

Original Article - Year 2019 - Volume 34 -
Nile tilapia skin xenograft versus silver-based hydrofiber dressing in the treatment of second-degree burns in adults
Xenoenxerto (pele da Tilápia-do-Nilo) e hidrofibra com prata no tratamento das queimaduras de II grau em adultos
ABSTRACT
Introduction: Recent studies have suggested the use of biological dressings made of aquatic animals as biomaterials in regenerative medicine since they demonstrate good adherence to the wound bed. The objective of this study was to evaluate the efficacy of Nile tilapia skin (Oreochromis niloticus) as an occlusive biological dressing in the management and treatment of second-degree burns in adults.
Methods: This clinical study included 30 patients randomly treated with Nile tilapia skin (n = 15) or Aquacel Ag® silver-based hydrofiber dressing (n = 15).
Results: The Nile tilapia skin yielded a similar mean treatment time (9.6 ± 2.4 days) to that of the comparative material (10.7 ± 4.5 days). There was no statistically significant intergroup difference (p > 0.68) in pain during dressing changes. No disadvantage in pain was noted, as 66.7% of patients treated with Nile Tilapia skin reported a decrease in pain events. Moreover, 60% of the patients treated with the Nile Tilapia skin did not require dressing replacement at any time during treatment. For the Aquacel AG® dressing, 53.3% of the patients required more than one dressing replacement.
Conclusions: Our findings suggest that the Nile tilapia skin is as effective as an occlusive biological dressing. The average treatment time (complete wound healing) and pain reports during dressing changes were similar between groups. Furthermore, pain after and number of dressing exchanges (when performed) were not worse.
Keywords: Burns; Occlusive dressings; Healing; Biological dressings; Cichlids
RESUMO
Introdução: Estudos recentes apontam a utilização do curativo biológico com base em animais aquáticos como biomaterial na medicina regenerativa, apresentando boa aderência ao leito das feridas. O objetivo foi avaliar a eficácia da utilização da pele da Tilápia-do-Nilo (Oreochromis niloticus) como curativo biológico oclusivo, no manejo/tratamento de queimaduras de 2º grau em adultos.
Métodos: Estudo clínico com 30 pacientes aleatoriamente tratados com pele da Tilápia-do-Nilo (n = 15) e hidrofibra com prata Aquacel Ag® (n =1 5).
Resultados: Em relação à duração, o tratamento com a pele da Tilápia-do-Nilo obteve uma média de dias de tratamento (9,6 ± 2,4) similar ao material comparativo (10,7 ± 4,5). Quanto ao relato de dor durante a troca de curativos, não houve diferença estatisticamente significante (p > 0,68) entre os grupos. Após a troca do curativo, não houve inferioridade no registro do valor na escala analógica de dor, em que 66,7% dos tratados com pele da Tilápia-do-Nilo relataram diminuição dos eventos álgicos. Constatou-se ainda que 60% dos pacientes tratados com a pele da Tilápia-do-Nilo não tiveram seus curativos substituídos em qualquer momento do tratamento. Para o curativo Aquacel AG®, 53,3% dos pacientes tiveram mais de uma substituição de curativos.
Conclusões: Com base na pesquisa, pode-se concluir que a pele da Tilápia-do-Nilo é eficaz como curativo biológico oclusivo. Houve similaridade entre os grupos para a média de dias de tratamento (completa cicatrização da ferida) e para o relato de dor durante a realização do curativo. Também, a não inferioridade relacionada a dor após os curativos e suas trocas (quando existentes) e na quantidade de substituições destes.
Palavras-chave: Queimaduras; Curativos oclusivos; Cicatrização; Curativos biológicos; Ciclídeos
INTRODUCTION
Approximately 1 million people in Brazil suffer burns every year, particularly second-degree superficial and/or deep burns1,2. The ideal dressing for such burns is easy to obtain, has good flexibility and adhesion to the wound bed, resists stretching, is easily handled, can suppress pain, if of low cost, is simple to store and, above all, prevents hydroelectrolytic losses and bacterial contamination, promotes epithelization, and encourages the adequate formation of granulation tissue in cases of grafting3.
Temporary skin substitutes and synthetic/biosynthetic dressing have been considered useful in the treatment of superficial burns because they reduce the frequency of dressing changes4. However, these materials are expensive and ineffective for deep burns5. Thus, alternative biological materials have been sought for this purpose. Tissues of animal origin, such as porcine skin and porcine intestinal submucosa, are among the materials used6. Recent studies have suggested the use of Nile tilapia skin (Oreochromis niloticus) as a biomaterial in regenerative medicine since it presented good adhesion to the wound bed in rats3 and satisfactory results of histological, histochemical, and tissue traction tests with human skin6.
Tilapia skin displays good tensile and compression resistance7, indicating that it may be usable as a biological dressing for burns. The presence of peptides with possible antimicrobial functions within this tissue reinforces this possibility 8-10.
OBJECTIVE
This study aimed to evaluate the efficacy of the use of Nile tilapia skin as an occlusive biological dressing compared to silver-based hydrofiber dressing (Aquacel AG®) in the management and treatment of superficial and deep second-degree burns in adults.
METHODS
This analytical interventional open clinical study with a convenience sample was performed at Hospital São Marcos, Recife/PE. The study was approved by the Research Ethics Committee of the Federal University of Pernambuco (no. 2.735.537). A clinical evaluation verified the general health conditions and the inclusion criteriA: Presence of superficial and/or deep second-degree burns affecting up to 10% of the burned body surface; maximum 72 hours since the burn occurred; age 20–60 years; and the absence of previous treatment for the current burns or significant comorbidities.
A total of 30 patients were selected. After receiving the initial explanation and providing written informed consent, they were randomly distributed into two groups: occlusive biological dressing with Nile tilapia skin (n = 15) or conventional treatment with the Aquacel AG® silver-based hydrofiber dressing (n = 15). The therapeutic process is described in Chart 1.
| Visit 1 (screening): | |
| • Collection of written informed consent | |
| • Clinical evaluation - physical examination, vital signs, anthropometric data; | |
| • Evaluation of the eligibility criteria (inclusion and exclusion criteria); | |
| • Allocation to the test or control group (according to randomization); | |
| • Photographs of the wound; | |
| • Preparation of the dressing; | |
| Guidelines on the procedures of the protocol and application of the visual analog scale (VAS) for pain | |
| Treatment visits: | |
| • Clinical evaluation; | |
| • Dressing evaluation - verification if dressing replacement is necessary in the test and control groups; | |
| • Photographs of the wound; | |
| • Application of the VAS | |
| Follow-up Visit - 7 (± 3) days after withdrawal of the dressing: | |
| • Clinical evaluation; | |
| • Photographs of the wound; | |
| • Study discharge | |
Nile tilapia skins are decontaminated (2% chlorhexidine and glycerol at high concentrations) and sterilized with gamma irradiation (Cobalt 60) to ensure the safety of their use in humans in addition to sampling microbiological testing for Gram-positive and -negative bacteria and fungi (Figure 1).
The procedures for both groups are described in Chart 2.
| Procedures | |
|---|---|
| First dressing | • Removal of blisters or loose skin |
| • Washing the lesion with running water and 2% chlorhexidine | |
| • Application of dressing | |
| • Test group: Occlusive biological dressing with Nile Tilapia skin (n = 15) | |
| • Control group: Conventional treatment with silver hydrogel (Aquacel AG®) (n = 15) | |
| • Coverage with cotton gauze, crepe bandages, and elastic tubular netting. | |
| Return | • Removal of the dressing and gauze layer |
| • Evaluation of the dressing for adherence to the wound bed | |
| • Replacement only when not adhered |
The outcomes for this study were:
Number of days required to achieve complete wound healing. The wound
was considered healed when 95% or more of the initial burn area was
re-epithelialized.
Pain assessment using a visual analog scale (VAS). ZERO corresponded
to no pain, while TEN indicated the worst pain felt during cleaning
and after application of the dressing. At each patient visit, the
dressing’s condition was evaluated and the pain score was
recorded.
Number of times a replacement Nile tilapia skin or Aquacel
AG® dressing was required.
The results were analyzed using descriptive statistics of absolute and relative frequencies and mean and standard deviation. The treatments were evaluated using Fisher’s exact test with a significance of p < 0.05 using SPSS version 20.0 software.
RESULTS
Of our cohort, 53.3% were treated with Nile tilapia skin, while 46.6% were treated with Aquacel AG®.
Table 1 shows that the mean treatment times in days were similar between the Nile tilapia skin and the Aquacel AG® (9.6 ± 2.4 and 10.7 ± 4.5 days, respectively).
| Categories | Treatment type | P value | ||
|---|---|---|---|---|
| Nile tilapia skin | Aquacel AG® | |||
| Number of days (discharge) | Minimum | 5 | 4 | 0.36 |
| Maximum | 14 | 19 | ||
| Average | 9.6 | 10.7 | ||
| Standard deviation | 2.4 | 4.5 | ||
Table 2 shows that the patients in both groups reported a VAS score greater than 5 during the exchange of dressings (p > 0.05; Fisher’s exact test).
| Treatment type | Total | P value | ||||
|---|---|---|---|---|---|---|
| Nile tilapia skin | Aquacel AG® | |||||
| Pain (during dressing exchange) | ≤ 5 points | n | 5 | 3 | 8 | 0.68 |
| % | 33.3% | 20.0% | 26.7% | |||
| > 5 points | n | 10 | 12 | 22 | ||
| % | 66.7% | 80.0% | 73.3% | |||
| Total | n | 15 | 15 | 30 | ||
| % | 100.0% | 100.0% | 100.0% | |||
After the dressing was changed, a new VAS pain score was collected. Table 3 shows that 86.7% of patients treated with Nile tilapia skin showed a reduced VAS score, and an analysis using Fisher’s exact test showed that it was not inferior to the Aquacel AG®.
| Treatment type | Total | P value | ||||
|---|---|---|---|---|---|---|
| Nile tilapia skin | Aquacel AG® | |||||
| Pain (after dressing application) | ≤ 5 points | n | 13 | 7 | 20 | ≤0.050 |
| % | 86.7% | 46.7% | 66.7% | |||
| > 5 points | n | 2 | 8 | 10 | ||
| % | 13.3% | 53.3% | 33.3% | |||
| Total | n | 15 | 15 | 30 | ||
| % | 100.0% | 100.0% | 100.0% | |||
Table 4 presents values regarding the number of skin substitutions or dressings required for complete re-epithelialization represented by patient discharge. Note that 60% of the patients who were treated with the Nile tilapia skin did not require skin replacement at any time during treatment, whereas 53.3% of patients treated with Aquacel AG® required more than one dressing replacement (p = 0.71), which indicates that the Nile tilapia skin was not inferior to the Aquacel AG®.
| Treatment type | Total | P value | ||||
|---|---|---|---|---|---|---|
| Nile tilapia skin | Aquacel AG® | |||||
| Number of exchanges | 0 | n | 9 | 7 | 16 | 0.71 |
| % | 60% | 46.7% | 53.33% | |||
| ≥ 1 | n | 6 | 8 | 14 | ||
| % | 40% | 53.3% | 46.67% | |||
| Total | n | 15 | 15 | 30 | ||
| % | 100% | 100.0% | 100% | |||
Figures 2 and 3 show the clinical results of two patients in the study from the first visit until medical discharge (complete re-epithelization).
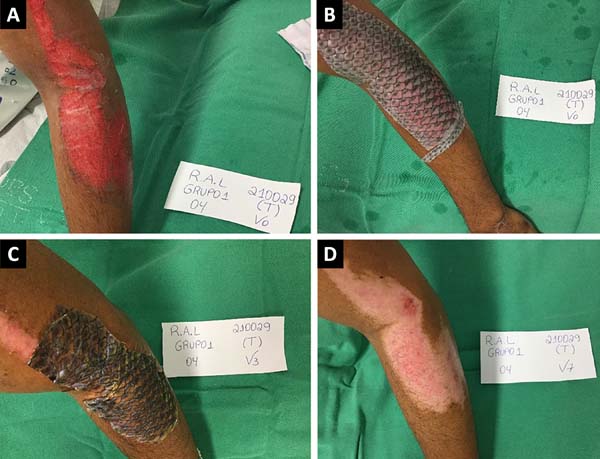
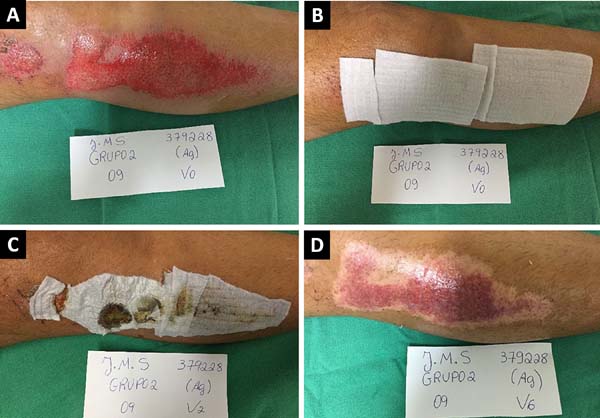
DISCUSSION
Studies have shown that hot liquids are the most common thermal agents that cause burn injuries1,2,11,12. In this study, 45% of the cases were due to overheated liquids.
The treatment and care of burns aim to provide a suitable environment for re-epithelialization and control the proliferation of microorganisms, which may delay the healing process13. Thus, biological dressings must display properties that prevent microbial growth, promote epithelization, and encourage the formation of granulation tissue6,14.
Records of the use of silver-based dressings date back to the 18th century15. Various properties of this material have been studied, including accelerating healing time, antimicrobial activity, and rapid re-epithelization. Despite its large-scale use, some disadvantages, including cytotoxicity, have inspired the study of other materials12,15,16.
Although we are far from an ideal temporary skin substitute, biological dressings have shown better functional and aesthetic results6,14. In this context, Nile tilapia skin is a promising product. Tilapia represents 45.4% of the total fish production in Brazil, but its skin is a waste byproduct of which only 1% is used in handicrafts. Tilapia skin must still be subjected to scientific analyses of its activity in humans. Several studies have compared human skin with Tilapia skin6,7,14,17-20, and favorable results were described regarding their histological and histochemical aspects and tensiometric properties18,20.
In this study, Nile tilapia skin was used in the treatment of 15 patients, 53.3% affected by second-degree superficial burns and 46.7% by second-degree deep burns.
To use animal skin as an occlusive dressing, a rigorous disinfection and sterilization protocol must be followed. Recent research indicates that chemical sterilization and radiosterilization are effective for the preparation of Nile tilapia skin18. The skins were provided by the Center for Research and Development of Medicines of the Federal University of Ceará, which is responsible for the sterilization processing.
Tilapia skin molds and adheres to the wound, creating a kind of tampon that prevents contamination and fluid loss.
The results of this study showed that the mean treatment time with Nile tilapia skin (9.6 ± 2.3 days) was similar to that with Aquacel AG® (10.7 ± 4.5 days).
Pain during and after the dressing change was assessed using a VAS. Patients in both groups reported a VAS score > 5 at the time of the initial cleaning and dressing application process. At the end of the dressing application, 86.7% of the patients in the Nile tilapia skin group reported feeling less pain, proven by VAS scores ≤ 5, compared to 46.7% of patients in the Aquacel AG® group (p = 0.05).
Skins and dressings are changed according to the amount of exudate. However, the higher the number of exchanges, the higher the risk of infection, the higher the cost of treatment, and the greater the possibility that the patient will feel pain. Given these aspects, it should be emphasized that fewer patients treated with Nile tilapia skin required dressing exchanges. In nine patients (60%) treated with Nile tilapia skin, there was no need for replacement of any dressing, while 53.3% of patients treated with Aquacel AG® required at least one exchange. Thus, considering the p value = 0.71 (p ≥ 0.05), skin healing with Nile tilapia skin was not inferior to that with Aquacel AG®.
The findings of this study suggest that Nile tilapia skin is as effective as Aquacel AG® in the management and treatment of second-degree burns in adults.
CONCLUSIONS
Based on the results of this study, Nile tilapia skin is an effective occlusive biological dressing in the management and treatment of second-degree burns in adults. The average treatment time of the patients treated with Nile tilapia skin (9.6 ± 2.4 days) was similar to that of patients treated with Aquacel AG® (10.7 ± 4.5 days). Furthermore, no significant intergroup difference was noted in pain level after dressing or the need for replacement during treatment.
COLLABORATIONS
|
MJBM |
Analysis and/or data interpretation, conceptualization, data curation, funding acquisition, investigation, methodology, realization of operations and/or trials, writing - original draft preparation, writing - review & editing. |
|
CTB |
Conceptualization, final manuscript approval, formal analysis, supervision, writing - review & editing. |
REFERENCES
1. Cruz BF, Cordovil PBL, Batista KNM. Perfil epidemiológico de pacientes que sofreram queimaduras no Brasil: revisão da literatura. Rev Bras Queimaduras. 2012;11(4):246-50.
2. Ministério da Saúde. Secretaria de Atenção à Saúde. Departamento de Atenção Especializada. Cartilha para Tratamento de Emergência das Queimaduras. Brasília: Ministério da Saúde; 2012. [acesso 2019 Jan 30]. [Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/cartilha_tratamento_emergencia_queimaduras.pdf
3. Ferreira E, Lucas R, Rossi L, Andrade D. Curativo do paciente queimado: uma revisão de literatura. Rev Esc Enferm USP. 2003;37(1):44-51. DOI: http://dx.doi.org/10.1590/S0080-62342003000100006
4. Hansbrough JF, Zapata-Sirvent RL, Cooper ML. Effects of topical antimicrobial agents on the human neutrophil respiratory burst. Arch Surg. 1991;126(5):603-8. PMID: 1850590 DOI: http://dx.doi.org/10.1001/archsurg.1991.01410290079016
5. Chanda J, Rao SB, Mohanty M, Muraleedharan CV, Arthur VL, Bhuvaneshwar GS, et al. Use of glutaraldehyde-gentamicin-treated bovine pericardium as a wound dressing. Biomaterials. 1994;15(1):68-70. DOI: http://dx.doi.org/10.1016/0142-9612(94)90200-3
6. Alves APNN, Lima Verde MEQ, Ferreira Júnior AE, Silva PGB, Feitosa VP, Lima Júnior EM, et al. Avaliação microscópica, estudo histoquímico e análise de propriedades tensiométricas da pele de tilápia do Nilo. Rev Bras Queimaduras. 2015;14(3):203-10.
7. Franco MLRS, Franco NP, Gasparino E, Dorado DM, Prado ME, Vesco APD. Comparação das peles de tilápia do nilo, pacu e tambaqui: Histologia, composição e resistência. Arch Zootec. 2013;62(237):21-32. DOI: http://dx.doi.org/10.4321/S0004-05922013000100003
8. Chem WY, Rogers AA, Lydon MJ. Characterization of biologic properties of wound fluid collected during early stages of wound healing. J Invest Dermatol. 1992;99(5):559-64. DOI: http://dx.doi.org/10.1111/1523-1747.ep12667378
9. Rajanbabu V, Chen JY. Applications of antimicrobial peptides from fish and perspectives for the future. Peptides. 2011;32(2):415-20. DOI: http://dx.doi.org/10.1016/j.peptides.2010.11.005
10. Hunag PH, Chen JY, Kuo CM. Three different hepcidins from tilapia, Oreochromis mossambicus: analysis of their expressions and biological functions. Mol Immunol. 2007;44(8):1922-34. DOI: http://dx.doi.org/10.1016/j.molimm.2006.09.031
11. Costa GOP, Silva JA, Santos AG. Perfil clínico e epidemiológico das queimaduras: evidências para o cuidado de enfermagem. Ciênc Saúde. 2015;8(3):146-55.
12. Farina JR JA. Novas tecnologias no tratamento de queimaduras. Rev Plastiko's. 2018:47-9.
13. Moser H, Pereima RR, Pereima MJL. Evolução dos curativos de prata no tratamento de queimaduras de espessura parcial. Rev Bras Queimaduras. 2013;12(2):60-7.
14. Miranda MJB. Viabilidade da pele da Tilápia-do-Nilo (Oreochromis niloticus). An Fac Med Olinda. 2018;1(1):49-52.
15. Hayneman A, Hoeksema H, Vandekerckhove D, Pirayesh A, Monstrey S. The role of silver sulphadiazine in the conservative treatment of partial thickness burn wounds: A systematic review. Burns. 2016;42(7):1377-86. DOI: http://dx.doi.org/10.1016/j.burns.2016.03.029
16. Tavares WS, Silva RS. Curativos utilizados no tratamento de queimaduras: uma revisão integrativa. Rev Bras Queimaduras. 2015;14(4):300-6.
17. Lima Junior EM, Bandeira TJPG, Miranda MJB, Ferreira GE, Parente EA, Piccolo NS, et al. Characterization of the microbiota of the skin and oral cavity of Oreochromis niloticus. J Health Biol Sci. 2016;4(3):193-7.
18. Alves APNN, Lima Júnior EM, Piccolo NS, de Miranda MJB, Lima Verde MEQ, Ferreira Júnior AEC, et al. Study of tensiometric properties, microbiological and collagen content in nile tilapia skin submitted to different sterilization methods. Cell Tissue Bank. 2018;19(3):373-82.
19. Lima Júnior EM. Tecnologias inovadoras: uso da pele da tilápia do Nilo no tratamento de queimaduras e feridas. Rev Bras Queimaduras. 2017;16(1):1-2.
20. Lima-Júnior EM, Picollo NS, Miranda MJB, Ribeiro WLC, Alves APNN, Ferreira GE, et al. Uso da pele de tilápia (Oreochromis niloticus), como curativo biológico oclusivo, no tratamento de queimaduras. Rev Bras Queimaduras. 2017;16(1):10-7.
1. Universidade Federal de Pernambuco, Recife, PE,
Brazil.
Corresponding author: Marcelo José Borges de Miranda Avenida Boa Viagem, nº 3296/102 - Boa Viagem, Recife, PE, Brazil Zip Code 51020-001 E-mail: mborgesmais@hotmail.com
Article received: October 30, 2018.
Article accepted: November 11, 2018.
Conflicts of interest: none.










 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter