

Original Article - Year 2021 - Volume 36 -
Viability of mesenchymal stem cells of adipose tissue from human liposuction
Viabilidade das células-tronco mesenquimais do tecido adiposo proveniente de lipoaspirado humano
ABSTRACT
Introduction: Lipografting is an alternative with important applicability for breast reconstruction and/or corrections of asymmetries resulting from cancer treatment. This technique consists of autologous fat transfer, whose stroma contains stem cells derived from adipose tissue that can differentiate itself throughout the mesodermal lineage. For adipose tissue preparation, Coleman-based centrifugation of syringe-aspirated material at 3000 revolutions per minute (rpm) for 3 minutes. However, studies question whether lower centrifugation speeds could be less harmful to cell viability.
Methods: An experimental study was conducted to evaluate the adipose cells of six patients; from 60mL of liposuction of each one. The sample collected was fractionated into four tubes and submitted to different protocols, decanting and centrifugation at speeds 500, 1000, and 3000rpm for 3 minutes. Afterward, the samples were processed with collagenase IA for 30 min, submitted to cell culture for 24 hours, and a cell viability analysis. The results were tabulated and analyzed by the ANOVA test using the Graphpad Prism 6.0® and SAS®.
Results: Cell viability was higher in the cell sample centrifuged at 3000rpm and lower in the decanted sample. Giemsa staining indicated maintenance of cell morphology on the samples.
Conclusion: Centrifuged cells at a speed of 3000rpm showed higher cell viability. Centrifugation was effective in compacting tissue and eliminating unwanted waste (blood and residual oil).
Keywords: Stem cells; Centrifuge; Lipectomy; Mammoplasty; Adipocytes.
RESUMO
Introdução: A lipoenxertia é uma alternativa com importante aplicabilidade para reconstrução de mama e/ou correções de assimetrias decorrentes do tratamento oncológico. Esta técnica consiste na transferência de gordura autóloga, cujo estroma contém células-tronco derivadas do tecido adiposo que tem capacidade de diferenciar-se em toda a linhagem mesodermal. Para o preparo do tecido adiposo, Coleman fundamentou a centrifugação, de material aspirado por seringa, em 3000 rotações por minuto (rpm) durante 3 minutos. Contudo, estudos questionam se velocidades menores de centrifugação poderiam ser menos deletérias para viabilidade celular.
Métodos: Foi realizado um estudo experimental, onde foram avaliadas as células adiposas de seis pacientes; a partir de 60mL de lipoaspirado de cada um. A amostra coletada foi fracionada em quatro tubos, e submetidos a diferentes protocolos, decantação e centrifugação nas velocidades 500, 1000 e 3000rpm por 3 minutos. Após as amostras foram processadas com colagenase IA por 30 min, submetidas ao cultivo celular por 24 horas e realizado a análise da viabilidade celular. Os resultados foram tabulados e analisados pelo teste ANOVA utilizando os programas Graphpad Prism 6.0® e SAS®.
Resultados: A viabilidade celular foi maior na amostra celular centrifugada a 3000rpm e menor na amostra decantada. A coloração com Giemsa indicou manutenção da morfologia celular entre as amostras.
Conclusão: As células centrifugadas na velocidade de 3000rpm apresentaram maior viabilidade celular. A centrifugação foi efetiva na compactação do tecido e eliminação de resíduos indesejados (sangue e óleo residual).
Palavras-chave: Células-tronco; Centrifugação; Lipectomia; Mamoplastia; Adipócitos
INTRODUCTION
Lipografting, a surgical technique known as lipomodeling or lipofilling, is an essential alternative to breast reconstruction techniques after surgical treatment of breast cancer1. This procedure consists of the transfer of autologous fat to the breast, aiming to restore the volume and revitalize and regenerate tissues damaged by adjuvant treatments such as radiotherapy; or more recently described, for minor volume adjustments conservative surgery for the treatment of breast cancer1,2. This regenerative potential is due to stem cells derived from adipose tissue (ATSC), present in subcutaneous cellular tissue1-4. A practical way to obtain a donor area of ATSCs, followed by preparation for injection in the receiving area, was described in the 1990s by Coleman3,4. In this technique, the liposuction is obtained using 3mm diameter needles coupled to 10mL syringes. After, the content is centrifuged at 3000rpm for three minutes1,3,4. In practice, there is significant variability of methods for obtaining liposuction and separating the elements that compose it5.
ATSCs come from the same embryological lineage as bone marrow stem cells. Because of the ease in obtaining surgery, they are considered an alternative source of stem cells5-8. ATSCs can differentiate beyond mesenchymal origin cells (adipocytes, myocytes, chondrocytes, and osteocytes) in non-mesenchymal cells, such as hepatocytes and neurons pancreatic cells, endothelial cells, and cardiomyocytes7,8.
The viability of ATSCs may be related to the form used for the separation of lipoaspirated elements4,5,7. Identifying management conditions that optimize the viability of the content used is essential to improve lipografting as an adjuvant in mammary reconstruction8,9. Despite being a widely used technique, there is still questioning about the effect on ATSCs and adipocytes’ viability after centrifugation; or after decanting of liposuction10,11.
OBJECTIVES
Therefore, the present study aimed to evaluate the cellular viability of isolated ASCs from decanting and centrifugation at different speeds (500, 1000, and 3000rpm) and collection methods: Coleman, assisted conventional liposuction (SAL), and associated with VASER (Vibration Amplification of Sound Energy at Resonance).
METHODS
An experimental clinical study was conducted with samples of liposuction of adipose tissue from patients older than 18 years, who agreed to participate in the research and signed the free and informed consent form, and who underwent liposuction procedures associated or not with lipofilling, performed in hospitals in Chapecó - Santa Catarina, between June and July 2018. The patients were randomly chosen during the proposed period for the collection of lipoaspirated material.
To verify the influence of liposuction processing by decanting or centrifugation, approximately 60mL of residual samples from each patient was evaluated, subdivided into 4 Falcon® tubes of 15mL, one subjected to decanting and each of the other three tubes subjected to centrifugation at speeds other than 500, 1000 and 3000rpm (equivalent to 43, 173 and 1560g, respectively) (centrifuge with rotor: 11133 Buzzer, Sigma®). For cell viability analysis, the ASCs were treated with 3-bromide (4,5-dimetiltiazol-2-il)-2,5-diphenyltetrazolium (MTT). In the control sample, 0.9% saline solution was added (SF 0.9%) in a 1:1 ratio and kept in decanting for 30 minutes12. The other samples were centrifuged at speeds of 500, 1000, and 3000 rpm, equivalent to 43, 173, and 1560g (considering that 2g is equivalent to 100 rpm in the rotor centrifuge: 11133 Buzzer, Sigma®); for 3 minutes. Then, the centrifuged samples were washed with 0.9% SF and centrifuged again for 10 minutes at 2000rpm. These four collagenase IA (C2674 - Sigma®) samples were added to a concentration of 0.075% at 1:1 ratio, and samples were incubated in a water bath at 37ºC for 30 minutes homogenization every 10 minutes12,13. For cell viability analysis, ATSCs were treated with 3-bromide (4,5-dimetiltiazol-2-il)-2,5-diphenyltetrazolium (TM).
From the 15mL Falcon® content, 200µL were aspirated from the pellet bottom (lower portion of the tube) and 200µL from the intermediate region and sent for cultivation in the 12-well plate, in triplicate, with the addition of 2mL of the DMEM medium (Vitrocell-Embriolife ®), containing 10% SFB, amphotericin and penicillin. The samples were grown in a greenhouse without CO2. Each sample’s aliquot was removed and colored with Giemsa dye to evaluate the cells’ morphology under optical microscopy. After 24 hours of cell culture, the cells were submitted to the cell viability assay by 3-bromide (4,5-dimetiltiazole-2-il)-2,5-diphenyltetrazozole (MTT). The plates were centrifuged at 2500rpm for 15 minutes in a thermo-shaker centrifuge (Agimaxx®.) The antinatant was discarded and 90 lµ0.9% at 37ºCand 10 µlof MTT reagent were added. He was in a dry bath at 37ºC for 60 minutes. It was centrifuged again at 2500rpm for 15 minutes and after discarding 70µL of each sample and adding 70µL dimethylsuf(DMSO). The centrifugal was incubated for 10 minutes, the centrifugal was repeated in the same conditions, and spectrophotometry of 560ηm12.14 was performed.
The data of cell culture of mesenchymal cells performed by the MTT technique were statistically analyzed using the MEANS and UNIVARIATE procedures and, by analysis of variance, using the statistical software SAS® (SAS INSTITUTE, 2002). With the plate reading, a nonparametric ANOVA study was performed by the NPAR1WAY procedure, and for comparison between the means, the nonparametric test of the SAS®/Graphpad Prism 6.0® software was adopted. The results of all analyses were considered significant with p<0.05. This project was approved by the Research Ethics Committee of Unochapecó, under Certificate of Presentation for Ethical Appreciation (CAAE) 73059017.4.0000.0116 and opinion number 2,576,847.
RESULTS
During the period determined for the collection of lipoaspirated, it was possible to evaluate 24 samples from six patients (five female and one male), with a median age of 43 years. The choice that the lipoaspirated were of different techniques was to verify whether there was the influence of the collection methodology (Coleman, conventional suction-assisted liposuction and VASER) according to the components’ separation of the lipoaspirated. The cell viability rate was considerably higher in the samples submitted to more intense centrifugations. The samples that went through centrifugation at 3000rpm showed better results regarding cell viability in ATSCs submitted to the MTT assay (0.309±0.08A). Centrifuged lipoaspirated with 500 and 1000rpm showed relatively close ATSC viability rates (0.246±0.12B and 0.253±0.08B, respectively). While the sample washed with SF 0.9% and decanted for 30 minutes, followed by exposure to MTT, presented the lowest viability rate of ATSCs (0.219±0.18AB) (Figure 1).
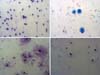
At the end of cell culture, samples stained with Giemsa dye and observed under optical microscopy showed similar morphologies; no karyorrhexis, a significant amount of binucleated cells, and no DNA evidence fragmentation in the different samples were observed (Figure 2).
DISCUSSION
Adipose cells’ viability may be related to several factors: characteristics of the patient’s adipose tissue; extraction procedure; centrifuge methodology; injection form of lipoaspirated; use of anesthetic solutions; topography of the donor area; among others10,11,15. There is no consensus on the most effective way to process fat (after aspiration of donor areas) for better graft viability; several factors can influence the cells’ viability that makes up liposuction5,16,17.
For this research, we chose to include samples from different liposuction techniques because the objective was to evaluate the impact of centrifugation speed on cell viability, regardless of the technique used in the collection. In a study conducted with lipoaspirated residues from three different techniques of obtaining adipose cells, it was verified that SAL is the one that provides the lowest amount of ATSCs18. On the other hand, in an in-vivo study, in which collection sites were treated with VASER, the authors concluded that similar results were found in ASLC considering the quality of adipose cell retention5. In another experimental study, which compared the technique of adipose tissue collection by SAL and the Coleman technique, it was verified that the technique of aspiration with a syringe would preserve the histological structure of adipocytes better than the ASC17.
The centrifugation process makes it possible to concentrate fat and provides a more significant number of cells per milliliter, separating from liquefied fat and blood cell components19. It was reported that the ATSCs present in the pellet (after washing the centrifuge) would have greater viability because there were no contaminants of the remains of the blood cells, which would be a constant in samples submitted only to decanting10. Centrifugation with lower processing speeds and time may have the same cell compaction capacity compared to speeds and times greater20,21. In our study, the sample submitted only to decanting, and those that were centrifuged at speeds of 500 rpm (43g) and 1000 rpm (173g) presented reduced cell viability values concerning the sample of 3000 rpm. The fact that the samples submitted to centrifugation at lower velocities (500 and 1000 rpm) showed similar results suggests that some factors such as cellular blood debris may have interfered in cell reproduction capacity. This was a limiter presented during the research; because, with the established methodology, it was not possible to identify the specific cause related to the lowest cell viability in samples that were not centrifuged at a speed of 3000 rpm.
Considering the variable cell morphology in an experimental study, no cellular changes were observed in optical microscopy examination at 3600 rpm centrifugation for 10 seconds9. There was no change in another experiment with lipoaspirated residues of eight patients, centrifuged at velocities (400g, 700g, 1200g, 3000g, or 4200g) microscopic morphology of cells16. While in another study, with similar methodology and centrifugation velocities of 1500 and 3000rpm for 3 minutes, no cell membrane changes were described, including the control sample (decanting)15.
In the present study, after 24-hour cultivation, it was possible to analyze the morphology of cell samples stained with Giemsa, and no morphological changes were observed between decanted and centrifuged samples at different speeds, but the cell viability evaluated by the MTT method was higher in the centrifuged sample at 3000rpm for 3 minutes. These findings suggest that both decanting and centrifugation velocities lower than 3000rpm may not interfere with cells’ microscopic aspect, but the higher speed would be related to maintaining cell multiplication capacity.
On the other hand, in experimental studies that used cell culture in rats, there was an association of deleterious effects in cell samples submitted to higher velocities when compared to those submitted to centrifugations with lower velocities9,16. This difference in results may be related to the form of cell culture methodology where most of the studies that associated better cell viability with higher centrifugation velocities, as in the present article, used cell culture in plates. In contrast, the other researchers used the murine model.
CONCLUSION
Fat grafting is widely applicable, especially in breast reconstruction, whose aesthetic and therapeutic effects are essential for the patient’s self-esteem, contributing to the quality of life during treatment. In searching for perfecting the technique, studies are carried out with the ASCs, who are mainly responsible for the reconstructive potential. In this context, it was possible to verify in this study that centrifugations with 3000 rpm for three minutes results in maintaining the biological characteristics of the mesenchymal cells of adipose tissue, necessary for cell colonization in the graft recipient areas. This reinforces the Coleman technique’s applicability, which can be performed in surgical treatment services for breast cancer, as long as the surgeon is used to the methodology.
ACKNOWLEDGMENT
To all the components of the research group “Molecular Biology and Biotechnology in Health” of Universidade Comunitária da Região de Chapecó (Unochapecó) and to the research group “Biological and Clinical Studies in Human Pathologies” of Universidade Federal da Fronteira Sul (UFFS) Campus Chapecó/SC.
To the Hospital Regional do Oeste, Hospital da Criança Augusta Muller Bohner and Hospital Unimed Chapecó for consenting the research. To surgeons: Cassiano Furtado Beller, Fabio Portanova Barros, Gustavo Colonheze, Jorge Diego Valentini, Liana Ortiz Streets Winkelmann, Rafael de Almeida Tirapelle and Tainara Cassol.
REFERENCES
1. Fatah F, Lee M, Martin L, O'Donoghue JM, Sassoon EM, Weiler-Mithoff M. Lipomodelling guidelines for breast surgery: joint guidelines from the association of breast surgery, the British Association of Plastic, Reconstructive and Aesthetic Surgeons, and the British Association of Aesthetic Plastic Surgeons; 2012.
2. Biazus JV, Falcão CC, Parizotto AC, Stumpf CC, Cavalheiro JAC, Schuh F, et al. Immediate reconstruction with autologous fat transfer following breast-conserving surgery. Breast J. 2015 Jun;21(3):268-75.
3. Coleman SR. Long-term survival of fat transplants: controlled demonstrations. Aesthetic Plast Surg. 1995 Out;19(5):421-5.
4. Coleman SR. Structural fat grafting. Aesthet Surg J. 1998 Set;18(5):386-8.
5. Fisher C, Grahovac TL, Schafer ME, Shippert RD, Marra KG, Rubin JP. Comparison of harvest and processing techniques for fat grafting and adipose stem cell isolation. Plast Reconstr Surg. 2013 Ago;132(2):351-61.
6. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001 Abr;7(2):211-28.
7. Kumar V, Abbas AK, Fausto N. Adaptação, lesão e morte celular. In: Kumar V, Abbas AK, Fausto N, eds. Robbins & Cotran - Patologia: bases patológicas das doenças. 7ª ed. Rio de Janeiro: Elsevier; 2005. p. 4-47.
8. Moreno M, Silva AV. Caracterização da célula-tronco derivada do tecido adiposo subcutâneo. Rev Bras Mastologia. 2012;22(4):138-43.
9. Hoareau L, Bencharif K, Girard AC, Gence L, Delarue P, Hulard O, et al. Effect of centrifugation and washing on adipose graft viability: a new method to improve graft efficiency. J Plast Reconstr Aesthet Surg. 2013 Mai;66(5):712-9.
10. Condé-Green A, Amorim NFG, Pitanguy I. Influence of decantation, washing and centrifugation on adipocyte and mesenchymal stem cell content of aspirated adipose tissue: a comparative study. J Plast Reconstr Aesthet Surg. 2010 Ago;63(8):1375-81.
11. Gabriel A, Champaneria MC, Maxwell GP. Fat grafting and breast reconstruction: tips for ensuring predictability. Gland Surg. 2015 Jun;4(3):232-43.
12. Yokomizo VMF, Benemond TMH, Bressan FF, Sangalli JR, Pieiri NCG, Casals JB, e tal. Adipose tissue derived stem cells: isolation, in vitro culture and potential uses in dermatology. Surg Cosmet Dermatol. 2011;3(1):55-9.
13. Ferreira RJ. Avaliação de diferentes métodos de detecção de alterações celulares para triagem de células-tronco mesenquimais: estudo pré-clínico (tese). Curitiba (PR): Universidade Federal do Paraná (UFPR); 2014.
14. Fukui M, Yamabe N, Zhu BT. Resveratrol attenuates the anticancer efficacy of paclitaxel in human breast cancer cells in vitro and in vivo. Eur J Cancer. 2010 Jul;46(10):1882-91.
15. Kim IH, Yang JD, Lee DG, Chung HY, Cho BC. Evaluation of centrifugation technique and effect of epinephrine on fat cell viability in autologous fat injection. Aesthet Surg J. 2009 Jan/Fev;29(1):35-9.
16. Kurita M, Matsumoto D, Shigeura T, Sato K, Gonda K, Harii K, et al. Influences of centrifugation on cells and tissues in liposuction aspirates: optimized centrifugation for lipotransfer and cell isolation. Plast Reconstr Surg. 2008 Mar;121(3):1033-41.
17. Pu LLQ, Coleman SR, Cui X, Ferguson REH, Vasconez HC. Autologous fat grafts harvested and refined by the Coleman technique: a comparative study. Plast Reconstr Surg. 2008 Set;122(3):932-7.
18. Oedayrajsingh-Varma MJ, Van Ham SM, Knippenberg M, Helder MN, Klein-Nulend J, Schouten TE, et al. Adipose tissue-derived mesenchymal stem cell yield and growth characteristics are affected by the tissue-harvesting procedure. Cytotherapy. 2006;8(2):166-77.
19. Ladeira PRS, Isaac C, Nakamura YM, Tutihashi RMC, Paggiaro AO, Ferreira MC. Cultivo de células-tronco derivadas de tecido adiposo: uma análise crítica. Rev Med (São Paulo). 2012 Dez;91(4):246.
20. Khoobehi K. Lipoenxertia na mama. In: Autor XX, ed. Contorno corporal e lipoaspiração. Rio de Janeiro: Elsevier; 2014. p. 43-53.
21. Pulsfort AK, Wolter TP, Pallua N. The effect of centrifugal forces on viability of adipocytes in centrifuged lipoaspirates. Ann Plast Surg. 2011;66(3):292-5.
1. Federal University of Fronteira Sul, Chapecó,
SC, Brazil.
2. Community University of the Region of Chapecó,
Medicine Course, Chapecó, SC, Brazil.
MM Analysis and/or data interpretation, Conception and design study, Final manuscript approval, Supervision, Writing - Original Draft Preparation, Writing - Review & Editing
JCS Analysis and/or data interpretation, Conception and design study, Final manuscript approval, Methodology, Supervision, Writing - Original Draft Preparation, Writing - Review & Editing
CDG Analysis and/or data interpretation, Data Curation, Realization of operations and/ or trials
LDM Analysis and/or data interpretation, Data Curation, Realization of operations and/ or trials
BSRB Analysis and/or data interpretation, Formal Analysis, Investigation, Realization of operations and/or trials
GCK Data Curation, Realization of operations and/or trials
FM Analysis and/or data interpretation, Formal Analysis
MDB Conception and design study, Final manuscript approval, Methodology, Supervision, Writing - Review & Editing
Corresponding author: Marcelo Moreno, Área Rural, Área Rural de Chapecó, Rodovia SC 484, Km 02, Sala 210, Chapecó, SC, Brazil. Zip Code: 89815-899. E-mail: marcelo.moreno@uffs.edu.br
Article received: March 10, 2020.
Article accepted: January 10, 2021.
Conflicts of interest: none






 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter