

Original Article - Year 2018 - Volume 33 -
Prevention and treatment of ecchymosis, edema, and fibrosis in the pre-, trans-, and postoperative periods of plastic surgery
Prevenção e tratamento de equimose, edema e fibrose no pré, trans e pós-operatório de cirurgias plásticas
ABSTRACT
Introduction: Fibrosis, intense edema, and ecchymosis are
complications that represent a great challenge to dermatofunctional
physiotherapists in the postoperative period of liposuction and
abdominoplasty. Here we aimed to propose a new approach in the
pre-, trans-, and postoperative periods to prevent and minimize
fibrosis, intense edema, and ecchymosis, thereby expediting
the patient's recovery and reducing the number of required
physiotherapy sessions.
Method: This controlled clinical trial
included 20 female patients aged 18-56 years who were divided
into the control group (CG; n = 10) and experimental group
(EG; n = 10). All were scheduled to undergo abdominoplasty or
abdominal liposuction at least 7 days after enrollment. The two
groups were evaluated preoperatively. The CG received care
only from the 4th postoperative day, while the EG received care
during the pre-, trans-, and postoperative periods.
Results: The
EG required fewer mean physiotherapy sessions (p = 0.0032)
and had lower perimetry values on the 4th postoperative day
(p ≤ 0.05) in the inframammary groove and iliac crest, lower mean
resolution of fibrosis (p = 0.0058), and lower mean resolution of
ecchymosis (p = 0.0002) compared to the CG.
Conclusion: The
findings of this unprecedented study indicate that treatment
in the pre-, trans-, and postoperative periods reduces edema,
ecchymosis, and fibrosis formation in the postoperative period.
It also decreases the number of required physiotherapy sessions
and accelerates patient recovery from abdominal plastic surgery.
Keywords: Postoperative complications; Lipectomy; Physical therapy modalities; Fibrosis; Ecchymosis; Edema
RESUMO
Introdução: A fibrose, o edema intenso e a equimose são as intercorrências que
representam um grande desafio ao fisioterapeuta dermatofuncional no
pós-operatório de cirurgia plástica de lipoaspiração e abdominoplastia. O
objetivo é propor uma abordagem inédita desde o pré, trans e pós-operatório
para prevenir e minimizar as fibroses, edema intenso e equimoses, acelerando
a recuperação do paciente e reduzindo o número de sessões.
Método: Ensaio clínico controlado, composto por 20 pacientes do sexo feminino, com
idade entre 18 e 56 anos, divididos em dois grupos: 10 no grupo controle
(GC) e 10 no grupo experimental (GE), que apresentavam indicação cirúrgica
de abdominoplastia ou lipoaspiração abdominal, associadas ou não, e que se
encontravam com no mínimo 7 dias de pré-operatório. Os dois grupos foram
avaliados no pré-operatório. O GC recebeu atendimento somente a partir do 4º
dia de pós-operatório, enquanto que o grupo GE recebeu atendimento durante o
pré, trans e pós-operatório.
Resultados: O grupo experimental apresentou uma média menor do número de sessões
(p = 0,0032), perimetria menor no 4º dia de
pós-operatório (p < 0,05) nos pontos antômicos: sulco
inframamário e crista ilíaca, média menor na resolução da fibrose
(p = 0,0058) e média menor na resolução da equimose
(p = 0,0002) em relação ao grupo controle.
Conclusão: Pode-se concluir com este estudo inédito que o tratamento no pré, trans e
pós-operatório reduz o edema, a formação de equimose e principalmente a
formação de fibrose no pós-operatório. Também diminui o número de sessões
fisioterapêuticas e acelera o restabelecimento do paciente no pós-operatório
das cirurgias abdominais.
Palavras-chave: Complicações pós-operatórias; Lipectomia; Modalidades de fisioterapia; Fibrose; Equimose; Edema
INTRODUCTION
According to the International Society of Aesthetic Plastic Surgery, Brazil ranks second globally for aesthetic surgical procedures. The implantation of a silicone breast enlargement prosthesis remains the most popular surgical procedure worldwide (15.8%), followed by liposuction (14%), blepharoplasty (12.9%), rhinoplasty (7.6%), and abdominoplasty (7.4%)1.
Of these surgical procedures, abdominoplasty and liposuction entail major intercurrences and complications2-4.
Fibrosis, intense edema, and ecchymosis are complications that greatly challenge the dermatofunctional physiotherapist, who, over the past few years, has sought effective treatments to act in the pre-, trans-, and postoperative periods, demonstrating the importance of this healthcare professional5.
The physiotherapist’s role during the preoperative period remains very restricted6. Lange7 reports the use of cosmetics, nutricosmetics, and low glycemic index diets to improve the healing process, rearrange collagen, and reduce the rates of fibrosis, intense edema, and ecchymosis formation.
The physiotherapeutic approach in the transoperative period of plastic surgery has not been well demonstrated. Lange7 mentioned only the use of Unna boots and compression stockings for the prevention of deep venous thrombosis.
Thus, the present study proposes a new approach to the preoperative (use of antiglycans, nutricosmetics and nutritional orientations), transoperative (lymphatic taping and containment foam), and postoperative (manual lymphatic drainage, microcurrent, red light-emitting diode [LED], and taping) management with the goal of preventing and minimizing fibrosis, severe edema, and ecchymosis, accelerating patient recovery, and reducing the number of required physiotherapy sessions.
OBJECTIVE
The objective of this study is to evaluate the occurrence of postoperative ecchymosis, edema, and fibrosis in patients undergoing liposuction and/or abdominoplasty and statistically correlate these occurrences with the pre- and transoperative treatment.
METHOD
This controlled clinical trial was performed from June to December 2016. The study received approval from the Ethics Committee of the Center of Higher Education of Campos Gerais, Ponta Grossa, PR (55410316.0.0000.5215).
Female subjects in the preoperative phase of abdominal plastic surgery who were aged 18-56 years, had the surgical indication of abdominoplasty and/or abdominal liposuction, and were scheduled at least 7 days before surgery were admitted to the study. The sample consisted of 20 patients divided evenly into the control group (CG) and the experimental group (EG).
Patients in the CG and EG were evaluated preoperatively and daily after the 4th postoperative day until the end of treatment.
The 20 patients were evaluated preoperatively, and personal details, surgical details, abdominal semiology, anthropometric measurements, and photodocumentation were collected.
The postoperative treatment in the CG was started from the 4th day for 1 hour each session for the first 3 sequential sessions, and the remaining alternated 3 times in the first week, 3 times in the second week, twice in the third week and once in the last 4 weeks for a total of 15 sessions.
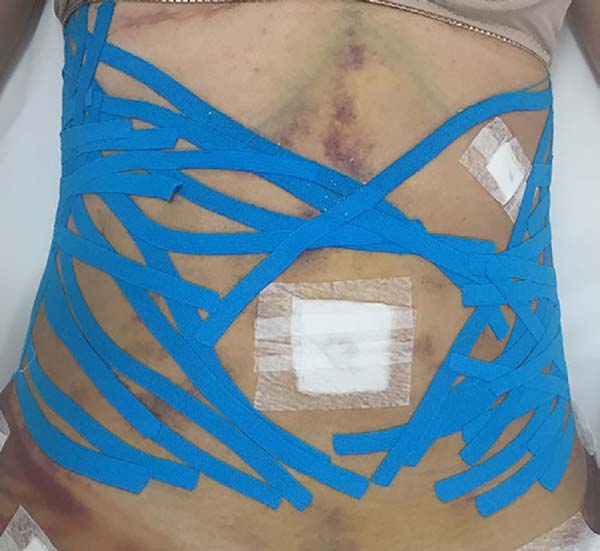
The therapeutic resources used in all sessions were: manual lymphatic drainage with the Leduc method in the lower limbs, upper limbs, abdomen and flanks, microcurrent (frequency, 250 Hz; intensity, 150 µA) for 20 minutes in the abdomen, red LED (650-959 nm) for 20 minutes in the abdomen, and application of taping in the operated area with the application chosen in accordance with the alteration found (edema, ecchymosis, or fibrosis), with a “web” or “basket” cut for fibrosis, “fan” or “octopus” for edema, and “hashtag” cut for ecchymosis, with a rest of 3-5 days between them and 1 day to the next application when necessary.
The EG group received care during the pre-, trans-, and postoperative periods. Preoperatively, guidelines regarding postoperative care, nutritional guidance with low glucose ingestion, a nutricosmetic oral antiglycant (Exsynutriment®, Glycoxil®, and Bioarct®, 100 mg each, 30 capsules, 1 capsule per day), and a topical antiglycant (Alistin® 10%, 30 g, 2 times a day in the area subjected to surgery), were provided and indicated to be used for 30 days or until the product supply was exhausted.
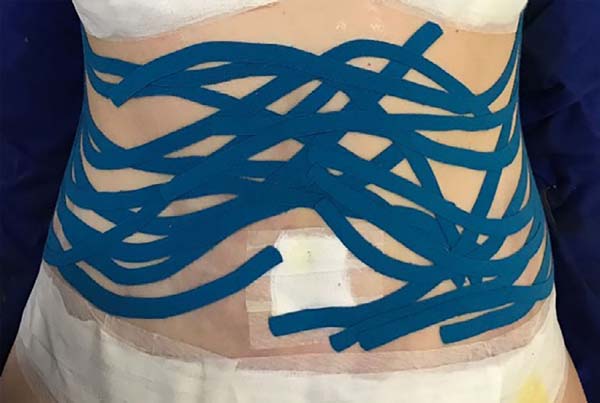
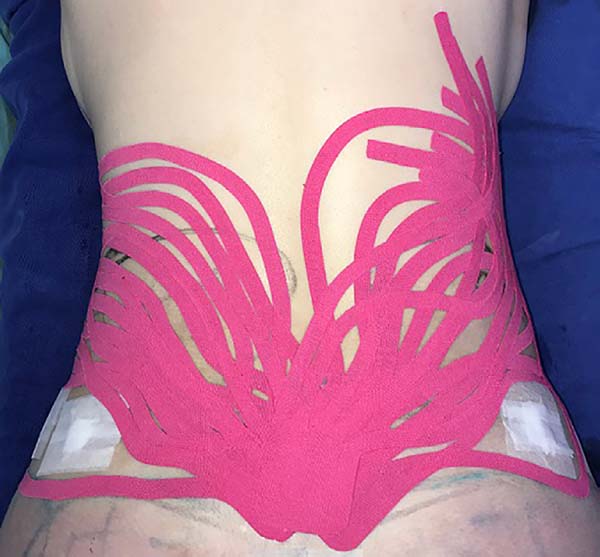
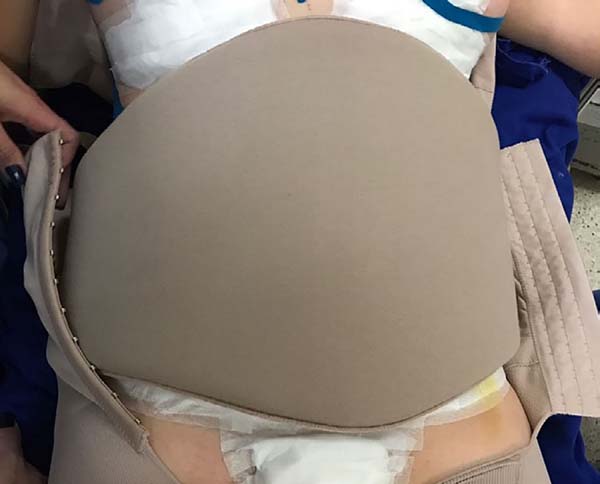
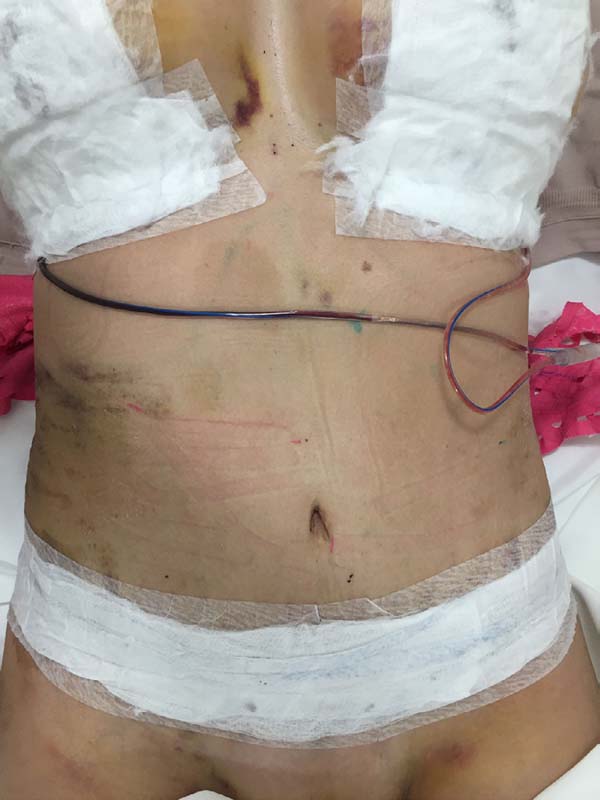
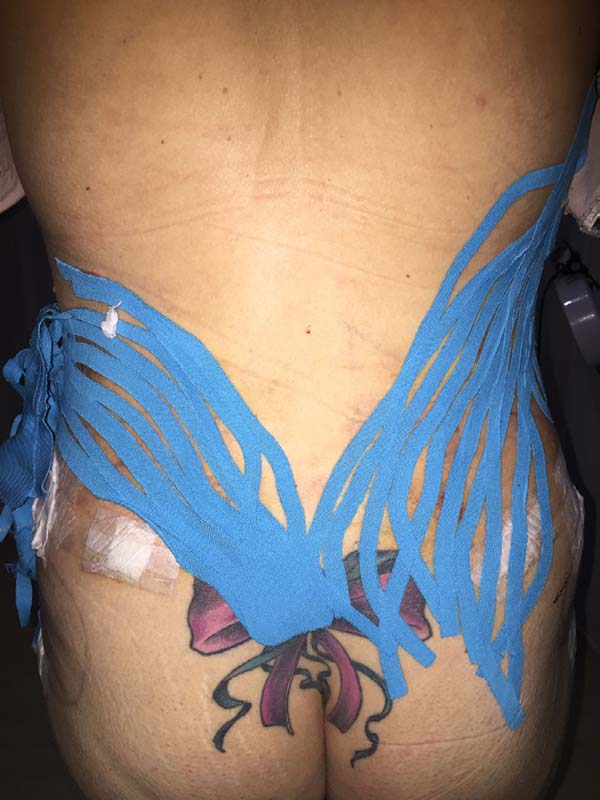
The intraoperative treatment of the EG group was performed with the application of lymphatic taping in a “fan” or “octopus” form in the operated regions. The dressings were cut into five different portions, positioned with minimum tension (0-15%) in the regions of the abdomen (with a fixed base in the axillary region bilaterally) (Figure 1) and flanks (with a fixed base in the coccygeal region) (Figure 2) and contention foam 360° in the operated region under the surgical mesh (Figure 3). The same postoperative treatment was offered to patients in the EG and CG.
Fibrosis formation was evaluated during all care sessions through palpation, visual inspection, contact thermography analysis, and photodocumentation in both groups. Edema was analyzed based on perimetry and body weight in both groups, while ecchymosis was evaluated using photodocumentation in both groups.
A database was created of the collected data that were subjected to analysis. Descriptive statistics and analysis of variance were the statistical methods used.
The following data were statistically analyzed in both groups: number of sessions; start of fibrosis; resolution of fibrosis; resolution of ecchymosis; perimetry of the iliac crest, inframammary groove, and navel; initial thermography; degree of fibrosis; ecchymosis type; and pain.
RESULTS
With the data collected during the preoperative and postoperative care of patients in the CG and EG, the study variables were assessed and compared.
The data of the quantitative variables were subjected to the Kolmogorov-Smirnov (KS) normality test to guide the choice between parametric or non-parametric tests.
The standard deviation of the variables by groups approved in the normality tests were subjected to the T test to guide the choice of the most suitable test for parametric analysis of variance of the means test (T test or T test with Welch correction). The data sets not approved in the KS test were subjected to the Mann-Whitney non-parametric test.
The p values of the analysis of variance and summary statistics of the numerical variables are presented in Table 1.
| Variable | Group | P value | |||||
|---|---|---|---|---|---|---|---|
| Control | Experimental | ||||||
| Age (years) | µ | 32.1 | 39.9 | 0.1251 | |||
| ± | 10.2 | 11.5 | |||||
| No. of sessions | µ | 23.1 | 14.6 | *0.0032 | |||
| ± | 6.7 | 0.7 | |||||
| Beginning of fibrosis | µ | 19 | 3.4 | *0.0019 | |||
| ± | 4.7 | 7.2 | |||||
| Resolution of fibrosis | µ | 48.6 | 11.7 | *0.0058 | |||
| ± | 22.2 | 16.3 | |||||
| Rest time | µ | 15 | 14.5 | 0.4704 | |||
| ± | 0 | 1.6 | |||||
| Resolution ecchymosis | µ | 17.6 | 7.8 | *0.0002 | |||
| ± | 5 | 4.3 | |||||
| Weight (kg) | PO | µ | 70.97 | 69.49 | 0.7071 | ||
| ± | 7.76 | 9.49 | |||||
| 4th PO | µ | 70.5 | 69.75 | 0.8177 | |||
| ± | 7.06 | 9.92 | |||||
| Final | µ | 66.51 | 65.66 | 0.8177 | |||
| ± | 7.58 | 8.63 | |||||
| Variation (from the PO) | 4th PO | µ | 20.92 | 23.6 | 0.3073 | ||
| ± | 12.52 | 16.36 | |||||
| Final | µ | -20.92 | -23.6 | 0.6852 | |||
| ± | 12.52 | 16.36 | |||||
| Perimetry (cm) | Pré-operatória | Inframammary groove | µ | 85.5 | 84 | 0.5601 | |
| ± | 5.8 | 5.5 | |||||
| Navel | µ | 92.9 | 91.9 | 0.78 | |||
| ± | 8 | 7.8 | |||||
| Iliac crest | µ | 97.3 | 97.5 | 0.9379 | |||
| ± | 5.2 | 6.1 | |||||
| 4th PO | Inframammary groove | µ | 88.1 | 81.7 | *0.0261 | ||
| ± | 5.4 | 6.4 | |||||
| Navel | µ | 95.1 | 88.6 | 0.0567 | |||
| ± | 8 | 6.2 | |||||
| Iliac crest | µ | 99.8 | 94.7 | *0.0303 | |||
| ± | 5.2 | 4.5 | |||||
| Final | Inframammary groove | µ | 81 | 77.3 | 0.1518 | ||
| ± | 5.4 | 5.7 | |||||
| Navel | µ | 86 | 81 | 0.133 | |||
| ± | 8.2 | 5.7 | |||||
| Iliac crest | µ | 89.3 | 88.5 | 0.7447 | |||
| ± | 5.7 | 5.1 | |||||
| Variation (from the PO) | 4th PO | Inframammary groove | µ | 3.09 | 2.78 | *0.0001 | |
| ± | 1.15 | 2.29 | |||||
| Navel | µ | 2.38 | 3.42 | *0.0022 | |||
| ± | 0.49 | 4.3 | |||||
| Iliac crest | µ | 2.58 | -2.76 | *0.0002 | |||
| ± | 1.08 | 2.84 | |||||
| Final | Inframammary groove | µ | -5.09 | -8.25 | *0.0027 | ||
| ± | 1.75 | 2.28 | |||||
| Navel | µ | -7.31 | -12.26 | *0.0133 | |||
| ± | 1.48 | 5 | |||||
| Iliac crest | µ | -8.04 | -9.48 | 0.1821 | |||
| ± | 1.71 | 2.8 | |||||
* Significant differences between the means (p < 0.05); PO: Postoperative day.
The variables that presented significant differences between the means (p < 0.05) included number of sessions, beginning of fibrosis, resolution of the fibrosis, resolution of the ecchymosis, perimetry on the 4th postoperative day of the iliac crest and the inframammary groove, and perimetry of the 4th postoperative day and final assessment of the inframammary groove and navel.
From the T test with Welch correction, significant (p < 0.05) differences were found between the EG and the CG with regard to the number of sessions; therefore, we rejected the null hypothesis.
The EG (μ =14.60 ± 0.70) presented a statistically significant lower mean number of sessions (p = 0.0032) than the CG (μ = 23.10 ± 6.71).
No pain was reported in the operated region in the EG, while the CG reported pain a mean 5.50 ± 1.58 postoperative days.
Perimetry was performed preoperatively, on the 4th postoperative day, and at the end of the treatment in the inframammary groove, navel, and iliac crest.
On the 4th postoperative day, significant differences (p < 0.05) were observed in the inframammary groove and iliac crest. The EG displayed a larger mean reduction in the perimetry than the CG in all different periods and anatomic parts except the iliac crest.
Significant differences (p < 0.05) were found between the EG and CG in relation to the resolution of fibrosis (Table 2); thus, we rejected the null hypothesis.
| Variable | Group | P value | ||
|---|---|---|---|---|
| Control | Experimental | |||
| Resolution of fibrosis | µ | 48.6 | 11.7 | 0.0058 |
| ± | 22.2 | 16.3 | ||
The EG displayed a statically significant (p = 0.0058) lower mean resolution of fibrosis (μ = 11.7 ± 16.3) than the CG (μ = 48.6 ± 22.2).
From the non-paired T test, there were significant differences (p < 0.05) between the EG and CG in resolution of ecchymosis (Table 3); thus, we rejected the null hypothesis.
| Variable | Group | P value | ||
|---|---|---|---|---|
| Control | Experimental | |||
| Resolution of ecchymosis | µ | 17.6 | 7.8 | 0.0002 |
| ± | 5.0 | 4.3 | ||
The EG displayed a statically significant (p = 0.0002) lower mean resolution of ecchymosis (µ = 7.8 ± 4.3) than the CG (μ = 17.6 ± 5.0).
p values < 0.05 indicate that the deviations are significant, that the variables are dependent, and that the samples differ significantly regarding the proportions of these classes; therefore, we rejected the null hypothesis (Table 4).
| Variable | Classes | Controle | Experimental | p valor |
|---|---|---|---|---|
| Fibrosis | No | 0 | 8 | *0.0003 |
| Yes | 10 | 2 | ||
| Initial fibrosis degree | 0 | 0 | 8 | *0.0002 |
| I | 0 | 2 | ||
| II | 6 | 0 | ||
| III | 4 | 0 | ||
| Initial thermography | I | 0 | 2 | *0.0002 |
| II | 6 | 0 | ||
| III | 4 | 0 | ||
| Normal | 0 | 8 | ||
| Intense edema | No | 5 | 10 | *0.0325 |
| Yes | 5 | 0 | ||
| Suggillation ecchymosis | Abdomen and flanks | 2 | 0 | *0.0056 |
| Lower abdomen | 0 | 1 | ||
| R and L lateral abdomen | 2 | 0 | ||
| Right flank | 0 | 1 | ||
| Flanks | 6 | 0 | ||
| R and L flanks | 0 | 1 | ||
| Lower lumbar | 0 | 1 | ||
| No | 0 | 6 |
* Significant differences between the means (p < 0.05).
The results indicate that:
The occurrence of fibrosis in the EG was significantly (p = 0.0003) less common than in the CG when all elements were presented;
The degree of fibrosis was lower in the EG (p = 0.0002);
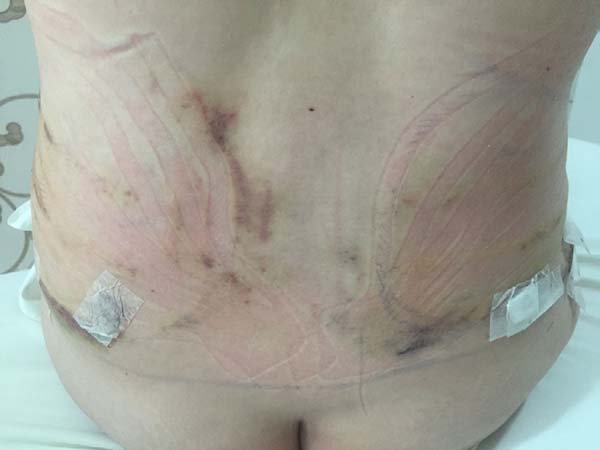
Initial thermography (Figures 4 and 5) was predominantly normal in the EG (p = 0.0002);
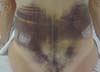
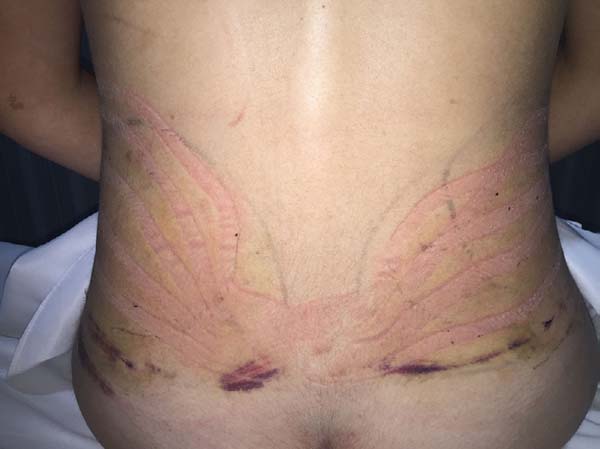
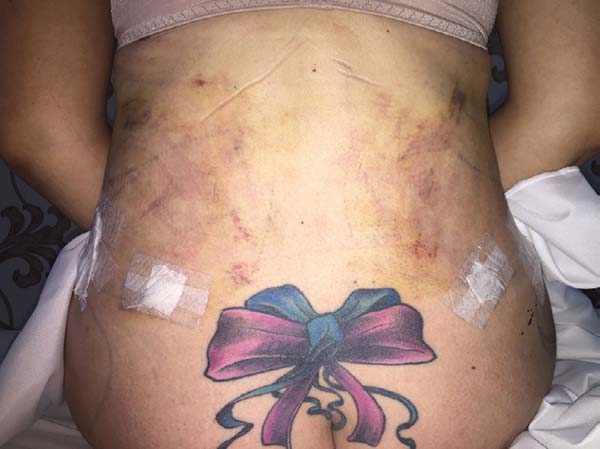
Non-occurrence of intense edema in the EG (p = 0.0325) as shown in Figures 6 and 7;
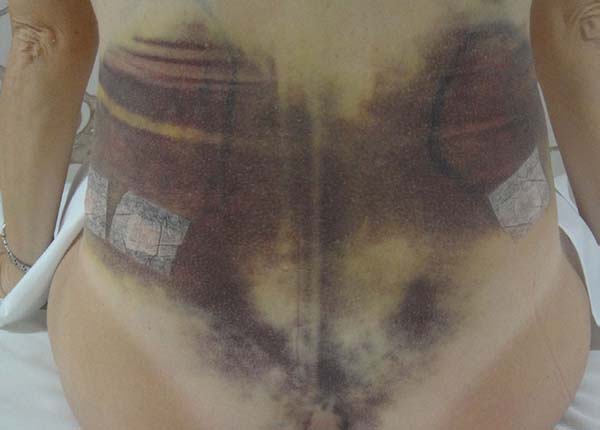
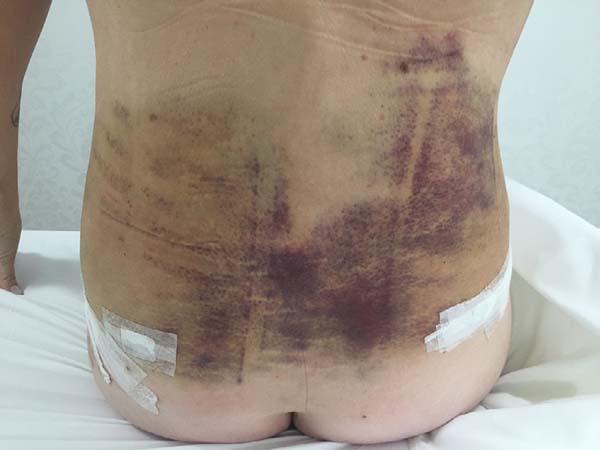
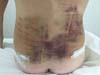
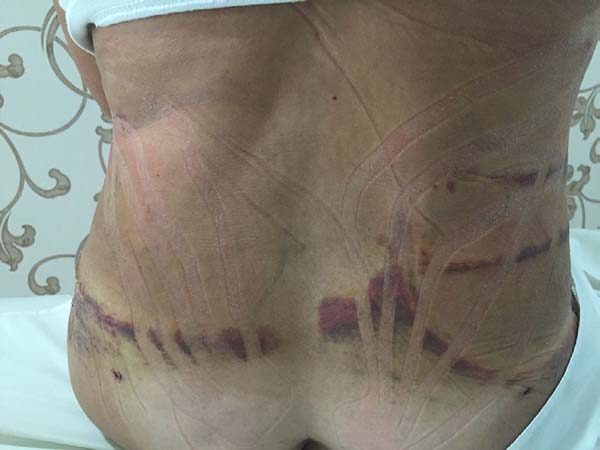
Non-occurrence of ecchymosis was higher in the EG (p = 0.0056) as shown in Figures 8 and 9;
Non-occurrence of ecchymosis in the flanks; abdomen and flanks; and lateral abdomen in the same proportions as in the CG (p = 0.0056) (Figures 10-13).
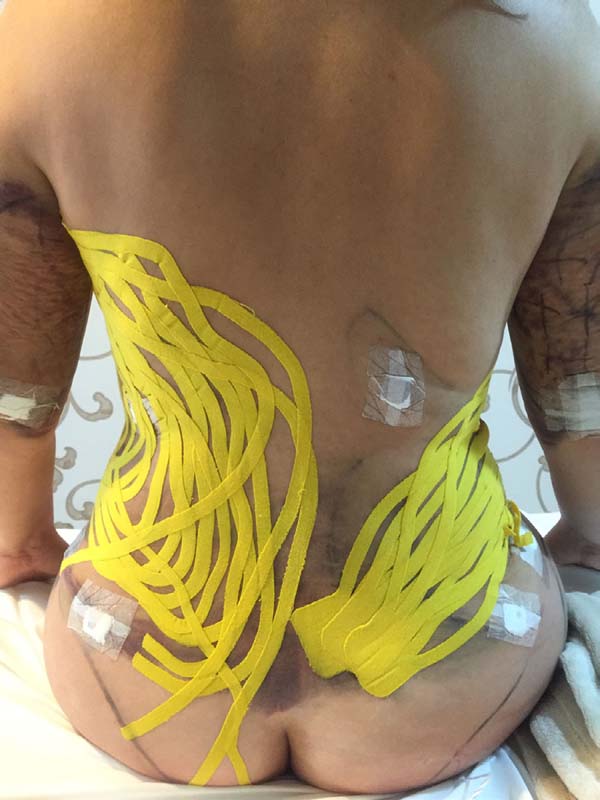
Moreover, in two patients in the EG, a difference was verified between the placement of the base of the taping compared to the axillary lymph node and coccygeal lymph node. There was no visual difference between the two positions in the two patients (Figures 14-17).
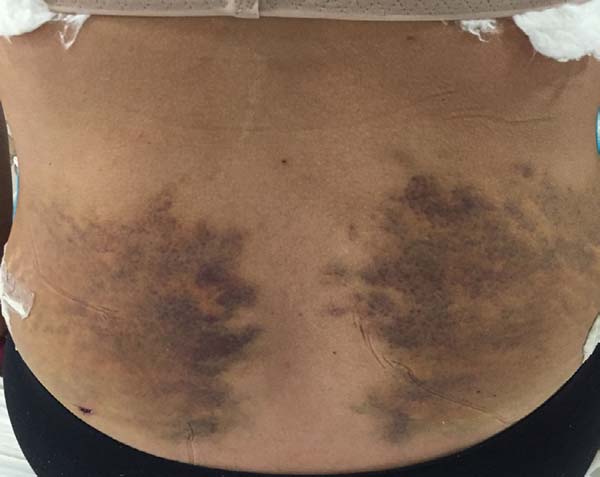
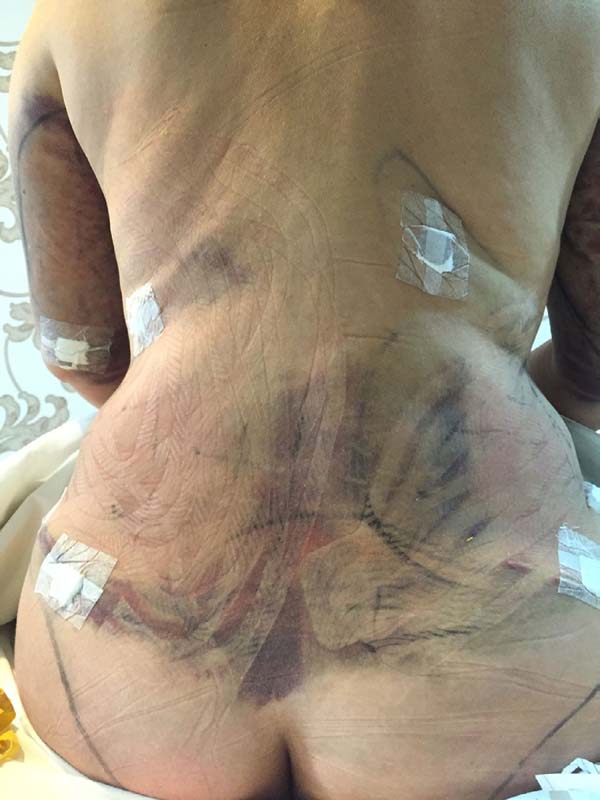
In regions in which taping was not used, a greater degree of visible ecchymosis was visible (Figures 18 and 19).
DISCUSSION
The variables of the EG that presented significant differences were number of sessions, beginning of fibrosis, resolution of fibrosis, resolution of ecchymosis, and perimetry on the 4th postoperative day.
The mean number of sessions was 14.6 in the EG and 23.1 in the CG, showing that the approach in the pre-, trans-, and postoperative periods reduces the number of sessions.
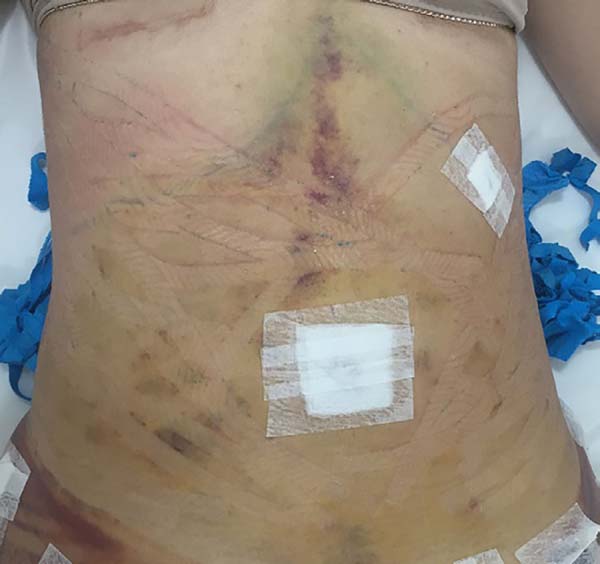
Fibrosis formation in the EG occurred in only 2 patients on the 18th and 16th postoperative day; in contrast, in the CG, all patients developed fibrosis on the 19th postoperative day. According to Lange7, Schwartz8, and Lange & Chi9, we usually palpate and/or visualize fibrosis after the first week due to the collagen synthesis being more intense between the 6th and 17th days. This corroborates the results of the EG in that 80% of those patients did not present fibrosis, as most did not present ecchymosis on the 4th postoperative day.
The resolution of fibrosis of the two patients in the EG occurred on the 45th and 40th postoperative days compared to 48.6 days in the CG. In this study, the preoperative period along with the lymphatic taping and containment foam transoperatively promoted improved metabolism due to constant lymphatic drainage.
Chi et al.10 showed results of lymphatic taping accompanied by manual lymphatic drainage in the postoperative period, in which they obtained a total reversal of fibrosis in patients who were in the proliferative phase suggesting the use of transoperative lymphatic taping to prevent fibrosis.
The EG obtained significant results (p = 0.0002) in the resolution of ecchymosis compared to the CG. This finding corroborates with those of Zanchet & Del Vecchio11, who found absorption of ecchymosis with application of taping.
Significant differences (p < 0.05) were observed on the 4th postoperative day in the inframammary groove and iliac crest, where we observed the largest perimetry in the CG compared to the EG. This shows that the preoperative period together with the use of the lymphatic taping and foam containment effectively reduced edema. There is no scientific evidence of this approach (pre-, trans-, and postoperative) in plastic surgery that would enable a comparison with our results.
For the treatment of edema, some studies are already been published, such as the case study of Chou et al.12 showing the effects of taping in a patient with lymphedema secondary to breast cancer. According to Bosman & Piller13, the application of taping favors the process of expansion of the initial lymphatic vessels, allowing the liquid present in the interstitium to be absorbed by the lymphatic network.
According to Van Zuilen et al.14, Neves et al.15, and Bosman & Piller13, the use of taping facilitates the drainage process due to the increase in space between the skin and muscle tissue, promoting the opening of lymphatic vessels and the sliding of the skin on the fascia, mechanism that consequently contribute to improvements in lymphatic and venous circulation.
The application of taping in its various forms aims to promote redirection of the lymphatic circulation, reducing edema in places where it is installed16. This finding corroborates the indications of Kase et al.17, who reported removal of edema from the directioning of exudates toward the lymphatic ducts and increasing the local circulation.
Furthermore, pain was not reported in the operated region in the EG. In this case, according to the findings of Chi et al.10, postoperative treatment should be started as early as possible to avoid possible postoperative complications such as seroma, prolonged edema, refractory ecchymosis, and intense pain.
Some authors have already reported several physiological effects after the use of taping such as reduction of pain or abnormal sensation and removal of lymphatic congestion, fluid, or bleeding under the skin18-22 as supported by our results.
There was a low occurrence of severe edema and ecchymoses in the EG. The use of a compression plaque for the operated region, involving the entire abdominal circumference of the patients, was associated to the application of the lymphatic taping technique to assist in the absorption of edema with the patient still in the surgical (transoperative) block and responsible for this low rate of edema and ecchymoses.
Another important factor in this study was the choice of cutting the lymphatic taping into the “fan” or “octopus” shapes. In this technique, the elastic band is cut into 4 or 5 strips with a fixed basis of approximately 5 cm. The articles identified in the literature using taping for edema20,21,23,24 used only the “fan” or «octopus» cut as described here.
According to Sijmonsma25, below the base of the “fan” (octopus) there is the possibility of triggering a skin irritation that does not occur below loose strips. In the present study, we observed no cases of skin irritation. Also, in accordance with the results of this study, Martins et al.26 described no cases of skin lesions, thus justifying the use of the fan cut for the treatment of edema.
Another aspect to highlight is the fact that use of the taping is associated with skin lesions or allergic reactions20; thus, we must prudently apply taping in smaller areas in an attempt to minimize the occurrence of these problems27. Therefore, the “fan” or “octopus” cut is suggested during trans- and postoperative plastic surgery care.
The treatment was initiated on the 4th postoperative day in both groups. The choice of the initiation of treatment was based on studies of Psillakis et al.28, who reported that the lymphatic anastomoses are more intense between the 4th and 7th postoperative days, in addition to the studies of Mendez et al.29, who mentioned that the absorption of interstitial fluid is hampered until the 5th postintervention day and returns to normal on the 10th day.
Flap adhesion occurs through a thin mesh of fibrin, which is infiltrated by fibroblasts that transform the tenuous adhesion into a definitive adhesion by fibrous tissue. An adhesion that allows the manipulation of the area repeatedly only occurs on the 4th postoperative day in agreement with the present study7.
The degree of fibrosis was lower in the EG (p = 0.0002) than in the CG. The excess edema favors fibrosis formation. The rapid absorption of local edema, through the action of the lymphatic taping associated with the uniform compression of the operated region, decreased even more when the accumulation of fluid inside the tunnels was triggered by the liposuction cannula. With these small spaces, scar tissue formation will be less likely, as will fibrosis formation30.
Fibrosis formation is mediated by the interaction between fibrinogenic growth factors and pro-fibrotic cytokines in addition to other influences such as mechanical stress, chronic inflammation, and oxidative stress31. For the EG, the use of oral and topical antiglycans and nutritional guidelines was indicated to better control inflammation and oxidative stress.
According to Rocha & Paula32, it is possible to prevent or alleviate complications such as fibrosis using functional foods. Modulating the inflammatory process, a diet rich in anti-inflammatory agents can prevent, combat, and even reverse some of the damage caused by inflammation36,33.
The analysis of fibrosis degree was also evaluated by contact thermography from the 4th postoperative day, and the findings were predominantly normal in the EG (p = 0.0002). Chi et al.10 used contact thermography as an instrument for the early detection and classification of fibrosis.
The difference between the placements of the base of the taping in comparison to the two regions, axillary lymph nodes and coccygeal lymph node, was analyzed in two patients in the EG, but no visual difference was noted between the two positions, so either was considered acceptable.
In all patients of the EG, in areas in which taping was not applied, no areas with ecchymosis and edema were observed.
Taping should cover the entire area with edema. When applied to the skin, it provides a greater opening of initial lymphatics, favoring the absorption of interstitial fluid into the lymphatic channel34-36.
The low rate of fibrosis formation in the EG was due to several factors. Among them, a preoperative conduct consisting of an anti-inflammatory diet and restriction of foods with a high glycemic index associated with the oral use of nutricosmetics and topical use of active antiglycants and anti-inflammatory agents; and conduct during surgery included the use of a compression plate and the placement of the lymphatic taping during surgery.
Individuals in the EG reacted differently in the postoperative period compared to those in the CG, with evidence of the influence of different classifications on the results of the statistical tests. Thus, the treatment result depends statistically on the pre- (nutritional guidelines, use of topical and oral nutricosmetics) and trans-operative (taping and containment foam) measures.
Our results suggest that the preoperative use of antiglycant cosmetics, nutricosmetics, and anti-inflammatory medications associated with the transoperative placement of lymphatic taping below the containment foam reduces edema, ecchymosis formation, and fibrosis formation in the postoperative period. It also decreases the number of required physiotherapy sessions and accelerates the patient’s recovery from abdominal surgeries.
COLLABORATIONS
|
AC |
Analysis and/or interpretation of data; final approval of the manuscript; conception and design of the study; completion of surgeries and/or experiments; writing the manuscript or critical review of its contents. |
|
AL |
Final approval of the manuscript; writing the manuscript or critical review of its contents. |
|
MVTNG |
Final approval of the manuscript. |
|
CBS |
Statistical analyses. |
REFERENCES
1. International Society of Aesthetic Plastic Surgery (ISAPS) [Internet]. 2017 [cited 2017 Ago 9]. Available from: https://www.isaps.org
2. Soncini JA, Baroudi R. Revisão da técnica de abdominoplastia com dissecção reduzida e fixação com pontos de Baroudi. Rev Bras Cir Plást. 2016;31(2):166-71.
3. Souza LS, Harada MN, Bolognani EMC. Comparação da ocorrência de seroma entre as técnicas de abdominoplastia convencional e em âncora nos pacientes pós-bariátricos. Rev Bras Cir Plást. 2017;32(1):78-86.
4. Carloni R, Naudet F, Chaput B, de Runz A, Herlin C, Girard P, et al. Are There Factors Predictive of Postoperative Complications in Circumferential Contouring of the Lower Trunk? A Meta-Analysis. Aesthet Surg J. 2016;36(10):1143-54. DOI: http://dx.doi.org/10.1093/asj/sjw117
5. Masson IF, de Oliveira BD, Machado AF, Farcic TS, Júnior IE, Baldan CS. Manual lymphatic drainage and therapeutic ultrasound in liposuction and lipoabdominoplasty post-operative period. Indian J Plast Surg. 2014;47(1):70-6. DOI: http://dx.doi.org/10.4103/0970-0358.129627
6. Macedo ACB, Oliveira SM. A atuação da fisioterapia no pré e pós-operatório de cirurgia plástica corporal: uma revisão de literatura. Cad Esc Saúde. 2011;1(5):169-89.
7. Lange A. Fisioterapia Dermato Funcional Aplicado à Cirurgia Plástica. Curitiba; 2017.
8. Schwartz SI. Princípios de Cirurgia. Rio de Janeiro: Guanabara Koogan; 1987.
9. Lange A, Chi A. Fibrose: da prevenção ao tratamento. Curitiba; 2018.
10. Chi A, Oliveira AVM, Ruh AC, Schleder JC. O uso do linfotaping, terapia combinada e drenagem linfática manual sobre a fibrose no pós-operatório de cirurgia plástica de abdome. Fisioter Bras. 2016;17(3):197-203.
11. Zanchet MA, Del Vecchio FB. Efeitos da bandagem kinesio taping(tm) na recuperação de hematoma. Decorrente de distensão durante a prática do tênis: um estudo de caso. In: Anais do XVII Congresso Brasileiro de Ciências do Esporte e IV Congresso Internacional de Ciências dos Esporte. Porto Alegre: CONBRACE; 2011.
12. Chou YH, Li SH, Liao SF, Tang HW. Case report: Manual lymphatic drainage and kinesio taping in the secondary malignant breast cancer-related lymphedema in an arm with arteriovenous (A-V) fistula for hemodialysis. Am J Hosp Palliat Care. 2013;30(5):503-6. DOI: http://dx.doi.org/10.1177/1049909112457010
13. Bosman J, Piller N. Lymph taping and seroma formation post breast cancer. J Lymphoed. 2010;5(2):12-21.
14. Van Zuilen M. Técnicas de aplicação de bandas neuro-musculares. Cascais: Aneid, Produtos Farmacêuticos; 2011.
15. Neves AC, Cruz AC, Pinho AM, Barreira AS, Tenreiro A, Fernandes D,, et al. Bandas neuromusculares: um complemento na abordagem da fisioterapia mastectomia [Internet]. Notícias de Bandas Neuromusculares; 2010 [acesso 2018 Maio 3]. Disponível em: https://www.aneid.pt/wp-content/uploads/2018/03/news_bandas_set10.pdf
16. Pyszora A, Krajnik M. Is Kinesio Taping useful for advanced cancer lymphoedema treatment? A case report. Adv Palliat Med. 2010;9(4):141-4.
17. Kase K, Lemos TV, Dias EM. Kinesio Taping - Introdução ao Método e Aplicações Musculares. São Paulo: Andreoli; 2013.
18. Tsai HJ, Hung HC, Yang JL, Huang CS, Tsauo JY. Could Kinesio tape replace the bandage in decongestive lymphatic therapy for breast-cancer-related lymphedema? A pilot study. Support Care Cancer. 2009;17(11):1353-60. DOI: http://dx.doi.org/10.1007/s00520-009-0592-8
19. Aguilar-Ferrándiz ME, Castro-Sánchez AM, Matarán-Peñarrocha GA, Guisado-Barrilao R, García-Ríos MC, Moreno-Lorenzo C. A randomized controlled trial of a mixed Kinesio taping-compression technique on venous symptoms, pain, peripheral venous flow, clinical severity and overall health status in postmenopausal women with chronic venous insufficiency. Clin Rehabil. 2014;28(1):69-81. DOI: http://dx.doi.org/10.1177/0269215512469120
20. Smykla A, Walewicz K, Trybulski R, Halski T, Kucharzewski M, Kucio C, et al. Effect of Kinesiology Taping on breast cancer-related lymphedema: a randomized single-blind controlled pilot study. Biomed Res Int. 2013;2013:767106. DOI: http://dx.doi.org/10.1155/2013/767106
21. Pekyavas NÖ, Tunay VB, Akbayrak T, Kaya S, Karatas M. Complex decongestive therapy and taping for patients with postmastectomy lymphedema: a randomized controlled study. Eur J Oncol Nurs. 2014;18(6):585-90. DOI: http://dx.doi.org/10.1016/j.ejon.2014.06.010
22. Tozzi U, Santagata M, Sellitto A, Tartaro GP. Influence of Kinesiologic Tape on Post-operative Swelling After Orthognathic Surgery. J Maxillofac Oral Surg. 2016;15(1):52-8. DOI: http://dx.doi.org/10.1007/s12663-015-0787-0
23. Taradaj J, Halski T, Rosinczuk J, Dymarek R, Laurowski A, Smykla A. The influence of Kinesiology Taping on the volume of lymphoedema and manual dexterity of the upper limb in women after breast cancer treatment. Eur J Cancer Care (Engl). 2016;25(4):647-60. DOI: http://dx.doi.org/10.1111/ecc.12331
24. Gatt M, Willis S, Leuschner S. A meta-analysis of the effectiveness and safety of kinesiology taping in the management of cancer-related lymphoedema. Eur J Cancer Care (Engl). 2017;26(5). DOI: http://dx.doi.org/10.1111/ecc.12510
25. Sijmonsma J. Lymph Taping. In: Sijmonsma J, ed. Lymph taping. Hof van Twente: Fysionair; 2010. p. 57-84.
26. Martins Jde C, Aguiar SS, Fabro EA, Costa RM, Lemos TV, de Sá VG, et al. Safety and tolerability of Kinesio Taping in patients with arm lymphedema: medical device clinical study. Support Care Cancer. 2016;24(3):1119-24. DOI: http://dx.doi.org/10.1007/s00520-015-2874-7
27. Pivetta HMF, Petter GN, Penna GB, Martins TNO, Santos LF, Pautz ACG. Efeitos do Kinesio Taping sobre o edema linfático. Fisioter Bras. 2017;3(18):382-90.
28. Psillakis JM, de Jorge FB, Villardo R, de Albano AM, Martins M, Spina V. Water and electrolyte changes in autogenous skin grafts. Discussion of the so-called "plasmatic circulation". Plast Reconstr Surg. 1969;43(5):500-3. PMID: 4889412 DOI: http://dx.doi.org/10.1097/00006534-196905000-00008
29. Mendez U, Brown EM, Ongstad EL, Slis JR, Goldman J. Functional recovery of fluid drainage precedes lymphangiogenesis in acute murine foreleg lymphedema. Am J Physiol Heart Circ Physiol. 2012;302(11):H2250-6. PMID: 22427513 DOI: http://dx.doi.org/10.1152/ajpheart.01159.2011
30. Gantwerker EA, Hom DB. Skin: histology and physiology of wound healing. Facial Plast Surg Clin North Am. 2011;19(3):441-53. DOI: http://dx.doi.org/10.1016/j.fsc.2011.06.009
31. Mu X, Bellayr I, Walters T, Li Y. Mediators leading to fibrosis - how to measure and control them in tissue engineering. Oper Tech Orthop. 2010;20(2):110-8. DOI: http://dx.doi.org/10.1053/j.oto.2009.10.003
32. Rocha CL, Paula VB. Nutrição funcional no pós-operatório de cirurgia plástica: enfoque na prevenção de seroma e fibrose. Rev Bras Cir Plást. 2014;29(4):609-24.
33. Pujol AP. Nutrição aplicada à estética. Rio de Janeiro: Rubio; 2011.
34. Fernandes J, Chandía PY, Garcia FE. Tape Neuro Muscular - Aplicaciones Prácticas. Buenos Aires: TNM Argentina; 2016.
35. Bosman J. Lymph taping for lymphoedema: an overview of the treatment and its uses. Br J Community Nurs. 2014;Suppl:S12, S14, S16-8. DOI: http://dx.doi.org/10.12968/bjcn.2014.19.Sup4.S12
36. Pamuk U, Yucesoy CA. MRI analyses show that kinesio taping affects much more than just the targeted superficial tissues and causes heterogeneous deformations within the whole limb. J Biomech. 2015;48(16):4262-70. DOI: http://dx.doi.org/10.1016/j.jbiomech.2015.10.036
1. Instituto Universitário Italiano de Rosário,
Santa Fe, Argentina.
2. Pontifícia Universidade Católica do Paraná,
Curitiba, PR, Brazil.
3. Universidade Tuiuti do Paraná, Curitiba, PR,
Brazil.
4. Instituto Marcus Thomé, Ponta Grossa, PR,
Brazil.
5. Faculdade IBRATE, Curitiba, PR,
Brazil.
6. Centro Universitário de Volta Redonda, Rio de
Janeiro, RJ, Brazil.
7. Associação Médica Brasileira, São Paulo, SP,
Brazil.
8. Sociedade Brasileira de Cirurgia Plástica, São
Paulo, SP, Brazil.
9. Universidade Estadual de Ponta Grossa, Ponta
Grossa, PR, Brazil.
10. Universidade Tecnológica Federal do Paraná,
Curitiba, PR, Brazil.
Corresponding author, Anny Chi, Rua Nestor Guimarães, 77 - Ponta Grossa, PR, Brazil. Zip Code 84040-130. E-mail: annychi10@hotmail.com
Article received: October 14, 2017.
Article accepted: September 5, 2018.
Conflicts of interest: none.

















 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter