

Special Article - Year 2021 - Volume 36 -
Nasal reshaping with hyaluronic acid: technique, risks, and benefits
Rinomodelação com ácido hialurônico: técnica, riscos e benefícios
ABSTRACT
Rhinoplasty is a common procedure performed by plastic surgeons. Nasal reshaping with hyaluronic acid (HA) is a fast and straightforward procedure that does not involve distancing recovery activities while providing results comparable to surgical rhinoplasty. Indications for nasal filling include patients who want to "test" the result of rhinoplasty, patients undergoing rhinoplasty who do not wish to further surgery to review a deformity, patients who are not candidates for surgery, and patients waiting for the appropriate time interval before undergoing a secondary rhinoplasty. The nose is the subunit of the face most at risk for fillings after glabella. As the number of patients submitted to fillings increases, so does the number of associated adverse effects.
Keywords: Hyaluronic acid; Rhinoplasty; Dermal fillers; Aesthetics; Postoperative complications
RESUMO
Rinoplastia é um procedimento comum realizado pelos cirurgiões plásticos. A rinomodelação com ácido hialurônico (AH) é um procedimento rápido e simples que não envolve afastamento das atividades na recuperação, ao mesmo tempo que fornece resultados comparáveis aos da rinoplastia cirúrgica. As indicações para preenchimento nasal incluem: pacientes que querem "testar" o resultado de uma rinoplastia, pacientes submetidos à rinoplastia que não desejam nova cirurgia para revisão de uma deformidade, pacientes que não são candidatos para cirurgia e pacientes que aguardam o intervalo de tempo adequado antes de passar por uma rinoplastia secundária. O nariz é a subunidade da face de maior risco para preenchimentos depois da glabela. À medida que aumenta o número de pacientes submetidos a preenchimentos, também aumenta o número de efeitos adversos associados.
Palavras-chave: Ácido hialurônico; Rinoplastia; Preenchedores dérmicos; Estética; Complicações pós-operatórias
INTRODUCTION
Rhinoplasty remains one of the most common aesthetic procedures1. More recently, technological advances in injectable products based on hyaluronic acid (HA) and refinements of application techniques have allowed HA to reach the gold standard as a volumizing agent2. Nasal reshaping with HA is a fast and simple procedure that does not involve distancing recovery activities while providing results comparable to conventional rhinoplasty3. The most common applications have been the correction of deformities of the tip, back, and columella4.
Indications for nasal filling include patients who want to “test” the result of rhinoplasty, patients undergoing rhinoplasty who do not wish to further surgery to review a residual deformity, patients who are not candidates for surgery, and patients waiting for the appropriate time interval before undergoing a secondary rhinoplasty5,6. The nose is the subunit of the face most at risk for fillings after glabella. As the number of patients submitted to fillings increases, so does the number of associated adverse effects.
OBJECTIVES
According to the authors ‘ experience, this study aims to demonstrate a safe technique, highlighting the anatomical knowledge and problems involved in nasal reshaping in a standard case report.
METHODS
The main techniques used for Nasal reshaping are bolus and retroinjection. For safety, we strongly suggest that this procedure be preferably performed with cannulas. We do not use needles to inject into the nose. Injections should be performed deep in the musculoaponeurotic layers and suprapericondral and supraperiostal layers to avoid injury or cannulation of the vessels (which are subdermal in this region) providing natural results and with greater safety.
In secondary rhinoplasty, extreme caution should be taken. Unpredictable blood vessel repositioning and a more tenuous blood supply in the operated nose may increase the risk of ischemia, necrosis, and vascular embolism after filler injection. Anatomical planes may have been violated or healed. The dermis may be adhered to deep planes; besides, natural anastomoses between contralateral vessels may no longer be present.
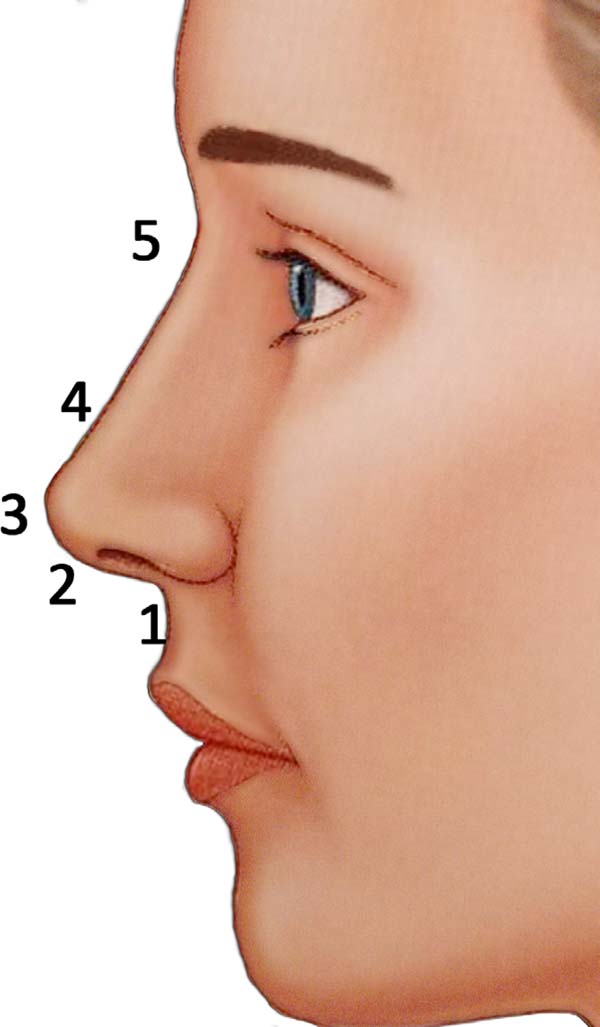
The amount of HA injected per patient is variable. On average, the total quantities range from 0.6 to 2ml. The desired modifications are elevation of the nasal tip, increased nasolabial angle, and correction of nasal dorsum irregularity with nasal root repositioning. These modifications will be described below:
- The first step is to photograph the patient. Frontal, lateral, and
caudocranial incidences are essential. Images should be standardized
to help evaluate results;
- The nose is one of the areas most colonized by bacteria. Extra care
in the antisepsis is mandatory for the safety of this procedure;
- Intraoral access is used to block the infraorbital nerve with 2%
lidocaine without epinephrine. This time, tiny additional anesthetic
buds using lidocaine with epinephrine are injected into the nasal
tip and nasolabial angle. Anesthetics with epinephrine help reduce
the risk of peri-cannula bleeding. Remember that there will be a
halo of pallor due to the vasoconstrictor;
- The cannulas should be long enough to reach from the tip to the
nasal root. In general, 50mm in length is a good measure. Our
preference is for the 22G 50mm cannulas;
- In a single step, with the entry point at the nasolabial angle (the
paramedian intake is better than the median, so the syringe does not
touch the chin), we start increasing the columella-labial angle. The
cannula is advanced along the subcutaneous plane to the nasal spine.
Then, the HA is injected slowly, carefully observing the filling of
the columella-labial angle (Figure 1 - step 1);
Once the nasolabial angle is corrected, the cannula is previously oriented to strengthen and rectify the columella. This maneuver straightens, lengthens, and supports the columella. This should produce a more anterior tip projection with less cranial rotation. When stretching the columella, the nose should appear more isosceles and the nostril more tear-shaped when viewed from the basal angle (Figure 1 - step 2);
• In a single step, with the entry point still at the
nasolabial angle, the third stage begins with the tip’s projection.
A single bolus is sufficient for this purpose
(Figure 1 - step3(;
• The last step is to correct the back. Injections before and
after the comic can disguise it. Subsequent injection projects the
root. The final point is to create a new root at the approximate
level of the supratarsal fold, to create an angulation of
approximately 135 degrees of the nasal dorsum with the forehead, and
to create a new nasal profile, more aesthetically pleasing (Figure 1 - steps 4 and 5);
- Cohesive gels can be easily molded and remodeled to sculpt the
nose, such as “modeling mass” immediately after injection.
Additional syringes may be required at this stage;
- In all areas, always apply deep injections; - In our technique, we start with the columella enhancement, then the
tip, and finish with the back;
- The right hand is used for smooth, measurable, and directional fill
injection. The left hand is used to guide placement, shaping, and
avoid inadvertent spreading of the injected load. This is
particularly important when injecting the dorsum since the lack of
accuracy of the injected filler creates a noticeable asymmetry;
- Slow and constant correction provides the safest way to achieve the
best results;
- Compression of the nasal and dorsal upper part of the angular
arteries is also recommended. It should always be injected more than
2 to 3 mm above the alar groove to avoid the lateral nasal
artery7;
- Stop injection immediately if there are ischemic changes in the
skin of the nose;
- Other additional fixes are always possible at a later date; - Avoid large volumes of superficial injections as this can cause
external vascular compression, which can cause ischemia and necrosis
of the skin. This is especially important at the tip and nasal
base;
- Closely monitor the nose after the procedure for signs of ischemia,
particularly in those patients with a history of the previous
rhinoplasty, as their vascular supply may be distorted and
compromised;
- Avoid unnecessary external compression of the nose after injection,
such as wearing glasses, for at least a few days;
- A small dressing is added at the entry points at the end of the
procedure to avoid ha reflux and the consequent increased risk of
fistulization.
RESULTS
One of the authors developed this technique (Fernandes RL) in 2013, and since then, it has been performed in approximately 60 patients. Results range from good to very good in almost all patients. The duration of the effect of correction of the dorsal gibbon is significant, with an average between 12 and 18 months. The duration of the tip lift is shorter, usually half that time. Pain is considered mild to moderate using this technique. The most common side effects are fistula (or HA vesicle) at the cannula’s entry points and persistent erythema of the nose. No serious adverse events were observed using this technique.
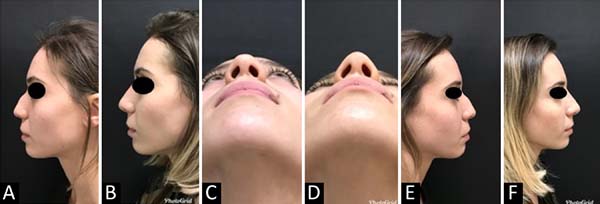
MCB, 28 years old, 1.2 ml HA (Teosyal Ultra Deep® - Teoxane Laboratory - Genéve) with 22G cannula - 50mm (Figures 2,3 and 4).
DISCUSSION
Anatomy
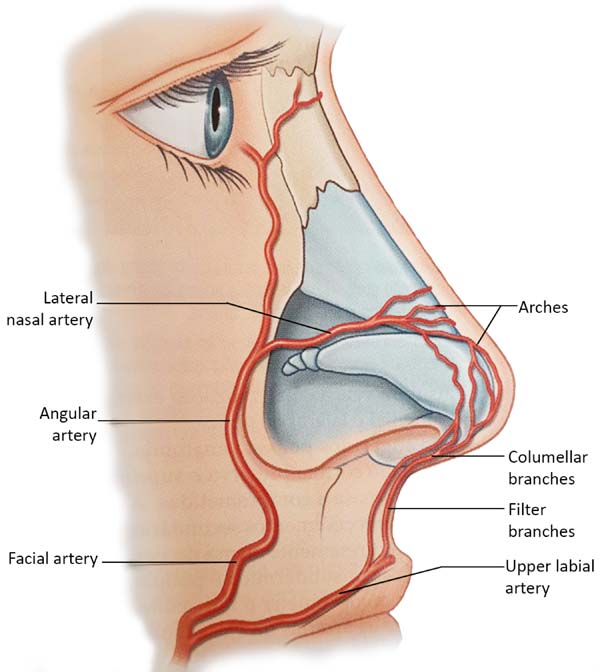
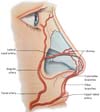
The extensive collateral blood supply of the nose makes this procedure relatively safe. Both a branch of the internal carotid artery, the supratroclear, and a branch of the external carotid artery, the facial artery, give rise to branches that cross the midline. These form a vascular network that runs through the back. Along the way, inferior to the nose, the facial artery originates the upper labial artery, which also originates the filter’s arteries, providing the ascending columellar arteries’ main contribution. Several arches arise from both the supratroclear arteries and the facial arteries (Figure 5); the lateral nasal artery is one of the main sources of blood supply to the nose.
Tansatit et al., in 20168, studied 50 cadaver noses and showed that the dorsal artery of the nose is not a constant artery. It is present as a pair of arteries in 34%. The typical pattern of the nose’s dorsal artery is a large and long artery that descends through the back or side of the nose and is anastomosed with the lateral nasal artery on one side or both. The lateral nasal artery (LNA) is a constant branch of the alar groove’s facial artery. It represents an anastomosis between the facial artery (FA) and ophthalmic artery (OA) in the paracentral zone of the middle third of the face. In 28% of the cadavers, a single and sizeable dorsal artery was presented8. The study used 57 adult hemifaces to allow precise observation of the branches of the facial artery. Four patterns were identified based on the detailed course and origin of the angular artery (AA) concerning the surrounding structures. In type I, the persistent pattern in which the AA traverses the LNA branch point toward the medial corner area. In type II, the deviation pattern, the AA originates from FA near the corner of the mouth and then heads towards the infraorbital area, finally rotating medially along with the nasal areas nasojugal corner. This pattern was the predominant type, and AA emerged along with the lower margin of the eye’s orbicularis. In type III (alternative pattern), AA originates from OA and runs down along the nose’s side. Finally, in type IV (latent pattern), AF ends up as ALN without producing an AA branch. In summary, AA originated from FA in 50.9% of the specimens dissected with persistent (type I) and deviation (type II) patterns 8.
Venous drainage of the nose consists mainly of vessels anastomosed with the facial vein, either through veins that travel from the back and lateral nasal wall or through vessels that accompany the filter and upper labial vessels8. The vascularization of the nose is superficially located below the dermis.
Fillers
Fillers are volumizing biomaterials that are injected into dermal and/or subcutaneous tissues for various reconstructive and cosmetic purposes, especially on the face9. The use of injectable fillers has skyrocketed in the last 25 years. The American Society of Plastic Surgeons reports an increase from 650,000 filling procedures in 2000 to 2.3 million in 20144. Hyaluronic acid (HA) injections comprise most of these procedures (80%), followed by calcium hydroxyapatite (CaHA) (10%), polylactic acid (5%), and autologous fat (3%)9.
From the 1990s to today, HA fillers have evolved technologically. They have improved durability, versatility (gels with distinct rheological characteristics and different tissue expansion capabilities), and safety. These changes allowed HA to become a good volumizing agent10.
There is no identical or similar HA filler if we compare different brands. The rheological characteristics of each product are unique. They differ among the most diverse products, mainly depending on the following features: being single-phase or biphasic, the molecular weight of its HA chains, the technology and its degree of crosslinking, and the concentration of HA.
The main rheological parameters are viscosity, cohesiveness, and elasticity. Viscosity is the spreading capacity of the gel. The more viscous, the smaller the tissue spread. Surface applications require less viscous gels to make results more natural without irregularities.
Cohesiveness is the resistance capacity of the gel to shear. The more cohesive, the more united its structure remains when subjected to external pressure. We give preference to the more cohesive HAs when we want to get better-defined forms.
Elasticity is the ability to resist deformation when subjected to external pressure. Deep planes are looser and require more elastic HAs for better tissue expansion.
Of course, there is no ideal filler, but we recommend high viscosity, cohesive, and elasticity HA if we want to perform a non-surgical rhinoplasty effectively and safely. They are known as volumizers and are indicated for subdermal application.
Adverse effects
The potential adverse events associated with filling are injection site reaction, inappropriate injection (hypercorrection, nodulation, asymmetry), product sensitivity, infection, and necrosis.
Although most complications are transient, some irreversible ones can cause severe functional and aesthetic deficits1. Complications, fortunately, are not typical and range from hematomas, edemas, and late granulomatous reactions to more severe skin necrosis11,12. Necrosis of the nose’s tip’s skin is particularly worrisome in the procedure, as it inevitably leads to permanent disfigurement. However, the nasal dorsum correction without considering the tip’s correction does not produce a comprehensive aesthetic improvement.
Inadvertent injection of intravascular filler would lead to irreversible necrosis of the skin. If an artery compression causes ischemia, it can eventually be reversed by dissolving the filling of AH13,14. Due to this reason, in case of use of needles, we recommend the filling injection only after the aspiration test, and we recommend that you observe closely immediately after rhinoplasty with HA and be ready to inject hyaluronidase.
Intermittent swelling followed by the development of palpable and/or painful nodular papulocystic lesions, from weeks to months after the injection, can progress to aseptic abscesses, the most common evolution being drainage through a fistula. These reactions usually occur after patients have their second or third injection. The histopathological analysis may show non-granulomatous inflammatory infiltrate (chronic suppurative inflammatory process with eosinophilia) or granulomatous reactions.
The mechanisms underlying the activation of the immune system and leading to chronic granuloma formation are still unclear. Various agents, including biomaterials, can trigger granulomatous reactions. These late reactions related to HA fillers are immunological in nature, but an infectious origin cannot be ruled out. It is important to differentiate two possible bacterial presence sources: 1 - bacteria are directly inoculated in the filling material or reach the filler from distant locations; 2 - systemic or remote infection can provoke inflammatory immunomediator reactions harmful to fillers in the absence of bacterial colonies in the filler. Clinically, a nodule of consistency and late-onset may suggest a granulomatous response. However, granuloma is a pathological diagnosis. True granuloma appears late (especially after 6 to 24 months) at all injected sites at approximately the same time; they grow quite fast.
The initial adverse effects described above tend to disappear within one or two weeks spontaneously with symptomatic treatment. In cases of severe or persistent swelling requiring corticosteroid use, betamethasone (0.05mg/kg/day) is preferable due to its higher mineralocorticoid action (antiedema) compared to the others.
Persistent hypercorrection can be treated early by incision and drainage. A blade is inserted directed to the nodule, and an expression is performed.
Hypersensitivity reactions usually regress without sequelae with the triple therapeutic regimen: hyaluronidase injection (once a week while the reaction persists) + antibiotics (macrolides such as clarithromycin or lymphomycins such as clindamycin) for 14 to 21 days + oral prednisolone (0.5mg/kg/day, while the reaction persists).
Due to frequent recurrence, corticosteroid treatment can last a long time. All precautions related to corticosteroid side effects should be taken: chest X-ray, bone densitometry, serum lipid dosage, blood pressure, and blood glucose monitoring should be considered. For treatment for more than three months, ophthalmological evaluation and supplementation of calcium carbonate (1.5g per day) and sodium alendronate (70mg per week) is recommended.
True granuloma usually reacts well to intralesional steroid injections (triamcinolone acetonide), despite or associated with oral corticosteroid ingestion.
The concomitance of HA reactions and other infectious conditions nearby is quite common. Investigation of periodontal disease and chronic sinusitis should be encouraged, especially in suggestive signs and symptoms.
The possibility of intravascular injection blindness is described in the literature, but cases with HA showed better results than other fillers due to hyaluronidase use, according to a meta-analysis study15. Another meta-analysis analyzed the cases described and found that most were unilateral cases with acute visual symptoms and signs, with a better prognosis in patients with partial loss of vision and cases of anterior artery branch obstruction, with a worse prognosis of complete blindness and obstructions in the central retinal artery or ophthalmic artery16. In arterial embolization cases, the immediate application of hyaluronidase and the emergency evaluation of a specialist in angiology and vascular surgery (possibly also an ophthalmologist qualified for retrobulbar injection) is the best way to minimize sequelae.
CONCLUSION
We are in the midst of a new era of rhinoplasty, in which surgery is not the only means to treat heart defects17. Non-surgical options seem more feasible than they would be before the advent of the new synthetic fillers18. In the literature, however, there are few prospective studies focused on the efficacy, safety, and longevity of HA fillers to support their usefulness as a non-surgical alternative to rhinoplasty. Several rhinoplasty surgeons have used fillers in the nose for many years, recognizing that HA can accurately smooth irregularities and asymmetries in the nose after aesthetic rhinoplasty. Indeed, the ability to smooth irregularities and asymmetries in the nose with an injectable material still has great appeal because imperfections after rhinoplasty are common.
The main advantage of using fillers in the nose is correcting a deformity without the financial cost, anesthetic risk, or downtime usually associated with surgical intervention. Disadvantages include potential damage to the nasal skin envelope, the need for serial treatments to maintain correction, and a decrease in the surgeon’s impulse to achieve the perfect intraoperative outcome5. Fear of occlusion or vascular compression is undoubtedly the most threatening. However, we believe that by following the simple steps of safety and having refined anatomical knowledge, fillers can be a good tool for a safe and comprehensive improvement of modeling rhinoplasty.
REFERENCES
1. Robati RM, Moeineddin F, Almasi-Nasrabadi M. The risk of skin necrosis following hyaluronic acid filler injection in patients with a history of cosmetic rhinoplasty. Aesthet Surg J. 2018 Jan;38(8):883-8.
2. Fernandes RL. Hyaluronic acid filler for the malar area. In: Issa MCA, Tamura B, eds. Botulinum toxins, fillers and related substances. Cham: Springer International Publishing; 2019. p. 271-80.
3. Youn SH, Seo KK. Filler rhinoplasty evaluated by anthropometric analysis. Am Soc Dermatol Surg. 2016 Ago;42(9):1071-81.
4. Thomas WW, Bucky L, Friedman O. Injectables in the nose. Facial Plast Surg Clin North Am. 2016 Ago;24(3):379-89.
5. Humphrey CD, Arkins JP, Dayan SH. Soft tissue fillers in the nose. Aesthet Surg J. 2009;29(6):477-84.
6. Kurkjian TJ, Ahmad J, Rohrich RD. Soft-tissue fillers in rhinoplasty. Plast Reconstr Surg. 2014 Fev;133(2):121-6.
7. Scheuer JF, Sieber DA, Pezeshk RA, Gassman AA, Campbell CF, Rohrich RJ. Facial danger zones: techniques to maximize safety during soft-tissue filler injections. Plast Reconstr Surg. 2017 Mai;139(5):1103-8.
8. Tansatit T, Apinuntrum P, Phetudom T. Facing the worst risk: confronting the dorsal nasal artery, implication for non-surgical procedures of nasal augmentation. Aesthetic Plast Surg. 2017 Fev;41(1):191-8.
9. Wang LL, Friedman O. Update on injectables in the nose. Curr Opin Otolaryngol Head Neck Surg. 2017 Ago;25(4):307-13.
10. Williams LCBA, Kidwai S, Mehta K, Kamel G, Tepper O, Rosenberg J. Nonsurgical rhinoplasty: a systematic review of technique, outcomes, and complications. Plast Reconstr Surg. 2020 Jul;146(1):41-51. DOI: https://doi.org/10.1097/PRS.0000000000006892
11. Johnson ON, Kontis TC. Nonsurgical rhinoplasty. Facial Plast Surg. 2016;32(5):500-6. DOI: https://doi.org/10.1055/s-0036-1586209
12. Alam M, Dover JS. Management of complications and sequelae with temporary injectable fillers. Plast Reconstr Surg. 2007 Nov;120(6 Suppl 1):98S-105S.
13. Daher JC, Silva SV, Campos AC, Dias RCS, Damasio AA, Costa RSC. Complicações vasculares dos preenchimentos faciais com ácido hialurônico: confecção de protocolo de prevenção e tratamento. Rev Bras Cir Plást. 2020;35(1):2-7.
14. Moon HJ. Use of fillers in rhinoplasty. Clin Plast Surg. 2016 Jan;43(1):307-17.
15. Chatrath V, Banerjee PS, Goodman GJ, Rahman E. Soft-tissue filler-associated blindness: a systematic review of case reports and case series. Plast Reconstr Surg Glob Open. 2019 Abr;7(4):e2173.
16. Kapoor KM, Kapoor P, Heydenrych I, Bertossi D. Vision loss associated with hyaluronic acid fillers: a systematic review of literature. Aesthetic Plast Surg. 2020 Dez;44(3):929-44. DOI: https://doi.org/10.1007/s00266-019-01562-8
17. Ramos RM, Bolivar HE, Piccinini PS, Sucupira E. Rinomodelação ou rinoplastia não-cirúrgica: uma abordagem segura e reprodutível. Rev Bras Cir Plást. 2019;34(4):576-81.
18. Jasin ME. Nonsurgical rhinoplasty using dermal fillers. Facial Plast Surg Clin North Am. 2013 Mai;21(2):241-52.
1. Institute of Plastic Surgery Santa Cruz,
São Paulo, SP, Brazil.
ACF Analysis and/or data interpretation, Conception and design study, Conceptualization, Data Curation, Final manuscript approval, Formal Analysis, Funding Acquisition, Investigation, Methodology, Project Administration, Realization of operations and/or trials, Resources, Supervision, Validation, Visualization, Writing - Original Draft Preparation, Writing - Review & Editing
BOB Analysis and/or data interpretation, Conceptualization, Final manuscript approval, Formal Analysis, Project Administration, Realization of operations and/or trials, Resources, Supervision, Validation, Visualization, Writing - Original Draft Preparation, Writing - Review & Editing
GHCT Analysis and/or data interpretation, Conception and design study, Data Curation
RLF Analysis and/or data interpretation, Conception and design study, Conceptualization, Data Curation, Final manuscript approval, Formal Analysis, Funding Acquisition, Project Administration, Realization of operations and/or trials, Resources, Supervision, Validation, Visualization, Writing - Original Draft Preparation
Corresponding author: André Campoli Frisina, Rua Bento de Andrade, 216, Jardim Paulista, São Paulo, SP, Brazil. Zip Code: 04503-000. E-mail: andrefrisina@yahoo.com.br
Article received: April 18, 2020.
Article accepted: April 18, 2020.
Conflicts of interest: none








 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter