

Original Article - Year 2018 - Volume 33 -
New proposal for the use of the Becker 35 expander in second large volume breast reconstruction
Nova proposta para utilização de Becker 35 no segundo tempo de reconstrução mamária de grandes volumes
ABSTRACT
Introduction: Immediate breast reconstruction performed
with the Becker expander is a versatile and accepted technique.
We present an original proposal for the use of the Becker
expander for a second breast implant.
Methods: A retrospective
study was performed between January 2014 and October
2016. Medical records were used to evaluate the indications
and complications, the use of radio- and chemotherapy, and
comorbidities in all patients, including 5 in whom combined
implantation was performed.
Results: The Becker expander
was used for 193 reconstructions in 168 patients, including
25 cases with bilateral reconstructions. The average patient
age was 47 years, and 33% had comorbidities. Postoperative
radiotherapy and chemotherapy were performed in 119
patients (62%). Complications included seroma in 7 patients,
hematoma in the mastectomy flap in 2 patients, partial necrosis
of the mastectomy flap in 3 patients, and necrosis/infection
with expander exposure in 2 patients. Complications occurred
in 9.5% of the cases. A total of 133 (69%) patients underwent
a second surgical stage to complement breast reconstruction,
with the Becker expander being replaced by a definitive
implant. Patients in whom the Becker 35 expander was used
in a second breast implant developed no complications.
Conclusions: the use of the Becker 35 expander for a second
breast implant was effective in achieving greater volume
in breast reconstruction with a low rate of complications.
Keywords: Mammaplasty; Reconstructive surgical procedures; Breast implantation; Tissue expansion devices; Breast
RESUMO
Introdução: A reconstrução imediata de mama realizada com o expansor de Becker é uma
técnica de reconstrução versátil e consolidada na prática médica.
Apresentamos uma proposta original com o uso do expansor de Becker associado
a um segundo implante mamário.
Métodos: Estudo retrospectivo realizado no período de janeiro de 2014 a outubro de
2016 no Hospital A.C. Camargo. A análise dos prontuários das pacientes
reconstruídas com Becker 35 avaliou o índice e tipo de complicações e a
associação de rádio e quimioterapia e comorbidades das pacientes com 5 casos
de associação de implantes.
Resultados: 193 reconstruções com Becker em 168 pacientes. Vinte cinco casos com
reconstrução bilateral. Idade média de 47 anos e 33% apresentaram
comorbidades. Cento e dezenove (62%) pacientes seguiram radioterapia e
quimioterapia pós-operatórias. Complicações: seroma em 7 pacientes, hematoma
no retalho da mastectomia em 2 pacientes, necrose parcial do retalho da
mastectomia em 3 pacientes, necrose/infecção com exposição do expansor em 2
pacientes. O total de complicações foi de 9,5%. Um total de 133 (69%)
pacientes foram submetidas a esta segunda etapa cirúrgica para
complementação da reconstrução da mama, sendo o expansor de Becker
substituído por um implante definitivo. As pacientes com uso de Becker 35
associado a outro implante não apresentaram complicações.
Conclusões: A associação do implante-expansor de Becker 35 a um segundo implante mamário
foi eficaz em atingir um volume maior nas reconstruções mamárias com baixo
índice de complicações.
Palavras-chave: Mamoplastia; Procedimentos cirúrgicos reconstrutivos; Implante mamário; Dispositivos para expansão de tecidos; Mama
INTRODUCTION
Breast reconstruction is part of a multidisciplinary approach to treat breast cancer. It can be performed using flaps (autologous tissue) and breast implants alone or in combination.
Immediate breast reconstruction (IBR) follows mastectomy. Collaboration between the mastologist and plastic surgeon is necessary. Advantages include a single hospital stay, a single operation under anesthesia, reduced hospital costs, ease of exposure of the surgical field by mastectomy for reconstruction, aesthetically restored balance, and psychological reassurance for the patient1,2.
In 1984, Becker advocated the use of a permanent expander, combining the benefits of silicone gel, saline implant, and expander implant into a single product, which could allow breast reconstruction in a single step.
The definitive Becker expander has a double lumen and a textured external surface and contains silicone gel as 35% of the expander volume in the anterior layer, with a posterior chamber for an additional 65% that can be filled with saline solution using an external valve3. IBR using the breast expander is simpler and faster, with shorter convalescence time compared to other reconstruction techniques4,5.
Despite being a simple method for breast reconstruction, the use of the Becker expander has unique features and like any technique is associated with some complications.
Late complications may be intense and deforming in a minority of patients and may be a cause of pain, especially after radiotherapy6. Other complications include seroma formation, infections, and tissue necrosis resulting from the surgical trauma of mastectomy7,8. Inconveniences include the acquisition and cost of expanders, quality control, risk of elastomer rupture, and need for outpatient follow-up for expansion sessions9.
Many implants are available, but in patients with very large breasts, no implants have sufficient volume for adequate reconstruction.
The original proposal of this study is the use of the Becker expander combined with a second breast implant. The Becker expander is the basis for the second implant, thus determining the final volume of the reconstructed breast.
OBJECTIVE
The objective of this work is to present a new proposal for breast reconstruction using the Becker 35 expander in large-volume breast reconstruction.
METHODS
This retrospective study examined the medical records of the senior author’s patients. The following inclusion criteria were established: patients who had undergone breast reconstruction using the Becker Expander 35, in which posterior breast symmetrization had been performed, but the volume of the new implant was insufficient for aesthetic adequacy even with the reduction in the size of the opposite breast. The study period was between January 2014 and October 2016.
The consultations, diagnostic investigation, surgical treatment, adjuvant oncological treatment, and clinical oncological follow-up were performed at the Hospital AC Camargo - Cancer Center of Fundação Antônio Prudente - São Paulo, SP.
The patients were followed-up by a multidisciplinary team from the Hospital do Câncer that included professionals from the following specialties:
Plastic Surgeons - Performed evaluation of the patient’s psychological motivation and general and specific clinical and physical conditions relevant to reconstruction, such as contralateral breast conditions, scars, shape, and volume of the breasts. The donor areas were determined with the participation of the patient.
Mastologists - Were involved in staging, on the basis of clinical parameters, imaging, and anatomic-pathologic assessment according to the TNM classification (Union for International Cancer Control). The mastologist performed mastectomy and was involved in ongoing oncological management.
Radiotherapists - Assisted in follow-up, methodology, and management of complications.
Clinical Oncologists - Were involved in the evaluation of clinical conditions, choice of drugs/doses, and management of complications of chemotherapy.
Anesthetists - Performed preoperative evaluation and determined the anesthetic technique.
Pathologists - Performed all tissue examinations including immunohistochemistry.
Psychiatrists - Provided support for mastectomy patients.
Physiotherapists - Enabled upper limb functional recovery after mastectomy.
A Dressing group - Provided nursing with specific techniques for use in breast reconstruction.
The expander is placed in submuscular position, following dissection of the pectoralis major, with additional coverage using the serratus anterior and anterior sheath of the rectus abdominis when needed.
Dissection was performed with reference to the original submammary sulcus and the anatomic limits of the breast. This ensures adequate coverage for the expander. The volume of the expander is determined by reference to the contralateral breast and the weight of the mastectomy product.
When implants overlapped, the Becker expander was completely emptied prior to removal of the valve and adjustments were performed when necessary.
Vacuum drainage was routinely used.
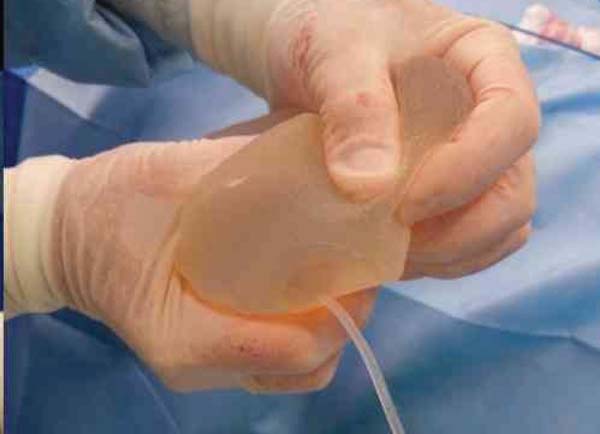
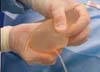
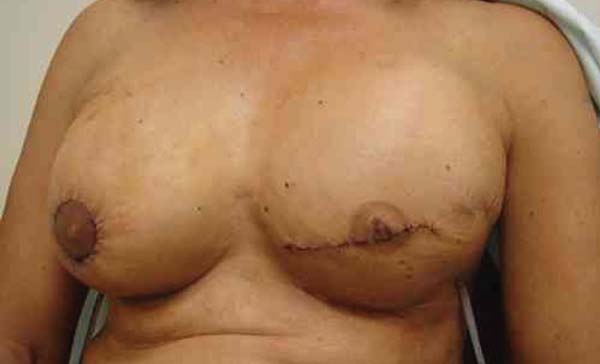
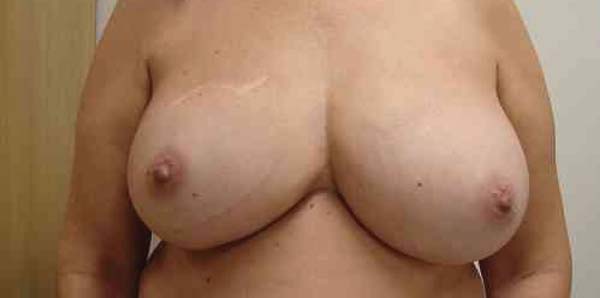
When the proposed technique was used in the second surgical step, the procedure was as follows. The expander was completely emptied (Figure 1), maintaining only the silicone, i.e., 35% of the original volume of the expander (Figure 2).
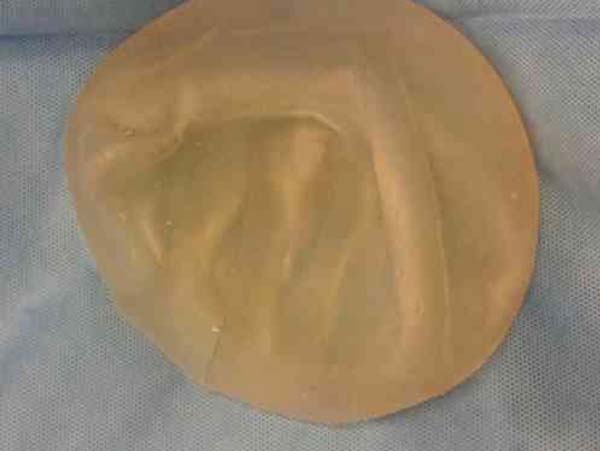
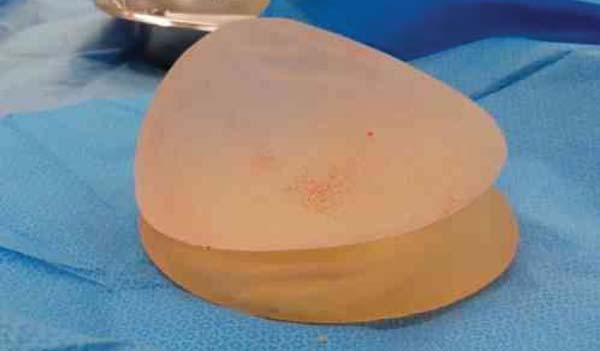
When the expander was drained, the valve and connector were also removed. The Becker expander was used as a basis to support a conventional breast implant with a round (Figure 3) or anatomical profile (Figure 4).
The study was approved by the institutional ethics committee.
RESULTS
Of 168 patients who underwent IBR, 25 had a bilateral procedure, for a total of 193 breast reconstructions. The mean age was 47 years; 49 patients were smokers, 57 had systemic arterial hypertension, 22 had diabetes, 9 had heart disease, and 5 had other diseases.
Complications in this group included seroma in 7 patients, hematoma in the mastectomy flap in 2, partial necrosis of the mastectomy flap in 3, and necrosis/infection with expander exposure in 2. One or more complications occurred in 9.5% of all cases.
One or more clinical aggravating factors were present in 33% of patients undergoing mastectomy and immediate reconstruction.
TNM stages ranged from Tis / T1n0m0 to T4n2m0. The mean surgical time for breast reconstruction was 1 hour.
Regarding adjuvant treatment, 119 (62%) patients had postoperative radiotherapy and chemotherapy and 83 received chemotherapy alone.
Only plastic surgery was performed in the second surgical phase of breast reconstruction. In this second phase, reconstruction of the papillary plate and mammaplasty for contralateral breast symmetrization were performed, when necessary, in unilateral cases, with or without the use of breast implants.
A total of 133 (69%) patients underwent this second surgical stage to complement breast reconstruction, with the Becker expander being replaced by a definitive implant. Complications in this group included seroma in 1 patient, hematoma in the mastectomy flap in 2, contralateral mammaplasty hematoma in 2, marginal necrosis in the mastectomy flap in 3, and necrosis/infection with breast implant exposure in 2 (more than one complication may have occurred in the same patient). One or more complications associated with the second surgical phase occurred in 4.5% of cases.
The technique proposed in this study was adopted in 5 patients (3.75%): the empty Becker expander was used with a second breast implant. There were no complications in this group. The aim of this technique was to increase the volume, base, and projection of the reconstructed breast, in which use of a mammary implant alone would not be satisfactory.
DISCUSSION
Appropriate selection of patients, planning, and good technical execution are essential for a good result in breast reconstruction without complications10,11. However, patient characteristics such as large breast volume, a wide breast base, and obesity are challenges often encountered by the plastic surgeon (Figuras 5, 6 e 7).
The Becker expander is a very useful tool for breast reconstruction, but its use should be limited6. Becker’s initial proposal for reconstruction in a single operation has not proved feasible in the long run, since many surgical adjustments are necessary to achieve an optimal outcome12. The volume of the implant is a factor that often limits its permanence.
Exchange of the expander with a definitive implant is generally well tolerated by the patient and is an opportunity to make new adjustments for adequate symmetrization13. In the present study, the need for capsulotomy was considered, as well as adequacy of lateral projection of the implant in the thoracic cavity with limited attachment points, adjustment in the height of the mammary groove, and vertical repositioning of the implant, since the expander tends to be in a higher thoracic position.
The largest available Becker expander accommodates 685 cc, with temporary overexpansion of up to 795 cc. The high-volume, high-profile round implant (Mentor) accommodates 800 cc. In 5 patients in this study, the volume provided by an available implant would not have been sufficient to reach the desired volume or sufficient to obtain adequate symmetry. Even if the volume provided by a single implant was sufficient, the breast base, breast height, and projection ratios would not have been not adequate in these 5 patients with very bulky breasts.
We believe that the technical features of the Becker 35 expander provide larger horizontal/vertical measurements than breast implants, despite smaller anterior projection, enabling overlap with a second breast implant to obtain more adequate symmetry.
In the present study, the replacement rate of the expanders was 69%. Chew et al.8 reported a 68% exchange and/or removal rate for expanders in 5 years of follow-up. In Brazil, Cammarota et al.6 reported a 28.57% replacement rate for expanders combined with implants. In 15.53% of the cases, the procedure was sufficient, without need for additional methods.
Complications did not occur with this procedure; moreover, no comparison is available in the literature for the proposed technique.
Adipose tissue grafting is a current proposal in mammary reconstruction enhancement and has proven to be very effective, as demonstrated by Bezerra et al.14 However, in cases requiring large volumetric increases, several approaches may be needed to achieve this goal. In Brazil, Blumenschein et al.15 reported the possible use of lipoenxertia in breast reconstruction to achieve an aesthetic volume increase.
CONCLUSION
The use of a Becker 35 implant with a second breast implant was effective in achieving larger volume in mammary reconstruction in carefully selected patients. The technique is reproducible and has a low rate of complications. More studies and longer postoperative follow-up are necessary to reach more specific conclusions.
REFERENCES
1. Cheville AL, Tchou J. Barriers to rehabilitation following surgery for primary breast cancer. J Surg Oncol. 2007;95(5):409-18. PMID: 17457830 DOI: http://dx.doi.org/10.1002/jso.20782
2. Parker PA, Youssef A, Walker S, Basen-Engquist K, Cohen L, Gritz ER, et al. Short-term and long-term psychosocial adjustment and quality of life in women under-going different surgical procedures for breast cancer. Ann Surg Oncol. 2007;14(11):3078-89. DOI: http://dx.doi.org/10.1245/s10434-007-9413-9
3. Becker H. Breast reconstruction using an inflatable breast implant with detachable reservoir. Plast Reconstr Surg. 1984;73(4):678-83. PMID: 6709750 DOI: http://dx.doi.org/10.1097/00006534-198404000-00031
4. Becker H. The expandable mammary implant. Plast Reconstr Surg. 1987;79(4):631-7. PMID: 3823256 DOI: http://dx.doi.org/10.1097/00006534-198704000-00023
5. Becker H. The permanent tissue expander. Clin Plast Surg. 1987;14(3):519-27. PMID: 3608362
6. Cammarota MC, Lima RQ, Almeida CM, Esteves BP, Curado DMDC, Ribeiro Júnior I, et al. Reconstrução de mama com expansor de Becker: uma análise de 116 casos. Rev Bras Cir Plast. 2016;31(1):12-8.
7. Farace F, Faenza M, Bulla A, Rubino C, Campus GV. Is mammary reconstruction with the anatomical Becker expander a simple procedure? Complications and hidden problems leading to secondary surgical procedures: a follow-up study. J Plast Reconstr Aesthet Surg. 2013;66(6):741-6. DOI: http://dx.doi.org/10.1016/j.bjps.2013.02.004
8. Chew BK, Yip C, Malyon AD. Becker expander implants: truly a long term single stage reconstruction? J Plast Reconstr Aesthet Surg. 2010;63(8):1300-4.
9. Camilleri IG, Malata CM, Stavrianos S, McLean NR. A review of 120 Becker per-manent tissue expanders in reconstruction of the breast. Br J Plast Surg. 1996;49(6):346-51. DOI: http://dx.doi.org/10.1016/S0007-1226(96)90001-6
10. De Vita R, Zoccali G, Buccheri EM, Costantini M, Botti C, Pozzi M. Outcome Evaluation after 2023 Nipple-Sparing Mastectomies: Our Experience. Plast Reconstr Surg. 2017;139(2):335e-47e. DOI: http://dx.doi.org/10.1097/PRS.0000000000003027
11. Munhoz AM, Aldrighi CM, Montag E, Arruda EG, Aldrighi JM, Gemperli R, et al. Clinical outcomes following nipple-areola-sparing mastectomy with immediate implant-based breast reconstruction: a 12-year experience with an analysis of patient and breast-related factors for complications. Breast Cancer Res Treat. 2013;140(3):545-55. PMID: 23897416 DOI: http://dx.doi.org/10.1007/s10549-013-2634-7
12. Eriksen C, Lindgren EN, Frisell J, Stark B. A prospective randomized study com-paring two different expander approaches in implant-based breast reconstruction: one stage versus two stages. Plast Reconstr Surg. 2012;130(2):254e-64e. DOI: http://dx.doi.org/10.1097/PRS.0b013e3182589ba6
13. Farace F, Faenza M, Bulla A, Rubino C, Campus GV. Is mammary reconstruction with the anatomical Becker expander a simple procedure? Complications and hidden problems leading to secondary surgical procedures: a follow-up study. J Plast Reconstr Aesthet Surg. 2013;66(6):741-6. DOI: http://dx.doi.org/10.1016/j.bjps.2013.02.004
14. Bezerra FJF, Moura RMG, Maia Neto JD. Lipoenxertia em reconstrução mamária. Rev Bras Cir Plást. 2013;28(2):241-6. DOI: http://dx.doi.org/10.1590/S1983-51752013000200012
15. Blumenschein AR, Freitas-Junior R, Tuffanin AT, Blumenschein DI. Lipoenxertia nas mamas: procedimento consagrado ou experimental? Rev Bras Cir Plást. 2012;27(4):616-22. DOI: http://dx.doi.org/10.1590/S1983-51752012000400025
1. Hospital A.C. Camargo, Cancer Center, São
Paulo, SP, Brazil.
2. Sociedade Brasileira de Cirurgia Plástica, São
Paulo, SP, Brazil.
Corresponding author: Alexandre Katalinic Dutra, Rua Prof. Antônio Prudente, 211 - Liberdade - São Paulo, SP, Brazil. Zip Code 01509-010. E-mail: akdutra@uol.com.br
Article received: February 1, 2017.
Article accepted: September 5, 2018.
Conflicts of interest: none.









 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter