

Special Article - Year 2019 - Volume 34 -
Facelift and Facial nerve injury: how to deal with?
Ritidoplastia e paralisia facial: como lidar?
ABSTRACT
Is largely known that the facial nerve virtually can be injured in every facial lift. Even though its importance, it is difficult to find in the literature articles related to this theme. Complete or incomplete facial paralysis after a facial procedures, may become a very uncomfortable situation between the patient and the surgeon, that is why we propose in this article a guide which intends to help avoid, identify and manage a facial nerve injury in the event of a face lift. Identifying Facial palsy before the surgery: Intending to make the pre-op facial assessment as simple as possible, we suggest a systematic approach examination. Avoiding facial nerve injury during face lifting: Several technical details are discussed along the text aiming to minimize the risk of nerve damage during the surgical procedure. What to in the event of a post-operative facial palsy: The authors propose a logical approach to the facial palsy, suggesting a planning for diagnosis, classification of the lesion and a rational treatment for the injury. Conclusion: This article presents a guide showing some safe technical options to avoid nerve damage during rythidoplasty, how to identify the lesion and treat it if necessary.
Keywords: Rhytidoplasty; Facial nerve; Facial paralysis; Physical therapy modalities; Reconstructive surgical procedures
RESUMO
Não é novidade para os cirurgiões plásticos, especialmente aos que se dedicam à cirurgia de face, que o nervo facial pode ser lesado virtualmente em qualquer cirurgia de ritidoplastia. Porém, apesar da importância contida nesse tema, não é tarefa simples encontrar na literatura artigos que abordem de forma objetiva a correlação entre lesão do nervo facial e cirurgia facial. Paralisias faciais, sejam elas completas ou não, podem se tornar situações potencialmente conflituosas na relação médicopaciente. Este artigo, portanto, visa propor de forma clara e sucinta, baseada na experiência dos autores, um guia de como evitar, identificar e tratar uma potencial lesão do nervo facial no contexto de uma cirurgia de face, em especial a ritidoplastia.
Palavras-chave: Ritidoplastia; Nervo facial; Paralisia facial; Modalidades de fisioterapia; Procedimentos cirúrgicos reconstrutivos
INTRODUCTION
It is well known that the facial nerve can be injured in virtually every face lift. Despite its importance, it is difficult to find literature on this topic. Most articles on rhytidoplasty and facial nerve injury are from the early 70’s to the late 80’s and focus almost exclusively on how to avoid facial nerve lesions through anatomic dissections. There are very few published studies on the management of nerve injury and they focus mostly on treatment of complete nerve transection which comprises 2.6% of operated cases. It is also surprising that on searching for the terms: “face lift/lifting + facial palsy (FP)” or “rhytidoplasty/rhytidectomy + facial palsy”, in the PubMed database, there are no answers that fulfill the search criteria1,2.
Currently there is no concise data regarding the incidence of partial or total facial nerve injury during cosmetic facial procedures, as it is probably under reported. A complete or an incomplete facial paralysis after a facial procedure, may lead to a very uncomfortable situation between the patient and the surgeon, which is why we recommend a guide in this article to help avoid, identify, and manage a facial nerve injury in the event of a face lift surgery.
DISCUSSION
Assessment - Facial palsy before surgery: Pre-operative Clinical Examination.
Despite our best efforts to avoid nerve injury during a face lift surgery, it may still occur. However, it is important to note that in a significant number of cases, patients may already have a certain degree of facial paralysis or pre-operative weakness that remains unnoticed in a routine consultation. It is quite difficult to identify the signs of a very mild palsy if the surgeon is not used to treat such cases on a regular basis. Even patients may not have noticed any degree of asymmetry until the surgeon points it out but will certainly refer to it in a post- op situation.
To make the pre-op facial assessment as simple as possible, we suggest a systematic approach to the facial examination. A structured history and clinical examination of the patient allows for accurate treatment planning and anticipation of problems that may be exacerbated by surgery3.
Evaluation of facial asymmetry and spontaneity of facial motion can be performed while eliciting a routine medical history. With regard to functional history, a “top down” approach is utilized in a systematic manner, beginning with the brow. An ophthalmic and nasal history is obtained, and the patient is questioned regarding oral continence and speech. The surgeon should always include a history of Bell’s palsy. Recurrence or new onset palsy, three to five weeks post-op in our experience is possible but rare and undesirable.
A physical examination is also performed from the brow downwards. The presence or absence of rhytids and brow ptosis is noted. An asymmetric brow ptosis is common and is seen clearly over a period of time in photos. The upper lid is also examined for dermatochalasis and lid retraction. The patient is asked to close the eyes and any lagophthalmos is measured. The lower lid position is inspected for an ectropion and a snap test is performed3.
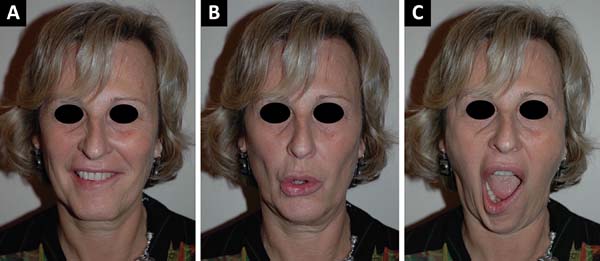
The nose is examined to exclude any fixed nasal obstruction and a Cottle’s test is performed to determine any nasal valve collapse4. Any midface ptosis or nasolabial crease asymmetry is evaluated. The mouth is examined at rest, with the amount of commissure droop and deviation of the philtrum to the contralateral side measured, if present. The excursion of the commissure is quantified and the degree of tooth show and shape of the smile are noted5-7. The lower lip is observed for any signs of weak depressor anguli oris function, indicating involvement of the mandibular division8. It is also very important to take sequential photos of the patient, at rest and in motion, before and after surgery, and note if there are any complications (Figura 1).
Avoiding facial nerve injury during a face lift
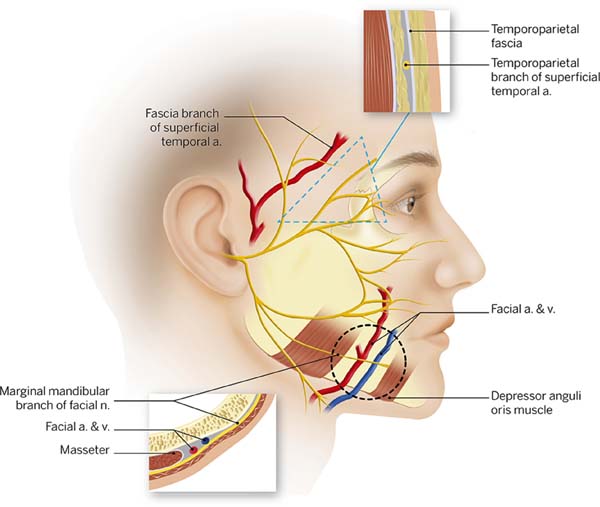
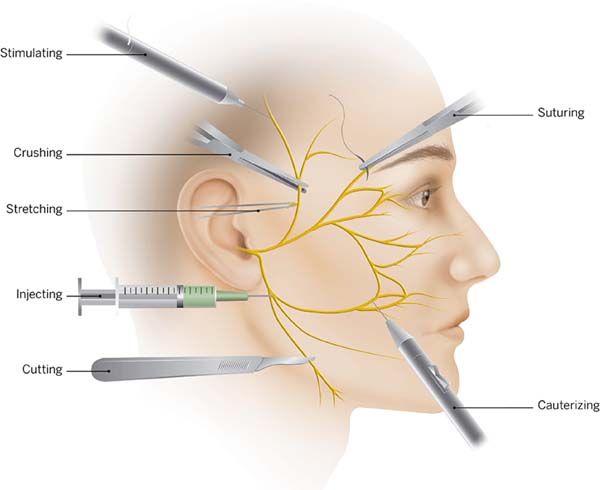
Several published articles have focused on this particular topic. It is clear that a deep knowledge of the facial anatomical structures and the anatomy of the facial nerve is very important. We recommend an article published in the late 70’s by Baker2 and a textbook by Brook Seckel titled: Facial Danger Zones (Figura 2)9, for surgeons who perform face lifts. Nevertheless, to master facial anatomy, we should always dissect in a very meticulous way, because the anatomy of high-risk areas is extremely variable and changes from patient to patient2,10.
Attention to minute details is as important as the dissection itself. Caution should be used while injecting solutions using fine needles or small cannulas. We are very permissive with the volume injected, using about 200cc of a 1:500.000 adrenaline solution. Intra-op anesthetic solutions are best avoided. Hydro dissection is recommended as it facilitates the undermining tissue and avoids unnecessary sharp dissections which could lead to less risk of inadvertent lesions and hematoma formation.
A blunt dissection is preferred especially for sub Superficial muscular aponeurotic system (SMAS) techniques. Trepsat dissectors (Pouret medical) are used routinely for both facial and neck approaches which makes a complete nerve transection virtually impossible (Figura 3).
Suturing and hemostasis techniques are also important. Avoid placing deep sutures in the SMAS, and preferentially place them along the axis of the major facial nerve branches. A surgeon must also prevent excessive traction and undue stretching2. During hemostasis, use of a bipolar cautery is advised and not large clamps or forceps, to minimize the electrocoagulation trauma (Figura 4). If there is a doubt regarding a nerve branch injury, a neurostimulator must be readily available at the operation room, to confirm any suspicion. It is important that the patient should not be curarized.
Nerve Section observed per-op
Occasionally the surgeon will be able to identify the damaged branch of the facial nerve. As the dissection goes towards the nasolabial fold, the nerves become less thick, which makes it difficult to correctly identify the anatomy with the naked eye. Several studies have been published describing surface anatomy landmarks correlating to the nerve divisions12-14 but the logical step is to test the assumed damaged branch and the surrounding ones with a neurostimulator, to avoid misidentification.
After this step, the nerve is sutured. Dissection of the proximal and distal ends of the nerve is done under magnification. A check is done to see if there is any loss of substance and an epineural suture is performed with non-absorbable 10 or 9.0 nylon. In case of traction of the nerve, nerve grafts are recommended, which are usually harvested from the great auricular nerve or the sural nerve15.
Post op paralysis: What should be done?
Diagnostic
In a majority of cases the surgeon will face the onset of paralysis during post-op. Whenever possible, evaluating the facial mimetic muscles at the end of the surgery is a good way to differentiate surgical trauma from other post-op causes such as Bell’s palsy. It is important to have a very strict follow-up and take pictures at every single contact with the patient.
To clinically assess the severity of peripheral facial nerve palsy various scoring systems are available. In our opinion, the most suitable facial nerve grading system is the Muscle testing of Freyss (Chart 1). This scale allows each muscle function to be evaluated separately, which is different from other grade systems, such as the House-Brackmann Grading System, which assesses groups of facial muscles. The system relies on evaluation of the degree of voluntary excursion of the facial muscles, evaluating ten muscles groups and attributing scores from 0 to 3 for each muscle, for a total score ranging from 0 to 3016. Evaluation is usually limited to two or three muscles, according to the injured nerve branch.
| Ten facial muscles | Score | Muscular contraction | |
|---|---|---|---|
| Frontalis | Wrinkles forehead and raises eyebrows | 0 | No contraction |
| Corrugator Supercilii | Pulls eyebrows medially and down | 1 | Minimal contraction |
| Orbicularis oculi | Closes eyelids | 2 | Wide excursion but weak contraction |
| Procerus | Pulls medial angle of eyebrow down producing wrinkles over bridge of nose | 3 | Normal contraction |
| Dilator naris muscle | Expands the nostrils | ||
| Orbicularis oris | Closes and protrudes lips | Total Score (0-30) | Grade of Facial palsy |
| Risorius | Pulls corner of mouth lateral | Score 20-30 | Slight |
| Zygomaticus major | Pulls corner of mouth up and lateral | Score 10-20 | Mild |
| Buccinator | Compresses cheek against teeth | Score 0-10 | Severe |
| Mentalis | Depresses lower lip and wrinkles chin skin | Score 0 | Total |
Facial nerve palsy can be categorized as complete if there is inability to voluntarily contract the facial muscles, or incomplete (partial). The degree of nerve damage can also be assessed by nerve conduction studies (electromiography-EMG) of the facial nerve. Reduction of the compound muscle action potential suggests axonal degeneration whereas increase in latency suggests demyelination of the nerve17.
Facial nerve palsy is an extremely frightening situation for the patient. The most frequently asked questions by these patients are whether their facial function will return to normal and how long it will take.
Among the wide variety of available prognostic tests, as discussed by Hughes18, the EMG seems to be the most reliable test to predict a patient’s prognosis19. To evaluate the lesion, an EMG is done early in the post-op, between day 4 and 6, although there is no rule ordering this first exam, because at this early stage, electromyography is used to detect any remaining voluntary activity. If voluntary potentials are detected, the palsy is labeled incomplete. The paralysis is considered complete only in the case of a totally silent electromyography19.
A diagnosis according to Sunderland and Seddon classifications (Chart 2) cannot be made at this stage, because pathologic spontaneous activity as a sign of neural degeneration does not occur earlier than 10 to 14 days after the onset of palsy. This early test is mainly intended to categorize the severity of an individual palsy and does not yield reliable prognostic information20,21.
| Sunderland | Seddon | Injury | Recovery potential |
|---|---|---|---|
| I | Neurapraxia I | Disruption of the nerve conduction, but nerve structure and axon remain intact. | Full (Up to 12 weeks) |
| II | Axonotmesis II | Disruption of the nerve conduction and axon degeneration, but remaining nerve structure remains intact. | Full (1mm/day) |
| III | Axonotmesis II | Disruption of the endoneurium, but the perineurium and epineurium remains intact. | Full (1mm/day) |
| IV | Axonotmesis II | Disruption of the endoneurium and perineurium, but epineurium remains intact. | Poor to none |
| V | Neurotmesis III | Total transection of the nerve fiber. | None |
A second examination should be performed not earlier than 10 to 14 days after the onset of palsy. After this period, all diagnostic criteria may have developed to establish a diagnosis according to Seddon, which predicts a prognosis on the expected clinical course19,20.
Steinner published that a fibrillation detected in EMG studies later than 10 to 14 days predicts that a patient has an 80% chance of an unfavorable result, but on the other hand, the absence of these signs implies an approximately 93% chance of total recovery20.
Treatment
Eye protection
One of the biggest problems with upper face facial palsy is the involvement of the eye if the lid commissure remains open. In this situation, eye care focuses on protection of the cornea from dehydration, drying or abrasions due to insufficient lid closure or tears. Eye ointments are recommended during day and night with protective glasses during the day22.
Physiotherapy
There are only a few controlled trials available on the effectiveness of physical therapy for facial palsies. In a randomized trial on 50 patients with Bell’s palsy and a House Brackmann scale grade of IV, mime therapy, speech therapy, including automassage, relaxation exercises, inhibition of synkinesis, coordination exercises, or emotional expression exercises, resulted in improvement of facial stiffness, lip motility, and the physical and social indices of the facial disability index23. A simple and reproducible technique has been used and advised by us: the mirror feedback therapy. It involves training the paralyzed side to reproduce symmetrical movements of the unaffected side in front of a mirror. Blanchin et al.24, in 2013, presented a paper proving that when the mirror therapy is applied to patients with long standing facial palsy and submitted to Labbe’s technique of facial reanimation it is more effective in recovering a spontaneous smile when compared to conventional therapies.
Corticosteroids
Till date to our knowledge, no study has discussed facial nerve trauma and the use of steroids for treatment. But since it is known that inflammation (particularly edema) of the facial nerve plays a key role in the pathogenesis of other types of facial paralysis, such as Bell’s palsy, we can extrapolate the concept to surgical trauma,. given the fact that a vast majority of cases are partial lesions.
Corticosteroids have been used due to its powerful anti-inflammatory effects in Bell’s palsy and this has recently been supported by a growing and well-designed evidence base. A Cochrane review included 1569 patients from 8 randomized controlled trials of adequate quality, and showed a benefit in improving the facial recovery, and a significant reduction in motor synkinesis in the steroid group25. Another high level systematic review published in the Journal of the American Medical Association (JAMA) concluded that corticosteroids used alone produced a reduced risk of unsatisfactory recovery26.
Even though the reviews support the use of steroids in facial palsy, there is no consensus on the prescription pattern. There are many authors who have suggested different protocols. We recommend the ones with the largest reviews:
- The Sullivan protocol27: Prednisolone 25 mg, by mouth (PO), twice daily, for 10 days, starting at a maximum of 72 h from the onset of palsy.
- The Engström protocol28: Prednisolone 60 mg PO for 5 days, then the dose is reduced by 10 mg per day for 5 days, also starting before 72 h of the onset.
- The Lagalla Protocol29: Prednisone 1 g, intravenous (IV), for 3 days, than 0.5 g IV for 3 days.
- The Stennert protocol (Table 1)30.
| Days of treatment | Cortisone (Prednisolone - equivalent dose - mg/day) | Dextran 40 with sorbite or mannite 5-10%c (ml) | Pentoxifylline (Trental) (ml) | ||||
|---|---|---|---|---|---|---|---|
| <70kg | >70kg | ||||||
| In-patient | 1 | Infusiona | 200 | 250 | 500 | 5 | |
| 2 | 200 | 250 | 500 | 10 | |||
| 3 | 150 | 500 | 15 | ||||
| 4 | 150 | 500 | 15 | ||||
| 5 | 100 | 500 | 15 | ||||
| 6 | 100 | 500 | 15 | ||||
| 7 | 75 | 500 | 15 | ||||
| 8 | 50 | 500 | 15 | ||||
| 9 | Oral circadianb | 40 | 500 | 15 | |||
| 10 | 20 | 500 | 15 | ||||
| Out-patient | 11 | (6-8 a.m.) | 15 | ||||
| 12 | 12.5 | ||||||
| 13 | 10 | ||||||
| 14 | 7.5 | ||||||
| 15 | 5 | ||||||
| 16 | 2.5 | ||||||
| 17 | 2.5 | ||||||
| 18 | 2.5 | ||||||
Stennert30 proposed a protocol based on an assumption that nerve damage is caused by edema and primary and secondary ischemia. To reduce the phlogistic and edematous reaction, he introduced steroids. Secondly he tried to increase the peripheral nerve perfusion, by adding pentoxifyline and dextrane to the IV infusion.
The effect of pentoxifylline on the recovery of Bell’s palsy has only been tested in association with other drugs, particularly steroids and low-molecular dextran. The studies showed a beneficial effect of a combination therapy, but it is not known which of these drugs is responsible for the beneficial effect16,28.
Botulinum toxin
When injected into facial muscles, botulinum toxin has been found to reduce the facial asymmetry in patients suffering from facial paralysis and has being used to treat synkinesis, hyperlacrimation, and hyperkinesis9. Most people have neglected the “physiotherapeutic” effect of the toxin. When applied to the healthy side, the toxin moderates movements, forcing the patient to exercise the affected side, which helps the muscle recover, and stimulates new neural connections. Therefore we recommend that it should be utilized even in the absence of hyperkinesia after confirmation of the diagnosis, around 12 days after the surgery (assumed facial palsy onset) and the EMG.
CONCLUSION
There is no consensus on management for a case of accidental nerve injury, therefore we have proposed a protocol with safe technical options to avoid nerve damage, identify, and treat if necessary. The proposed protocol is based on our own experience on treating facial palsy and published studies . Although it seems difficult to deal with such cases in general, patients with partial nerve lesions have an excellent prognosis with a recovery rate of 90 to 94%20, especially when the right decisions are made at the right time. In summary, we present a flowchart to help make clinical decisions (Figura 5).
COLLABORATIONS
|
FSR |
Analysis and/or data interpretation, conception and design study, data curation, final manuscript approval, methodology, project administration, supervision, visualization, writing - original draft preparation, writing - review & editing. |
|
CMR |
Data curation, supervision, visualization, writing - review & editing. |
|
FV |
Supervision, writing - review & editing. |
|
DL |
Analysis and/or data interpretation, conception and design study, data curation, final manuscript approval, methodology, project administration, supervision. |
REFERENCES
1. Castañares S. Facial nerve paralyses coincident with, or subsequent to, Rhytidectomy. Plast Reconstr Surg. 1974;54(6):637-43.
2. Baker DC, Conley J. Avoiding facial nerve injuries in rhytidectomy. Anatomical variations and pitfalls. Plast Reconstr Surg. 1979;64(6):781-95.
3. Fattah A, Borschel GH, Manktelow RT, Bezuhly M, Zuker RM. Facial Palsy and Reconstruction. Plast Reconstr Surg. 2012;129(2):340e-52e.
4. Howard BK, Rohrich RJ. Understanding the nasal airway: principles and practice. Plast Reconstr Surg. 2002;109(3):1128-46.
5. Manktelow RT, Zuker RM, Tomat LR. Facial paralysis measurement with a handheld ruler. Plast Reconstr Surg. 2008;121(2):435-42.
6. Paletz JL, Manktelow RT, Chaban R. The shape of a normal smile: implications for facial paralysis reconstruction. Plast Reconstr Surg. 1994;93(4):784-9.
7. Rubin LR. The anatomy of a smile: its importance in the treatment of facial paralysis. Plast Reconstr Surg. 1974;53(4):384-7.
8. de Maio M, Bento RF. Botulinum toxin in facial palsy: an effective treatment for contralateral hyperkinesis. Plast Reconstr Surg. 2007;120(4):917-27.
9. Seckel BR. Facial danger zones: avoiding nerve injury in facial plastic surgery. 1a ed. St Louis: Quality Medical Publishing; 1994.
10. Roostaeian J, Rohrich RJ, Stuzin JM. Anatomical considerations to prevent facial nerve injury. Plast Reconstr Surg. 2015;135(5):1318-27.
11. May M, Schaitkin BM. The facial nerve. New York: Thieme; 2000.
12. Chatellier A, Labbé D, Salamé E, Bénateau H. Skin reference point for the zygomatic branch of the facial nerve innervating the orbicularis oculi muscle (anatomical study). Surg Radiol Anat. 2013;35(3):259-62.
13. Stuzin JM, Wagstrom L, Kawamoto HK, Wolfe SA. Anatomy of the frontal branch of the facial nerve: the significance of the temporal fat pad. Plast Reconstr Surg. 1989;83(2):265-71.
14. Dorafshar AH, Borsuk DE, Bojovic B, Brown EN, Manktelow RT, Zuker RM, et al. Surface anatomy of the middle division of the facial nerve: Zuker’s point. Plast Reconstr Surg. 2013;131(2):253-7.
15. Sameem M, Wood TJ, Bain JR. A systematic review on the use of fibrin glue for peripheral nerve repair. Plast Reconstr Surg. 2011;127(6):2381-90.
16. Dubreuil C, Charachon R. Clinique: la paralysie faciale peripherique. In: Charachon R, Bebear JP, Sterkers O, Magnan J, Soudant J, eds. La paralysie faciale. Le spasme hemifacial. Paris: Société Française D’oto-Rhino-Laryngologie et de Pathologie Cervico-Faciale / L’européenne D’éditions; 1997. p. 135-57.
17. Finsterer J. Management of peripheral facial nerve palsy. Eur Arch Otorhinolaryngol. 2008;265(7):743-52.
18. Hughes GB. Prognostic tests in acute facial palsy. Am J Otol. 1989;10(4):304-11.
19. Grosheva M, Wittekindt C, Guntinas-Lichius O. Prognostic value of electroneurography and electromyography in facial palsy. Laryngoscope. 2008;118(3):394-7.
20. Sittel C, Stennert E. Prognostic value of electromyography in acute peripheral facial nerve palsy. Otol Neurotol. 2001;22(1):100-4.
21. Chhabra A, Ahlawat S, Belzberg A, Andreseik G. Peripheral nerve injury grading simplified on MR neurography: As referenced to Seddon and Sunderland classifications. Indian J Radiol Imaging. 2014;24(3):217-24.
22. Holland NJ, Weiner GM. Recent developments in Bell’s palsy. BMJ. 2004;329(7465):553-7.
23. Beurskens CH, Heymans PG. Positive effects of mime therapy on sequelae of facial paralysis: stiffness, lip mobility, and social and physical aspects of facial disability. Otol Neurotol. 2003;24(4):677-81.
24. Blanchin T, Martin F, Labbe D. Rééducation des paralysies faciales après myoplastie d’allongement du muscle temporal. Intérêt du protocole « effet-miroir ». Ann Chir Plast Esthet. 2013;58(6):632-7.
25. Salinas RA, Alvarez G, Daly F, Ferreira J. Corticosteroids for Bell’s palsy (idiopathic facial paralysis). Cochrane Database Syst Rev. 2010;(3):CD001942. DOI: 10.1002/14651858.CD001942.pub4
26. de Almeida JR, Al Khabori M, Guyatt GH, Witterick IJ, Lin VY, Nedzelski JM, et al. Combined corticosteroid and antiviral treatment for Bell palsy: a systematic review and meta-analysis. JAMA. 2009;302(9):985-93.
27. Sullivan FM, Swan IR, Donnan PT, Morrison JM, Smith BH, McKinstry B, et al. Early treatment with prednisolone or acyclovir in Bell’s palsy. N Engl J Med. 2007;357(16):1598-607.
28. Engström M, Berg T, Stjernquist-Desatnik A, Axelsson S, Pitkäranta A, Hultcrantz M, et al. Prednisolone and valaciclovir in Bell’s palsy: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2008;7(11):993-1000.
29. Lagalla G, Logullo F, Di Bella P, Provinciali L, Ceravolo MG. Influence of early high-dose steroid treatment on Bell’s palsy evolution. Neurol Sci. 2002;23(3):107-12.
30. Stennert E. New concepts in the treatment of Bell’s palsy. In: Malcolm D, House G, House WF, eds. Disorders of the facial nerve. New York: Raven Press; 1981. p. 313-8.
1. Clínica Cirurgia Plástica Beauté, Belém, PA,
Brazil.
2. Universidade Estadual de Botucatu, Botucatu,
SP, Brazil.
3. Clínica Particular, Caen, Normandia,
França.
Corresponding author: Franklin de Souza Rocha, Travessa Dom Romualdo de Seixas 1560, Belém, Brazil. Zip Code: 66055-028. E-mail: franklinrocha1@hotmail.com
Article received: March 02, 2018.
Article accepted: April 16, 2019.
Conflicts of interest: none.








 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter