

Review Article - Year 2021 - Volume 36 -
Fat grafting for the treatment of radiodermatitis after treatment of head and neck cancer
Lipoenxertia para tratamento de radiodermite após tratamento de câncer de cabeça e pescoço
ABSTRACT
Introduction: Although treatment of head and neck cancer has evolved in recent years, it is still a challenge to preserve and restore facial symmetry afterward; this fact significantly compromises the quality of life of patients. In reconstructive surgery, autologous fat grafting has been used to treat volume and contour defects. The objective is to review the effects of fat grafting in patients with radiodermatitis due to head and neck cancer radiotherapy treatment.
Methods: Literature review in PubMed and SciELO databases, looking for studies published in the last 20 years in Portuguese, English, or Spanish. The descriptors "adipose tissue", "transplantation", "neoplasms", "head and neck neoplasms" and "radiotherapy" and the non-descriptive terms "fat grafting", "fat transplantation" and "fat graft" were used.
Results: Of 212 articles found, only seven complied with the eligibility criteria.
Conclusion: The review suggests that fat grafting in head and neck cancer reconstruction is a safe treatment and optimizes aesthetic and functional results. Prospective studies are lacking for a better definition of techniques and results.
Keywords: Adipose tissue; Transplant; Neoplasms; Head and neck neoplasms; Reconstructive surgical procedures
RESUMO
Introdução: Embora o tratamento do câncer de cabeça e pescoço tenha evoluído nos últimos anos, ainda é um desafio preservar e restaurar a simetria facial após o mesmo, comprometendo significativamente a qualidade de vida dos pacientes. Em cirurgia reconstrutiva, o enxerto de gordura autóloga vem sendo usado para tratar defeitos de volume e de contorno. O objetivo é revisar os efeitos do enxerto de gordura em pacientes com radiodermite, devido ao tratamento radioterápico de câncer de cabeça e pescoço.
Métodos: Revisão de literatura nas bases PubMed e SciELO, buscando estudos publicados nos últimos 20 anos nas línguas portuguesa, inglesa ou espanhola. Foram utilizados os descritores "adipose tissue", "transplantation", "neoplasms", "head and neck neoplasms" e "radiotherapy"; e os termos não descritores "fat grafting", "fat transplantation" e "fat graft".
Resultados: De 212 artigos encontrados, apenas 7 estavam em conformidade aos critérios de elegibilidade.
Conclusão: A revisão sugere que a lipoenxertia em reconstrução oncológica de cabeça e pescoço é um tratamento seguro e permite otimizar os resultados estéticos e funcionais. Faltam estudos prospectivos para melhor definição de técnicas e de resultados.
Palavras-chave: Tecido adiposo; Transplante; Neoplasias; Neoplasias de cabeça e pescoço; Procedimentos cirúrgicos reconstrutivos
INTRODUCTION
Head and neck cancer care has evolved in recent years in diagnosis, prognosis, and treatment. Its therapeutic arsenal includes chemotherapy, radiotherapy, and surgery, whose significant advances are concentrated in reconstructive microsurgery1,2. Despite this, preserving and restoring facial symmetry after treatment is still a challenge, significantly compromising the quality of life of patients3-5.
In the last twenty years, there has been an increase in autologous fat grafts to treat volume and contour defects in reconstructive cosmetic surgery6-8. Its popularization can be attributed to the material’s wide availability, the low morbidity of the liposuction site in the donor area, and the intrinsic benefits as a minimally invasive procedure9-11.
Adipose tissue contains multipotent stromal cells, which secrete various trophic factors, such as transforming growth factor-beta (TGF-β). Among its actions, inflammatory and apoptotic suppression and the promotion of angiogenesis and parenchymal cell mitosis stand out12-15. The survival and longevity of fat grafts are variable after the procedure. Methods to increase their survival and that of their cellular constituents are still under investigation16-18.
Despite the advances, the medical literature lacks studies regarding complications, safety, and the results of aesthetic and functional improvement in cancer patients undergoing reconstructive surgery through the use of a fat graft for head and neck radiodermatitis.
OBJECTIVES
This study aims to review the medical literature regarding the side effects, benefits, and harms of the therapeutic use of fat grafting in radiodermatitis after treating head and neck cancer.
METHODS
It is an observational, analytical, and secondary study. The approval of the Federal University of São Paulo’s research ethics committee was issued under number 127310/2017. A literature review was carried out following the PRISMA statement. The descriptors “adipose tissue”, “transplantation”, “neoplasms”, “head and neck neoplasms” and “radiotherapy” were used; and the non-descriptive terms “fat grafting”, “fat transplantation” and “fat graft”. The health science descriptor system validated all descriptors. The PubMed and SciELO databases were accessed in April 2020 to search for studies published between 01/01/2001 and 01/01/2020 in Portuguese, English, or Spanish.
Chart 1 summarizes the attributes used to evaluate the studies found when searching the databases. Studies that met the following criteria were included: (a) randomized or quasi-randomized clinical trials, prospective/retrospective cohorts, and prospective/retrospective case series; (b) population undergoing surgical and radiotherapy treatment for head and neck cancer; (c) disease-free population for at least two years; (d) intervention with fat grafting technique on the face and neck comparable to another type of reconstruction or absence of reconstruction; (e) outcome with analysis of side effects, benefits, and harms of fat grafting. Studies that did not meet these criteria were excluded.
| Population | Patients treated for head and neck cancer, with radiodermatitis after and disease-free for at least two years |
| Intervention | Fat grafting intervention for radiodermatitis after head and neck cancer reconstruction |
| Comparison | Absence of radiodermatitis due to head and neck cancer |
| Outcome | Analysis of side effects, benefits and harms |
RESULTS
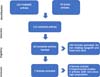
Altogether, 212 articles were found, 133 in the PubMed database and 79 articles in the SciELO database. Eighteen articles were selected for a thorough evaluation, three of which were excluded because they addressed the non-cancer population19-21, four reviews22-25, and four experimental studies26-29. Only seven studies met the inclusion criteria, being these case series4,30-35. No randomized clinical trials or cohort studies in humans were found. Figure 1 illustrates the inclusion and exclusion flows of the studies.
Phulpin et al.31, in 2009, submitted eleven patients with remission of head and neck cancer after treatment with external radiotherapy in the amount of 50 Gy or more to fat grafting. The group consisted of seven men, and the age ranged from 44 to 73 years (median = 54 years). The postoperative follow-up periods ranged from 2 to 88 months (mean = 39.9 months). Only two patients were followed for less than 24 months. All patients tolerated surgery and the immediate postoperative period. Transient ecchymosis, hyperemia, and edema occurred within the first 48 hours. Clinically, fat grafts provided structural improvement. However, the resorption of grafted fat, estimated at approximately 20 to 40%, was observed for all patients. Due to the defects’ importance, reinjections were performed in six patients at least three months after the first injection. In the areas of fat filling, there was an improvement in the quality of the irradiated skin. Clinically, the skin test showed greater malleability, the skin being softer and allowing for some functional improvement. Improvement in the quality of life of all patients has also been reported.
Rigotti et al.35, in 2007, also studied fat grafting in the reconstruction of irradiated tissues. The fat was collected using 2cc syringes and 2mm cannulas after tumescent infiltration. The aspirate was purified by centrifugation at 2700rpm, and the oil and liquid layers were discarded. Flow cytometry was used to isolate the stromal vascular fraction, cultured to obtain mesenchymal stem cells for further study. All stem cells were induced to differentiate into adipocytes, osteoblasts, or chondrocytes, depending on the culture medium. Ultrastructural analyzes were performed on the irradiated tissue before any grafting. It was found that it resembled tissues affected by chronic ischemia, characterized by damaged adipocytes, decreased capillary density, and abnormal capillary morphology. After transplantation of adipose stem cells, the structure of the tissue improved. Based on tissue analysis at different times in the post-treatment, it was hypothesized that adipose stem cells release angiogenic factors that lead to neoangiogenesis and, thus, improve tissue oxygenation. It has also been suggested that most mature adipocytes are irreversibly damaged in the harvesting process. The real effects of lipofilling, therefore, would come from the regenerative contributions of adipose stem cells.
Karmali et al.30, in 2015, conducted a retrospective analysis of 119 patients undergoing autologous fat grafting for head and neck cancer reconstruction. The primary endpoint was cancer recurrence. A total of 190 fat grafting procedures were performed on 116 eligible patients. The average time between radiotherapy and the first fat grafting was 40.5 months. The average time from cancer surgery for the first fat graft treatment was 35.1 ± 59 months. The average number of fat grafts per patient was 1.6 ± 1 (range 1 to 6), with an average injection volume of 24.8 ± 20.2mL. The follow-up time was 24 ± 22.3 months. Oncological recurrence was observed in five patients, two of whom were local, one regional, and two evolved with metastasis. All local and regional recurrences occurred in areas without continuity with the injected area. The overall complication rate was 4% and included two infections, two cysts at the injection site, and one overcorrection.
Coleman34, in 2006, performed fat grafting with the lower abdomen as a donor area. Fat grafting was performed in the cross direction with overlapping planes in the deep and superficial subcutaneous tissue. The mean volume of fat grafted was 24ml. The average procedure time was 1.8 hours with 21 hours of subsequent hospitalization. All patients underwent general anesthesia and were followed for one year. It was observed that injections of fat filled the soft tissue defects and improved the underlying skin. One case has been reported with radical excision of facial rhabdomyosarcoma and adjuvant radiotherapy. After lipofilling of the irradiated area, a sustained increase in facial volume was observed clinically. During its segment, no complications were observed. The result was assessed using the Aesthetic and Functional Evaluation System questionnaire, which evaluates the skin aesthetically by the dimension of the skin’s defect and flexibility and functionally by quantifying dysphonia dysphagia, changes in the head, and neck mobility, and changes in swallowing or chewing. There was an aesthetic improvement in 83% and functional improvement in 92% of the patients. A 25 to 50% fat graft reabsorption was observed through photographic recording in all patients.
Gutiérrez et al.4, in 2016, conducted a study to evaluate the duration of the procedure, anesthetic technique, length of hospital stay, complications, and the aesthetic and functional result of the treatment. Inclusion criteria were patients with a history of oral cancer and surgical and radiotherapy treatment with at least three years of complete remission; grade 3 or 4 in the Aesthetic and Functional Evaluation System, good health condition confirmed by preoperative and anesthetic evaluation; and consent to the consent form. Patients with previous fat grafting or those who did not have enough adipose tissue for the procedure were excluded. Twelve patients were included, ten of whom underwent surgical treatment and radiotherapy and two only for surgery.
Mojallal and Foyatier33, in 2004, published some cases involving grafting adipose tissue into irradiated tissue. They reported positive results, including better tissue growth; the improvement after the autologous fat graft was not limited to the subcutaneous.
In 2018, Karmali et al.32 carried out a new study with 116 patients (71 women). The average age was 55.9 years (ranging from 30 to 79 years), and the average body mass index was 26.7 ± 5.7 kg / m2. Eleven patients (9%) were active smokers. Eighty-one (69%) received radiation therapy before fat grafting, and two patients (1.7%) received radiation therapy after the graft. Seventy-six patients (66%) underwent some form of reconstruction with a free flap for a defect caused by cancer surgery, while 40 patients (34%) did not undergo a flap and only received autologous fat grafting for reconstruction. Thirteen patients (11%) underwent neck surgery for tumors with benign histology, while 103 (89%) had malignancy. Benign tumors were locally aggressive, indicating radical resection. The average time between the end of radiotherapy and the first treatment of fat grafting was 40.5 ± 24.3 months. The average time between head and neck cancer surgery and the first fat grafting was 35.1 ± 59.0 months. The average time between the first fat grafting treatment and the last follow-up was 35.8 ± 23.1 months.
In total, 190 fat grafting procedures were performed on 116 patients. The average number of treatments per patient was 1.6 ± 1.0 (range 1 to 6), with an average injection volume of 24.8 ± 20.2mL per session. All procedures were performed under general anesthesia. The main donor areas were the abdomen, hips, and flanks. Three local recurrences, one regional nodal recurrence, and two distant metastases were observed. Complications related to the procedure were observed in 5.1% of cases: two cases of infection, oil cysts, and fat necrosis. There was no morbidity at the donor site. All complications were resolved without further surgery or hospitalization
DISCUSSION
The effects of radiation on tissues can be classified as acute, consequential, or delayed. Acute effects are more noticeable in tissues with rapid cell turnover, such as epithelium or intestine. An increase in the pro-inflammatory cytokines interleukins 1 and 8 is observed, overexpressing TGF-ß, VEGF (vascular endothelial growth factor), TNF-α (tumor necrosis factor), and interferon-γ, and functional cell death. The mechanisms of the proliferation of stem cells damaged by radiation slowly replace lost cells. Acute effects can persist and are then classified as consequential36.
Consequent effects continue after the completion of radiotherapy and are more common in the urinary and intestinal tract, skin, and mucosa tissues. Late effects, in turn, develop months to years after treatment. Although not fully understood, the mechanisms integrate a defective cascade in healing: cytokines infiltrate irradiated tissues, fibrin leaks into the interstitium, collagen is deposited, and fibrosis occurs ischemia, atrophy, and associated circulatory damage37. The irradiated keratinocytes express low molecular keratins 5 and 14 instead of high molecular keratin cells 1 and 10, expressed in normal wounds. Radiation also disrupts matrix metalloproteins and tissue-inhibiting metalloproteins, causing deposition of collagen disorganized by fibroblasts38.
Flacco et al.29, in 2018, found that tissues chronically damaged by radiation presented epidermal thinning with homogenized eosinophilic sclerosis of the dermal collagen, large and atypical fibroblasts, and fibrous thickening with luminal obliteration of the deep vessels. Such changes are products of the expression of cytokines induced by radiation and reactive oxygen species, leading to cell apoptosis. Despite the obvious efficacy of radiotherapy to reduce local recurrence rates in head and neck cancers, collateral damage to the surrounding soft tissues can be deforming and debilitating.
Garza et al., In 201426, reported that the fat graft in the irradiated tissues decreased the dermal thickness and the collagen content while increasing the vascularization. In his study, human fat was obtained by liposuction and isolated by gravity. The liposuction was injected into a rat’s scalp irradiated with 16-gauge needles within 2 hours after collection. The skin was histologically inspected before and after the fat graft. The tissues irradiated before the fat graft showed dermal thickening and increased collagen with low vascular density, as measured by immunohistochemical staining of CD31. The retention of fat graft volume was significantly lower in irradiated tissues. Fat graft survival, assessed with computed tomography (CT), decreased significantly in irradiated tissues.
The experiences of Luan et al.27, in 2016, also reported an improvement in the microstructure and vascularization of the irradiated tissues after the fat graft. In their studies, irradiated mice were injected with human liposuction with stem cells derived from supplementary adipocytes. The volume retention of the fat graft was measured using 3D CT images. Fat grafts and overlapping skin were harvested eight weeks after injection and examined histologically for vascularity, dermal thickness, and collagen density. Significantly increased volume retention of fat grafts was found when they were enriched with adipose stem cells. Interestingly, the expected histological changes in radiation, such as increased dermal thickness, hypovascularity, and increased collagen density, were slightly attenuated with fat grafting. These effects were accentuated with the addition of stem cells. S
Similarly, Hadad et al.28, in 2010, found that when a combination of adipose stem cells and platelet-rich plasma was injected into irradiated tissues, microvascular density increased, and the wound healing time accelerated significantly.
The review by Haubner et al.24, in 2012, investigated the possible mechanisms by which adipose stem cells improve wound healing in irradiated tissue. It has been reported that stem cells derived from adipose tissue synthesize growth factors and cytokines, such as VEGF, platelet-derived growth factor (PDGF), and TGF-β, which have accelerated the healing of irradiated tissues in some cases.
Specifically, the skin, mucosa, and salivary glands are prone to acute radiation effects on the head and neck. Skin cells are depleted, causing erythema, desquamation, itching, hypersensitivity, and pain39. Damage to the salivary glands causes dry mouth, edema, and pain. Mucositis can develop with dysphagia and severe pain that disables food. Damage caused by radiation to the neck muscles aggravates dysphagia and causes trismus in severe cases. Other known complications include fibrosis and osteoradionecrosis40.
The use of fat grafting has been gaining ground in plastic surgery, with increasingly refined techniques and a better understanding of mesenchymal cells’ action, abundant in fat tissue6-8,21. Autologous fat transplantation is effective and inexpensive for the correction of facial deformities, preferable to synthetic implantation since it has more natural texture, contour, and facial expressions19,24,25,30-34.
Some alternative methods to treat fat grafting used are dermal filling agents, such as bovine collagen implants, injections and silicone prostheses, or hyaluronic acid. These, however, lead to a greater local inflammatory reaction and loss of skin texture. For bone correction, alternatives include bone autografts, acrylic prostheses, hydroxyapatite, and alloplastic implants. However, these materials can cause infection or extrusion19,30.
Autologous fat grafting may need new interventions since the entire structure designed in cosmetic surgery is lost over time due to gravity or tissue reabsorption19,35. Although lipofilling has been explored and used for more than a century to improve aesthetic concerns or deformities, fat injection in head and neck cancer reconstruction is relatively new. There are published reports of fibrosis regression after lipotransfer. Many studies are limited by small patient populations, fragmented evidence, and varying patterns of analysis.
However, motivated by these sporadic reports, Kumar et al.22, in 2016, carried out a systematic review to define the role and mechanism of lipotransfer in radiation-induced tissue fibrosis. They emphasized the complex interaction between fibroblasts, fibrogenic cytokines, and myofibroblasts, leading to various patterns of adhesion formation to cytoskeletal proteins with consequent scarring fibrosis. Some possible mechanisms were pointed out: activation of myofibroblasts by the cascade transformation of growth factor ß1 and connective tissue, fibrogenesis induced by chronic hypoxia, and radiation-stimulated release reactive oxygen species and free radicals with direct DNA damage.
The meta-analysis by Krastev et al.25, in 2018, regarding autologous fat injections in facial reconstruction revealed a general complication rate of 3.7%. The most-reported complications were asymmetries or irregularities after the injection. Other complications included infection (0.1%), fatty necrosis (1%) and hematoma (0.6%). Many plastic surgeons have avoided injecting fat into areas of previous cancer because of cancer concerns. However, based on current evidence, we believe that the cautious use of fat grafting is recommended to reconstruct head and neck cancer. More strong recommendations cannot be made until the publication of studies with a higher volume of cases and follow-up time. Prospective human studies are lacking for a better understanding of the procedure after head and neck cancer surgery. As the fifth most common cancer type and causing significant aesthetic and functional sequelae after surgical treatment, studies are necessary12,26.
No comparative analysis of results were found between fat grafting and other reconstructive cancer head and neck surgery techniques. Due to the small number of eligible articles, results are limited.
CONCLUSION
The sequelae of radiation from the head and neck can be challenging. Adipose tissue can be a valuable complement to improve these complications. Given the known benefits and the lack of obvious contraindications, we suggest that the fat graft can be considered carefully for reconstruction when faced with complications from the head and neck’s radiation.
REFERENCES
1. Kao SS, Ooi EH. Survival outcomes following salvage surgery for oropharyngeal squamous cell carcinoma: systematic review. J Laryngol Otol. 2018 Abr;132(4):299-313.
2. Graboyes EM, Zenga J, Nussenbaum B. Head & neck reconstruction: predictors of readmission. Oral Oncol. 2017 Nov;74:159-62.
3. Aksu AE, Uzun H, Bitik O, Tunçbilek G, Şafak T. Microvascular tissue transfers for midfacial and anterior cranial base reconstruction. J Craniofac Surg. 2017 Mai;28(3):659-63.
4. Gutiérrez SJ, Gridilla JM, Romero JP, López-de-Sagredo JG, Atín MSB. Fat grafting is a feasible technique for the sequelae of head and neck cancer treatment. J Craniomaxillofac Surg. 2016 Jan;45(1):93-8.
5. Leonetti JP, Nadimi S, Marzo SJ, Anderson D, Vandevender D. Facial reanimation according to the postresection defect during lateral skull base surgery. Ear Nose Throat J. 2016;95(12):E15-E20.
6. Laloze J, Varin A, Bertheuil N, Grolleau JL, Vaysse C, Chaput B. Cell-assisted lipotransfer: current concepts. Ann Chir Plast Esthet. 2017 Abr;62(6):609-16.
7. Hivernaud V, Lefourn B, Robard M, Guicheux J, Weiss P. Autologous fat grafting: a comparative study of four current commercial protocols. J Plast Reconstr Aesthet Surg. 2017 Fev;70(2):248-56.
8. Negenborn VL, Groen JW, Smit JM, Niessen FB, Mullender MG. The use of autologous fat grafting for treatment of scar tissue and scar-related conditions: a systematic review. 2016 Jan;137(1):31e-43e.
9. Khansa I, Harrison B, Janis JE. Evidence-based scar management: how to improve results with technique and technology. Plast Reconstr Surg. 2016 Set;138(3 Suppl):165S-78S.
10. De Decker M, De Schrijver L, Thiessen F, Tondu T, Van Goethem M, Tjalma WA. Breast cancer and fat grafting: efficacy, safety and complications-a systematic review. Eur J Obstet Gynecol Reprod Biol. 2016 Dez;207:100-8.
11. Kaoutzanis C, Xin M, Ballard TN, Welch KB, Momoh AO, Kozlow JH, et al. Autologous fat grafting after breast reconstruction in postmastectomy patients: complications, biopsy rates, and locoregional cancer recurrence rates. Ann Plast Surg. 2016 Mar;76(3):270-5.
12. Spiekman M, Przybyt E, Plantinga JA, Gibbs S, Van Der Lei B, Harmsen, MC. Adipose tissue-derived stromal cells inhibit TGF-ß1-induced differentiation of human dermal fibroblasts and keloid scar-derived fibroblasts in a paracrine fashion. Plast Reconstr Surg. 2014 Out;134(4):699-712.
13. Corselli M, Chen CW, Sun B, Yap S, Rubin JP, Péault B. The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. 2012 Mai;21(8):1299-308.
14. Mizuno H, Tobita M, Uysal AC. Concise review: adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells. 2012 Mai;30(5):804-10.
15. Philips BJ, Marra KG, Rubin JP. Adipose stem cell-based soft tissue regeneration. Expert Opin Biol Ther. 2012;12(2):155-63.
16. Tan SS, Ng ZY, Zhan W, Rozen W. Role of adipose-derived stem cells in fat grafting and reconstructive surgery. J Cutan Aesthet Surg. 2016 Jul/Set;9(3):152-6.
17. Zhou Y, Wang J, Li H, Liang X, Bae J, Huang X, et al. Efficacy and safety of cell-assisted lipotransfer: a systematic review and meta-analysis. Plast Reconstr Surg. 2016 Jan;137(1):44e-57e.
18. Ross RJ, Shayan R, Mutimer KL, Ashton MW. Autologous fat grafting: current state of the art and critical review. Ann Plast Surg. 2014 Set;73(3):352-7.
19. Alencar JCG, Andrade SHC, Pessoa SGP, Dias IS. Lipoenxertia autóloga no tratamento da atrofia hemifacial progressiva (síndrome de Parry-Romberg): relato de caso e revisão da literatura. An Bras Dermatol. 2011;86(4 Supl 1):S85-8.
20. Amarante MTJ. Análise da lipoenxertia estruturada na redefinição do contorno facial. Rev Bras Cir Plást. 2013;28(1):49-54.
21. Chia CY, Rovari DA. Lipoenxertia autóloga periorbitária no rejuvenescimento facial: análise retrospectiva da eficácia e da segurança em 31 casos. Rev Bras Cir Plást. 2012;27(3):405-10.
22. Kumar R, Griffin M, Adigbli G, Kalavrezos N, Butler PEM. Lipotransfer for radiation-induced skin fibrosis. Br J Surg. 2016 Jul;103(8):950-61.
23. Hammond SE, Samuels S, Thaller S. Filling in the details: a review of lipofilling of radiated tissues in the head and neck. J Craniofac Surg. 2019 Jun;30(3):667-71.
24. Haubner F, Ohmann E, Pohl F, Strutz J, Gassner H. Wound healing after radiation therapy: review of the literature. Radiat Oncol. 2012 Set;7:162.
25. Krastev TK, Beugels J, Hommes J, Piatkowski A, Mathijssen I, Van Der Hulst R. Efficacy and safety of autologous fat transfer in facial reconstructive surgery: a systematic review and meta-analysis. JAMA Facial Plast Surg. 2018 Set;20(5):351-60.
26. Garza RM, Paik KJ, Chung MT, Duscher D, Gurtner GC, Longaker MT, et al. Studies in fat grafting: part III. Fat grafting irradiated tissue: improved skin quality and decreased fat graft retention. Plast Reconstr Surg. 2014 Ago;134(2):249-57.
27. Luan A, Duscher D, Whittam AJ, Paik KJ, Zielins ER, Brett EA, et al. Cell-assisted lipotransfer improves volume retention in irradiated recipient sites and rescues radiation-induced skin changes. Stem Cells. 2016 Mar;34(3):668-73.
28. Hadad I, Johnstone BH, Brabham JG, Blanton MW, Rogers PI, Fellers C, et al. Development of a porcine delayed wound-healing model and its use in testing a novel cell-based therapy. Int J Radiat Oncol Biol Phys. 2010 Nov;78(3):888-96.
29. Flacco J, Chung N, Blackshear CP, Irizarry D, Momeni A, Lee GK, et al. Deferoxamine preconditioning of irradiated tissue improves perfusion and fat graft retention. Plast Reconstr Surg. 2018 Mar;141(3):655-65.
30. Karmali RJ, Nguyen AT, Skoracki RJ, Hanasono MM. Outcomes following autologous fat grafting in head and neck oncologic reconstruction. Plast Reconstr Surg. 2015 Set;136(4):49-50.
31. Phulpin B, Gangloff P, Tran N, Bravetti P, Merlin JL, Dolivet G. Rehabilitation of irradiated head and neck tissues by autologous fat transplantation. Plast Reconstr Surg. 2009 Apr;123(4):1187-97.
32. Karmali RJ, Hanson SE, Nguyen AT, Skoracki RJ, Hanasono MM. Outcomes following autologous fat grafting for oncologic head and neck reconstruction. Plast Reconstr Surg. 2018 Set;142(3):771-80.
33. Mojallal A, Foyatier JL. The effect of different factors on the survival of transplanted adipocytes. Ann Chir Plast Esthet. 2004 Out;49(5):426-36.
34. Coleman SR. Structural fat grafting: more than a permanent filler. Plast Reconstr Surg. 2006 Sep;118(3 Suppl):108S-20S.
35. Rigotti G, Marchi A, Galiè M, Baroni G, Benati D, Krampera M, et al. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: a healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg. 2007 Abr;119(5):1409-22.
36. Stone HB, Coleman CN, Anscher MS, McBride WH. Effects of radiation on normal tissue: consequences and mechanisms. Lancet Oncol. 2003 Set;4(9):529-36.
37. Goessler UR, Bugert P, Kassner S, Stern-Straeter J, Bran G, Sadick H, et al. In vitro analysis of radiation induced dermal wounds. Otolaryngol Head Neck Surg. 2010 Jun;142(6):845-50.
38. Medrado AP, Soares AP, Santos ET, Reis SRA, Andrade ZA. Influence of laser photobiomodulation upon connective tissue remodeling during wound healing. J Photochem Photobiol B. 2008 Set;92(3):144-52.
39. Cooper JS, Fu K, Marks J, Silverman S. Late effects of radiation therapy in the head and neck region. Int J Radiat Oncol Biol Phys. 1995 Mar;31(5):1141-64.
40. Beetz I, Schilstra C, Van Der Schaaf A, Van Der Heuvel ER, Doornaert P, Van Luijk P, et al. NTCP models for patient rated xerostomia and sticky saliva after treatment with intensity modulated radiotherapy for head and neck cancer: the role of dosimetric and clinical factors. Radiother Oncol. 2012 Out;105(1):101-6.
1. Federal University of São Paulo, Discipline
of Plastic Surgery, São Paulo, SP, Brazil.
RGFF Analysis and/or data interpretation, Final manuscript approval, Methodology, Writing - Original Draft Preparation, Writing - Review & Editing
RSA Analysis and/or data interpretation, Methodology, Writing - Original Draft Preparation, Writing - Review & Editing
ÉLFN Data Curation, Methodology
FCI Analysis and/or data interpretation, Writing - Review & Editing
PGD Analysis and/or data interpretation, Investigation, Writing - Original Draft Preparation
AGF Project Administration, Supervision, Writing - Review & Editing
Corresponding author: Roney Gonçalves Fechine-Feitosa, Rua Napoleão de Barros, 715, 4º andar, Vila Clementino, São Paulo, SP, Brazil. Zip Code: 04024-002. E-mail: roneyfechine@gmail.com
Article received: June 21, 2020.
Article accepted: January 10, 2021.
Conflicts of interest: none





 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter