. PTCH2 has a distinct action from PTCH1 since its overexpression cannot compensate for the inactivation of PTCH1. |
| Rahnama F, et al.11 | 2004 | Experimental (tumor sample and blood) | PTCH2 | PTCH2 presents itself as a target of SHH-N signaling, promoting regulation, but I could not actively inhibit the SMO-M2, in contrast to the PTCH1. PTCH2 is 57% identical to PTCH1, diverging in the transmembrane hydrophilic region between domains 6 and 7. The fact that PTCH1 mutated in BCC cannot be compensated by the overexpression of PTCH2 implies that PTCH2 is related to the PTCH1 pathway, but PTCH2 has a weak inhibiting function when compared to PTCH1. PTCH2 has multiple variants, with ptch2-∆22 being those with clinical importance and a variant that Splice exons 9 and 10 and maintains exons 8 and 11 - junction - PTCH2-∆9,10. PTCH2, in its 3 variants, produce proteins in the cytoplasm - vesicular intracellular. PTCH2 can change the location of moS dispersed in the cytoplasm to a pattern of overlap with PTCH1 or PTCH2. There is the combination of PTCH2 and PTCH1 - they are interacting in 20% of immunoprecipitate analysis. PTCH2 acts in the internalization of the SHH-N. The presence of GLI1 linked to the site in the position between bases 472-463 of the ATG initiation codon demonstrates the direct effect of the signaling SSH on the activation of PTCH2. The inclusion of exons 9 and 10 and the exclusion of exon 22 are necessary for dose-dependent inhibition. PTCH2-∆22 does not decrease its inhibition of MOS when linked to SHH - demonstrating that only PTCH1 presents a ligand-dependent transcriptional response. |
| Fujii K, et al.12 | 2013 | Experimental (Tumor Sample and Blood) | PTCH2 | Germline heterozygosity with a missense mutation in PTCH2 identified in Nevoid BCC Syndrome - there is a frameshift 2-bp deletion (c.1172_1173del CT in exon 9) in PTCH2 creating a premature stop codon - resulting in a truncated PTCH2 protein - p. S391X. No mutations were found in PTCH1, SUFU and SMO in peripheral blood. |
| Friedman E, et al.13 | 1993 | Experimental (Tumor sample) (Tumor sample) | RASA1 | Three different mutations in BCC were found in the C-terminal SH2 of ras-GAP (catalytic domain). Suggest that such mutation results in a dysfunctional domain that has no interaction with modified phosphotyrosine. There is the loss of lysine in the SH2 part, losing interaction with phosphotyrosine, and there is an interaction with amino-aromatic grouping or causing hydrophobicity. Lysine is replaced by glutamate in BCC - altering the phosphotyrosine binding site. ras-GAP acts on the growth factors (e.g., PDGF) - family of tyrosine kinases. The system used to detect DGGE mutations may be flawed and less sensitive. It has no relevant action in BCC - a 50% reduction in GAP activity does not sufficiently increase GTP (guanosine-triphosphate). |
| Xie J, et al.14 | 1998 | Experimental | SMO | Activation of the SMO gene through somatic SMO missense mutation. This gene is a signaling component of the SHH-receptor complex and provides evidence that the mutation in SMO makes it an oncogene in BCC. Activation of the hedgehog pathway in BCC may result from failure of inhibition performed by PTCH or due to abnormal activation by a mutation in SMO. Found heterozygosity of missense mutation in base pair 1,685 - M1 changing Arg 562 (CGG) to Gln (CAG) - somatic origin, not present in the blood sample. It is hypothesized that the mutated SMO binds with PTCH and causes the inhibition of this cessation in other SMO receptors. Mutant SMO signaling occurs independently of ligands. There is effective signaling in the SHH cascade in the domain of zinc as a transcriptional factor of GLI. This gene is amplified in BCC, and its activity is increased. SMO-M2 is an intrinsic membrane protein; when overexpressed, it only acts on basal cells, with no action on other tissues. SMO is a proto-oncogene, being a target for BCC treatment. |
| Kunstfeld R15 | 2014 | Revision | SMO | Via hedgehog is identified as a key element for the development of various neoplasms. Inhibition of SMO is the best way to interfere with the hedgehog pathway. The hedgehog pathway is fundamental in embryonic development and typically becomes silent/inactive in adult tissues. The pathway is initiated by connecting one of the hedgehog ligands - Desert, Indian or Sonic in the PTCH1 transmembrane receiver. In the inactive form, PTCH exerts inhibition in SMO receptors, and no internal signaling occurs. When hedgehog ligands bind in PTCH, a relaxation in inhibition occurs and signaling by SMO begins, regulating the expression and transcription of factors - GLI 1-3. SMO inhibitor drugs: Vismodegib, Sonidegib, BMS-833923 (XLI139), Taladegib. Drugs that do not target MOS: Itraconazole (prevents the accumulation of MOS in the primary eyelash), Vitamin D3 (direct binding to OSS). |
| Zhang H, et al.16 | 2018 | Revision | SMO | Hedgehog has importance in embryonic development, control of cell maturation, differentiation and proliferation. In adult tissues is silent, except in maintenance and repair when necessary. The components of Via Hedgehog are 3 HH binders (S, D and I), two 12-transmembrane receptors - PTCH1 and PTCH2, a frizzled receptor of the g-protein class - SMO and 3 factors of the GLI 1-3 transcription cascade. The transduction of HH signaling begins in the primary eyelash - an organelle similar to an antenna that protrudes out of cells. The inhibitory effect of inactive PTCH prevents the translocation of THE to the primary eyelash. The complete GLI2/3 is negatively regulated by the SUFU protein and left in the form of GLI2/3Repressor, and the track signal is off. When HH ligands attach to PTCH1, relax the repression on the SMO and induce the primary eyelash to traffic with MOS. Activated SMO promoters perform GLI2/3FL dissociation from SUFU (GLI2/3R), forming GLI2/3A - activated, inducing GLI2/3A transport to the inside of the nucleus and target transcription triggers of the gene. 80% of BCCs mutate in PTCH1, with no changes in MOS, so they are responsive to MOS inhibitors. Resistance occurs with the missense mutation of The SMO in the Locus D473H - aspartic acid loss. Several mutations at the drug binding site and at the sites with significant affinity reduction for GDC-0449 (vismodegib). There is resistance caused by chromosomal amplification of the GLI2 effector cascade or overexpression of the phosphoinositide-3-kinase signaling pathway. The binding of MOS antagonists induces a conformational alteration in both the transmembrane and extracellular domains, demonstrating how they manipulate the activity of the SMO protein. The D473 mutation causes the loss of hydrogen bonds, reducing affinity to GDC-0449 to SMO. |
| Souza AM, et al.17 | 2020 | Case Study - Retrospective control (Patient samples with BCC and controls) | SMO | CpG-SNPs rs375350898 and rs75827493 showed significant association with BCC, and SNP
rs75827493 showed a relationship with nodular BCC. Thus, such SNPs have been shown
to be potential markers of susceptibility to BCC. The presence of SNPs in the CpG-promoting
region of the SMO gene can modify methylation and cause susceptibility to BCC. BCC
presents aberrant overregulation of the hedgehog pathway, typically with the loss
of PTCH1 and activation of SMO-protein G receptor, resulting in the deregulation of
GLI transcription and processes involving cell growth and proliferation factors. SNPs represent the region of the code that is associated with many diseases. The study of SNPs in promoter and intron regions is important to understand the mechanisms of genetic regulation in carcinogenesis. |
| Drug response test | |
|---|---|
| Test name | Laboratory |
| Basal cell carcinoma, somatic, 605462 (SMO gene) (sequence analysis-all coding exons)(postnatal) | Intergen Genetic Diagnosis and Research Centre |
| Basal cell carcinoma, somatic, 605462(Gorlin syndrome) (PTCH2 gene) (sequence analysis- all coding exons)(postnatal) | Intergen Genetic Diagnosis and Research Centre |
| Basal cell carcinoma, somatic, 605462(RASA1 gene) (sequence analysis-all coding exons)(postnatal) | Intergen Genetic Diagnosis and Research Centre |
| Basal cell carcinoma, somatic, 605462(PTCH1 gene) (sequence analysis-all coding exons)(postnatal) | Intergen Genetic Diagnosis and Research Centre |
Source: GTR = Genetic testing registry.
| Diagnostic test | |
|---|---|
| Test name | Laboratory |
| PTCH1 Gene sequencing & del/dup | GeneDx |
| PTCH1 Gene sequence and deletion/duplication | Ambry Genetics |
| PTCH1 Gene sequencing and deletion/duplication analysis (PTCH1) | DDC Clinic Molecular Diagnostics Laboratory |
| Single gene testing PTCH1 | CeGaT GmbH |
| RASA1 Single Gene | Fulgent Genetics |
| PTCH1 Single Gene | Fulgent Genetics |
| PTCH2 Single Gene | Fulgent Genetics |
Source: GTR = Genetic testing registry.
| Test for tracking | |
|---|---|
| Name of the test | Laboratory |
| PTCH2 | Institute for Human Genetics) |
| RASA1 | Institute for Human Genetics) |
Source: GTR = Genetic testing registry.
| PTCH 1 - GENE Exclusive variants of non-syndromic BCC: chromosome 9 (Source: ClinVar) |
||
|---|---|---|
| Base replacement | Altered proteins | Associated conditions |
| c.3340A>T(p.Arg1114Trp) | R1048W, R1114W, R963W, R1062W, R1113W | Basal cell carcinoma, somatic |
| 451C-T, PRO-SER | - | Basal cell carcinoma, somatic |
| PRO-SER | - | Basal cell carcinoma, somatic |
| PTCH 2 GENE - Exclusive variants of non-syndromic BCC: chromosome 1 (Source: ClinVar) |
||
|---|---|---|
| Base replacement | Altered proteins | Associated conditions |
| c.3357+5C>T | - | Basal cell carcinoma, somatic |
| RASA 1 Gene - Exclusive non-syndromic BCC variants: chromosome 5 (Source: ClinVar) |
||
|---|---|---|
| Base replacement | Altered proteins | Associated conditions |
| c.1193G>T (p.Arg398Leu) | R398L, R221L | Basal cell carcinoma, somatic |
| c.1198A>G (p.Lys400Glu) | K223E, K400E | Basal cell carcinoma, somatic |
| c.1201A>;G(p.Ile401Val) | I401V, I224V | Basal cell carcinoma, somatic |
| SMO Gene - Exclusive variants of non-syndromic BCC: chromosome 7 (Source: ClinVar) |
||
|---|---|---|
| Base replacement | Altered proteins | Associated conditions |
| c.1604G>T (p.Trp535Leu) | W535L | Basal cell carcinoma, somatic |
| c.1685G>A(p.Arg562Gln) | R562Q | Basal cell carcinoma, somatic |
DISCUSSION
The genetic heterogeneity related to susceptibility to the development of basal cell carcinoma denotes a great complexity in understanding all the genes and signaling pathways involved. It represents a great challenge to developing drugs and tests for early detection and/or pre-visualization, which are effective. This heterogeneity is characterized by subtypes: CBC1 - occurring on chromosome 1p36, CBC2 - on chromosome 1q42, CBC3 - 5q15, CBC4 - 12q13, CBC5 - 9p21 and CBC6 - 7q32. Variations in the early transcription region of TP53 increase susceptibility to BCC (BCC7). Somatic mutations, present only in lesions, not found in the constitutional cells of affected individuals, were identified in the RASA1, PTCH1 and PTCH 218genes.
Mutations are characterized by any stable alteration of the DNA chain. They can occur at three different levels: 1. molecular (genetic or point): affect the chemical constitution of genes, i.e., the nitrogenous bases of DNA; 2. chromosomal: a larger segment, involving more than one gene, is affected, it is not the affected constitution, but the structure; 3. genomics: mutations that affect the whole of the genome, increasing (polyploidy)or decreasing (haploid or monoploid) the total number of chromosomal games, or changing the number of chromosomes of each individual pair, by defector by excess 19-21.
Not representing the scope of the current review, a brief characterization of the main types of molecular or point mutations will be outlined. There is a change of DNA bases in the silent mutation, which leads the nucleotide triplet to differ from the normal sequence, although it codes the same amino acid. In polymorphism, there is a change of one of the DNA bases, altering the nucleotide triplet of which it is part and may or may not alter the corresponding amino acid. Even if there is a change of the amino acid by a distinct from the original code, there is little or no repercussion on protein function. In the studies enrolled and analyzed in this review, the mutations related to BCC genesis most found were missense, nonsense, and frameshift mutations. Missense mutations are characterized by the alteration of a single DNA base, altering the nucleotide triplet in which it occurs, having therefore encode an incorrect amino acid, different from what is expected in the corresponding position of the protein, and may lead to alteration of protein function to a greater or lesser degree, depending on the location and importance of the amino acid. However, in nonsense mutations, there is also a change in a single DNA base, changing the affected triplet. However, such change generates a termination codon - “stop codon”, that is, the nascent protein is truncated/cut prematurely, which, depending on where it occurs, may or may not preserve part of the protein’s function22.
In the second group of mutations, frameshift, inserts, dislocations, duplications, and expansions can be added by repetition. There is a change in the DNA reading grid in this group, greatly altering the transcription and translation. There is an addition of bases in the original DNA sequence in the insertions, which may result in the change in the reading grid or the insertion of an “extra” amino acid, which can alter the function of the protein or its activity. Dichotomically, the deletions are losses of one or more bases, a DNA trunk is lost, altering the protein chain that should be formed and its function. In some cases, the deletions are so extensive that they can compromise an entire gene or even several contiguous genes. In duplications, a fragment of DNA appears copied one or more times concerning the original DNA sequence. Thus, the protein reading grid can be changed, or the insertion of “extra” amino acids occurs, which are inadequate even if not changing the reading grid. Distinctly from the previous ones, in frameshift, by inserting or losing bases, the reading grid is always changed. In translation, the bases are read in triplets; every three bases determine an amino acid. Thus, by changing the reading grid, the way of grouping these three bases is changed and incorrect amino acids are included, with the possibility of forming a premature “stop codon” with the truncation of the protein. By ending, the expansions by repetition are characterized by the repetition of small DNA sequences of 3 or 4 pairs of bases repeated in series. Expansion mutation is a mutation in which the number of repetitions has increased, leading to the translation of a protein with altered or inactive function22.
Having as archetype the concepts mentioned above, this review outlined the current “state of the art” in the genomic bases of sporadic, non-syndromic BCC. The literature is robust in relating the hedgehog signaling pathway (HH) with the syndromic and sporadic BCC genesis. The HH pathway is extremely important in vertebrates’ embryogenesis, orchestrating the development, proliferation and conformation of multiple organs and systems. However, this pathway becomes silent in adult tissues, only being activated when there is a need for tissue repair16. The components of the HH pathway are characterized by 3 HH binders (S, D, and I), two 12- transmembrane receptors - PTCH1 and PTCH2, a frizzled receptor of the g-protein class - SMO and 3 factors of the GLI 1-3 transcription cascade. The transduction of HH signaling begins in the primary eyelash - an organelle similar to an antenna that protrudes out of cells. The inhibitory effect of inactive PTCH prevents the translocation of SMO to the primary eyelash. The complete GLI2/3 is negatively regulated by the SUFU protein (fused protein suppressor) and left in the form of GLI2/3R, and thus the signal of the track is off. When coupled to PTCH1, HH ligands relax repression on MOS and induce the primary eyelash to traffic/interact with SMO - promoters. Activated SMO promoters dissociate GLI2/3FL from SUFU (GLI2/3R), forming GLI2/3A - activated, inducing GLI2/3A transport to the inside of the nucleus and target transcription triggers of the gene, initiating protein synthesis16.
The analysis of the articles selected for review (Chart 1) revealed that the genes involved in the genesis, maintenance and/or development of BCC are in order of importance: PTCH1, SMO, PTCH2 and RASA1. Others are related but not specific to sporadic BCC as PT537. The role of such genes in the normal functioning of the HH pathway is above reported; already, the effects of mutations in these genes - creating their variants, below will be delineated. It should be emphasized that the RASA1 gene does not have much evidence in the literature. The mechanism of its action in BCC carcinogenesis is still unknown, being related to a change in the protein binding site of the tyrosine kinase family, which could intervene in growth factors, such as PDGF, enabling the stimulation of cell proliferation of BCC13. Its variants are listed in chart 7, and the chromosome involved is 5.
In the literature5-8, PTCH is a gene that encodes a protein with transmembrane, outer (loop) cellular and intracellular sections. It has clear action in the HH pathway, serving as a connecting site for the ligands of this pathway (Indian, desert and sonic) and presenting an activator role for this. The PTCH1 variant represents the most important of these proteins. When it is inactive, that is, without HH ligands, it exerts strong inhibition in SMO receptors, preventing them from interacting with the promoter region of the primary eyelashes. At the moment there is binding, in the case of CBC of the SHH ligand, there is a loss in the intensity of this inhibition, and the HH pathway is initiated. On the tangent of the variant PTCH29-12, there is still in the literature a complete elucidation of the mechanism of action in BCC carcinogenesis. It presents as a protein homologous to PTCH1 with the internalization function of SHH-N ligands such as PTCH1. It presents interaction with PTCH1 itself forming complexes, but there is no decrease in its inhibition to SMO as coupled to HH ligands. This determines that they are related to the HH pathway of PTCH1 but with a distinct action. Another aspect that corroborates this proposition is that in PTCH1 mutations with its loss of function, there is no compensation for PTCH2, and the genesis of BCC occurs. PTCH2 is 57% identical to PTCH1, diverging in the transmembrane hydrophilic region between domains 6 and 7. PTCH2 can change the location of SMO dispersed in the cytoplasm to a pattern of overlap with PTCH1 or PTCH211. The chromosomes involved are chromosome 9 in PTCH1 and chromosome 1 in PTCH2.
About the variants of these genes, the most found alteration was related to the action of UVB in the somatic cells of the lesions, i.e., a substitution (missense) of C>T or CC>TT at pyrimidine sites. Other mutations, with lower prevalence and lower clinical significance, have been described and summarized in tables 5 and 6, respectively, of the PTCH1 and PTCH2 genes. It should be noted that PTCH2 has multiple variants, but those with clinical significance are three: PTCH2; PTCH2-∆22 (exclusion of exon 22) and PTCH2-∆9,10 (inclusion of exons 9 and 10 per splice). Therefore, the variants determine the genesis of sporadic BCC by losing their functions of HH pathway suppressors, resulting in the pathway’s activation and its nuclear cascade and consequent basal cell proliferation.
SMO represents the gene of the greatest importance for pharmacogenetics. It is located on chromosome 7 and its related variants in Chart 8. It is the main target for drugs aimed at treating CBCs that do not respond to traditional therapies23-27. They are responsible for transporting HH ligands into the cellular interior through their interaction with the primary eyelashes, representing the true activators of the HH pathway, with constant activity without the inhibitory effect of PTCH. Therefore, they are considered proto-oncogenes. When they undergo mutation and are no longer inhibited by PTCH, activate the HH pathway and feed on PTCH inhibition, performing tumor genesis, thus characterized as oncogenes14-17. Mutant SMO signaling occurs independently of ligands. There is effective signaling in the SHH cascade in the zinc domain as a transcriptional factor of GLI. This gene is amplified in BCC, with its activity increased. SMO-M2, a variant with great clinical significance, intrinsic membrane protein, when in overexpression only acts on basal cells, with no action in other tissues. SMO is a proto-oncogene, being a target for BCC treatment14.
Activated SMO promoters perform GLI2/3FL dissociation from SUFU (GLI2/3R), forming GLI2/3A - activated, inducing GLI2/3A transport to the inside of the nucleus and target transcription triggers of the gene. The complete GLI2/3 is negatively regulated by the SUFU protein and left in the form of GLI2/3R (inactive/repressor), and the track signal is in silent/off state 16. The variants of SMO are SMO-M1 and SMO-M2(this being the one with clinical and therapeutic importance). Such variants present the ability of HH pathway actives without HH ligands in receptor promoters; thus, an amplification of the pathway occurs, which is associated with a loss of PTCH inhibition, as described above, resulting in the development of sporadic BCC. SMO is a “G-coupled protein” receptor (frizzled family)28, being related as possible markers of susceptibility to BCC development, besides being the best target for pharmacological therapies. CpG-SNPs rs375350898 and rs75827493 showed significant association with BCC, and SNP rs75827493 showed a relationship with nodular BCC. Thus, such SNPs are potential markers of susceptibility to BCC. The presence of SNPs in the CpG-promoting region of the SMO gene can modify methylation and cause susceptibility to BCC. This present aberrant overregulation of the hedgehog pathway, typically with the loss of PTCH1 and activation of SMO-protein G receptor, resulting in the deregulation of GLI transcription and processes involving cell growth factors proliferation17.
The myriad interactions necessary for neoplastic genesis can be portrayed by new pathways related to the HH pathway in the genomic bases of sporadic BCC. An example is found in the Hippo pathway (YAP-TED-AP1), which directly acts on intranuclear GLI promoters and seems to be related to the resistance of some BCC to SMO8 inhibitors. However, there are still not enough studies to confirm these findings to date.
Study limitations
Framed as a non-systematic literature review, the present has the biases and weaknesses inherent in this type of study. Moreover, the analyzed studies are mostly experimental without a control group and with analysis of a small number of samples, as well as there is no homogeneity and randomization of the participants, that is, no distinction of ethnicity, Fitzpatrick, gender, age, environmental exposure, origin, labor, use of medications (e.g., hydrochlorothiazide), comorbidities, etc. Such considerations may lead to biases and impair the analysis. The mutation analysis methods used in most studies do not have good sensitivity to detect some types of variations, impairing the analysis of this review.
CONCLUSION
Sporadic BCC represents an imminent health problem, representing the most prevalent type of neoplasia and causing a great impact on the public health system. The present review presents robust literature demonstrating the importance of genomic knowledge of this disease for better therapies, prevention, and screening of susceptibility. Furthermore, drugs developed based on this knowledge are important means of treatment for patients who do not have surgical conditions and/or do not respond to radiotherapy. In this respect, drugs such as vismodegib, itraconazole, vitamin D3, and another phase I and II study23-27 will be responsible for curing these patients soon, in addition to immunomodulatory drugs and gene therapy.
For all those mentioned above, it is extremely important for professionals working in the diagnosis and treatment of BCC, including plastic surgeons, essential and most challenged by this condition, to gather knowledge about the genomic bases of this pathology, as this way they can better conduct their cases, with more accurate diagnoses. Prevention conducts based on the individual susceptibility of each patient, as well as targeted and individualized therapies with better success rates.
COLLABORATIONS
|
DSSR |
Analysis and/or data interpretation, Conception and design study, Conceptualization, Data Curation, Final manuscript approval, Formal Analysis, Investigation, Methodology, Project Administration, Realization of operations and/or trials, Resources, Supervision, Visualization, Writing - Original Draft Preparation, Writing - Review & Editing. |
REFERENCES
1. Browne J. A origem das espécies de Darwin: uma biografia. Rio de Janeiro: Editora Zahar; 2007.
2. Dick SJ. The biological universe. Netherlands: Springer; 1999.
3. Department of Energy, Genomics and Its Impact on Science and Society (US). Human genome program project information archive - 1990-2003 [Internet]. Washingtion: 2008. Disponível em: http://www.ornl.gov/sci/techresources/Human_Genome/publicat/primer2001/4.shtml
4. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Estimativa 2020: incidência de câncer no Brasil. Rio de Janeiro: INCA; 2019.
5. Gailani MR, Ståhle-Bäckdahl M, Leffell DJ, Glynn M, Zaphiropoulos PG, Pressman C, et al. The role of the human homologue of Drosophila patched in sporadic basal cell carcinomas. Nat Genet. 1996 Set;14(1):78-81.
6. Aszterbaum M, Rothman A, Johnson RL, Fisher M, Xie J, Bonifas JM, et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol. 1998 Jun;110(6):885-8.
7. Zhang H, Ping XL, Lee PK, Wu XL, Yao YJ, Zhang MJ, et al. Role of PTCH and p53 genes in early-onset basal cell carcinoma. Am J Pathol. 2001 Fev;158(2):381-5.
8. Maglic D, Schlegelmilch K, Dost AF, Panero R, Dill MT, Calogero RA, et al. YAP-TEAD signaling promotes basal cell carcinoma development via a c-JUN/AP1 axis. EMBO J. 2018 Set;37(17):e98642.
9. Smyth I, Narang MA, Evans T, Heimann C, Nakamura Y, Chenevix-Trench G, et al. Isolation and characterization of human patched 2 (PTCH2), a putative tumour suppressor gene in basal cell carcinoma and medulloblastoma on chromosome 1p32. Hum Mol Genet. 1999 Fev;8(2):291-7.
10. Zaphiropoulos PG, Undén AB, Rahnama F, Hollingsworth RE, Toftgård R. PTCH2, a novel human patched gene, undergoing alternative splicing and up-regulated in basal cell carcinomas. Cancer Res. 1999 Fev;59(4):787-92.
11. Rahnama F, Toftgård R, Zaphiropoulos PG. Distinct roles of PTCH2 splice variants in Hedgehog signalling. Biochem J. 2004 Mar;378(Pt 2):325-34.
12. Fujii K, Ohashi H, Suzuki M, Hatsuse H, Shiohama T, Uchikawa H, et al. Frameshift mutation in the PTCH2 gene can cause nevoid basal cell carcinoma syndrome. Fam Cancer. 2013 Dez;12(4):611-4.
13. Friedman E, Gejman PV, Martin GA, McCormick F. Nonsense mutations in the C-terminal SH2 region of the GTPase activating protein (GAP) gene in human tumours. Nat Genet. 1993 Nov;5(3):242-7.
14. Xie J, Murone M, Luoh SM, Ryan A, Gu Q, Zhang C, et al. Activating smoothened mutations in sporadic basal-cell carcinoma. Nature. 1998 Jan;391(6662):90-2.
15. Kunstfeld R. Smoothened inhibitors in the treatment of advanced basal cell carcinomas. Curr Opin Oncol. 2014 Mar;26(2):184-95.
16. Zhang H, Sun Z, Liu Z, Song C. Overcoming the emerging drug resistance of smoothened: an overview of small-molecule SMO antagonists with antiresistance activity. Future Med Chem. 2018 Dez;10(24):2855-2875.
17. Souza AM, Lopes OS, Liberato AL, Oliveira PJR, Herrero SST, Nascimento ALD, et al. Association between SNPs and loss of methylation site on the CpG island of the promoter region of the smoothened gene, potential molecular markers for susceptibility to the development of basal cell carcinoma in the Brazilian population. Asian Pac J Cancer Prev. 2020 Jan;21(1):25-29.
18. Hamosh A, Ramussen SA; Online Mendelian Inheritance in Man (OMIM). Basal cell carcinoma, susceptibility to 1 BCC1 [Internet]. Baltimore: OMIM/Johns Hopkins University; 2000. Disponível em: https://omim.org/entry/605462?search=BCC&highlight=bcc
19. Leroi A. Mutants: on the form, varieties & errors of the human body. London: HarperCollins; 2003.
20. Maki H. Origins of spontaneous mutations: specificity and directionality of base-substitution, frameshift, and sequence-substitution mutageneses. Annu Ver Genet. 2002;36:279-303.
21. Taggart R. Starr C. Biology the unity and diversity of life: mutated genes and their protein products. Pacific Grove: Thompson Brooks/Cole; 2006.
22. Universitat de Barcelona (ES). Sant Joan de Déu - Hospital Materno Infantil. Guía metabólica - tipos de mutações [Internet]. Barcelona: Sant Joan de Déu. Disponível em: https://metabolicas.sjdhospitalbarcelona.org/sites/default/files/tipos_de_mutacoes_ptg.pdf
23. Lacroix C, Fish I, Torosyan H, Parathaman P, Irwin JJ, Shoichet BK, et al. Identification of novel smoothened ligands using structure-based docking. PLoS One. 2016 Ago;11(8):e0160365.
24. Byrne EFX, Sircar R, Miller PS, Hedger G, Luchetti G, Nachtergaele S, et al. Structural basis of smoothened regulation by its extracellular domains. Nature. 2016 Jul;535(7613):517- 522.
25. Atwood SX, Sarin KY, Whitson RJ, Li JR, Kim G, Rezaee M, et al. Smoothened variants explain the majority of drug resistance in basal cell carcinoma. Cancer Cell. 2015 Mar;27(3):342-53.
26. Pricl S, Cortelazzi B, Dal Col V, Marson D, Laurini E, Fermeglia M, et al. Smoothened (SMO) receptor mutations dictate resistance to vismodegib in basal cell carcinoma. Mol Oncol. 2015 Fev;9(2):389-97.
27. Sharpe HJ, Pau G, Dijkgraaf GJ, Basset-Seguin N, Modrusan Z, Januario T, et al. Genomic analysis of smoothened inhibitor resistance in basal cell carcinoma. Cancer Cell. 2015 Mar;27(3):327-41.
28. GeneCards - The Humam Gene Database. Homepage [Internet]. Rehovot: Weizmann Institute of Science; 1996. Disponível em: https://www.genecards.org/cgi-bin/carddisp.pl?gene=SMO
1. Federal University of São Carlos, Postgraduate Program - PPGBiotec, São Carlos,
SP, Brazil.
2. Brazilian Society of Plastic Surgery, São Paulo, SP, Brazil.
3. Sundfeld Institute of Plastic Surgery, São Carlos, SP, Brazil.
Corresponding author: Daniel Sundfeld Spiga Real, Rua Dr. Domingos Faro, nº 285 - Jardim Alvorada, São Carlos, SP, Brazil, Zip Code 13562-003. E-mail: dplasticsurgery@hotmail.com
Article received: November 26, 2020.
Article accepted: April 23, 2021.
Conflicts of interest: none.












 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter