

Original Article - Year 2018 - Volume 33 -
Breast reconstruction with latissimus dorsi myocutaneous flap and silicone implant in the single lateral decubitus position: systematization and case series
Reconstrução mamária em decúbito único lateral com retalho miocutâneo do músculo grande dorsal e implante de silicone: sistematização e série de casos
ABSTRACT
Introduction: Breast cancer is an important public health
condition, and its surgical treatment and the subsequent breast
reconstruction has evolved significantly over time. The use of the
latissimus dorsi myocutaneous flap (LDMF) with silicone breast
implants is a mainstay of plastic surgeons due to the anatomical
reliability and safety. Performing this surgery in the single lateral
decubitus position makes the procedure shorter and maintains the
results of the conventional technique. The objective of this study
was to present a systematization of this surgery from a series of
cases.
Method: Retrospective study using medical records and
photographic documentation of 29 patients operated on by the
author and submitted to breast reconstruction with a LDMF
and silicone implant in a lateral decubitus position.
Results: Silicone implant extrusion due to seroma and infection at the
receptor site was noted in one patient (3.5%). One patient had
a seroma on the back (3.5%); there was one patient with partial
necrosis of the skin island of the flap (3.5%), and two patients with
mastectomy skin envelope (7.0%). One patient required removal
of the silicone implant due to extensive local recurrence (3.5%)
while another patient showed Baker III capsular contracture
after adjuvant radiotherapy (3.5%). Four patients had scar
review (14%), and three patients showed limitation of movement
(10.5%). The results were comparable to those reported for the
conventional technique.
Conclusion: The systematization of this
technique requires no change of the decubitus position and makes
the procedure shorter, while maintaining safety and reliability.
Keywords: Breast cancer; Mastectomy; Breast Implant; Breast
RESUMO
Introdução: O câncer de mama é afecção de grande relevância para a saúde pública, sendo
que seu tratamento cirúrgico e a reconstrução mamária evoluíram bastante ao
longo do tempo. A utilização do retalho miocutâneo do grande dorsal (RMGD)
com implante mamário de silicone apresenta-se como um verdadeiro cavalo de
batalha ao cirurgião plástico em função de sua confiabilidade anatômica e
segurança. Realizar esta cirurgia em decúbito único lateral torna o
procedimento mais breve e mantém os resultados da técnica convencional. O
objetivo do estudo é apresentar a sistematização desta cirurgia em uma série
de casos.
Método: Estudo retrospectivo realizado por análise de prontuários e documentação
fotográfica de 29 pacientes operadas pelo autor e submetidas à reconstrução
mamária com RMGD e implante de silicone em decúbito único lateral.
Resultados: Houve um caso de extrusão do implante de silicone por seroma e infecção no
sítio receptor (3,5%). Uma paciente cursou com seroma no dorso (3,5%). Um
caso de necrose parcial da ilha de pele do retalho (3,5%) e dois casos de
sofrimento do envelope cutâneo da mastectomia (7,0%). Uma paciente
necessitou retirada do implante de silicone por extensa recidiva local
(3,5%). Um caso de contratura capsular Baker III após radioterapia adjuvante
(3,5%). Quatro pacientes tiveram suas cicatrizes revisadas (14%). Três
pacientes tiveram limitação de movimentos (10,5%). Os resultados
mostraram-se compatíveis com os apresentados para a técnica convencional na
literatura vigente.
Conclusão: A sistematização desta técnica dispensa mudança de decúbito e torna o
procedimento mais breve, mantendo sua segurança e confiabilidade.
Palavras-chave: Neoplasias da mama; Mastectomia; Implante mamário; Mama
INTRODUCTION
Breast cancer is an important public health condition, the most prevalent female cancer excluding non-melanoma skin cancers, and the second cause of cancer death in women behind only lung cancer.
According to the American Cancer Society, there were an estimated 316,120 new cases of breast cancer in the United States in 20171. According to data from the National Cancer Institute, there were an estimated 57,960 new cases of breast cancer in Brazil in 20162.
The mortality rate is stable in women aged less than 50 years but has been decreasing in older women, probably due to greater access to information for a part of the population, early diagnosis, and improved treatment modalities3.
Mastectomy is an essential part of the treatment for breast cancer. The reconstruction of the breast helps affected women better preserve their self-esteem and is a right warranted by law in Brazil since 19994. Breast reconstruction does not interfere with the sequential steps in cancer treatment and does not compromise the detection of local recurrence5. Several breast reconstruction techniques have been described, with individualized assessments defining the technique best suited for each patient.
The use of the latissimus dorsi myocutaneous flap (LDMF) to cover defects caused by mastectomy was initially described by Tansini in 1906. However, it was not until 1978 that this flap was used for the reconstruction of the breast by Bostwick6.
Since then, the LDMF technique has become a mainstay in breast plastic surgery. The use of silicone breast implants has helped restore the volume of reconstructed breasts, since the flap itself does not usually provide sufficient soft parts to recreate the breast mound. Since the latissimus dorsi muscle is located in the posterior portion of the trunk, its use for breast reconstruction would usually require a change of decubitus position during the surgical procedure, which would imply an increase in surgical time.
The performance of this surgery in a lateral decubitus position has the advantage of eliminating the change of decubitus position, and as a result, the procedure is shorter. However, anatomical familiarization is required by the surgeon, not only with regard to the structures to be dissected, but also to the position in which the flap is sutured to the receptor site and the suitability for better customization of the remaining breast skin envelope.
The systematization of this procedure in the lateral decubitus position involves several aspects, from the positioning of the patient on the surgical table to a description of the details of dissection and customization of the skin island and envelope. A predetermined sequence is then configured, without change of the decubitus position, to reduce the duration of the surgery without affecting the final result.
OBJECTIVE
The objective of this study was to present a surgical systematization with the description of a series of cases.
METHOD
This was a retrospective primary study conducted using the medical records and photographic documentation of patients who underwent breast reconstruction with the LDMF with a silicone implant in the lateral decubitus position between October 2015 and April 2017. The patients were operated on by the author in both, a private clinic and two public services where he is a plastic surgeon (Hospital Napoleão Laureano-PB and Hospital das Clínicas-PE). The principles of the Declaration of Helsinki, as revised in 2000, and Resolution 196/96 of the National Health Council were duly followed.
Inclusion criteria
Patients who underwent mastectomy due to breast cancer underwent immediate or delayed breast reconstruction using the LDMF with a silicone implant in the single lateral decubitus position between October 2015 and April 2017 and were operated on by the author were included in this study.
Exclusion criteria
Patients who were smokers, had a body mass index greater than or equal to 30 kg/m2, and/or missed the minimum 3-month postoperative follow-up were excluded from this study.
Surgical systematization
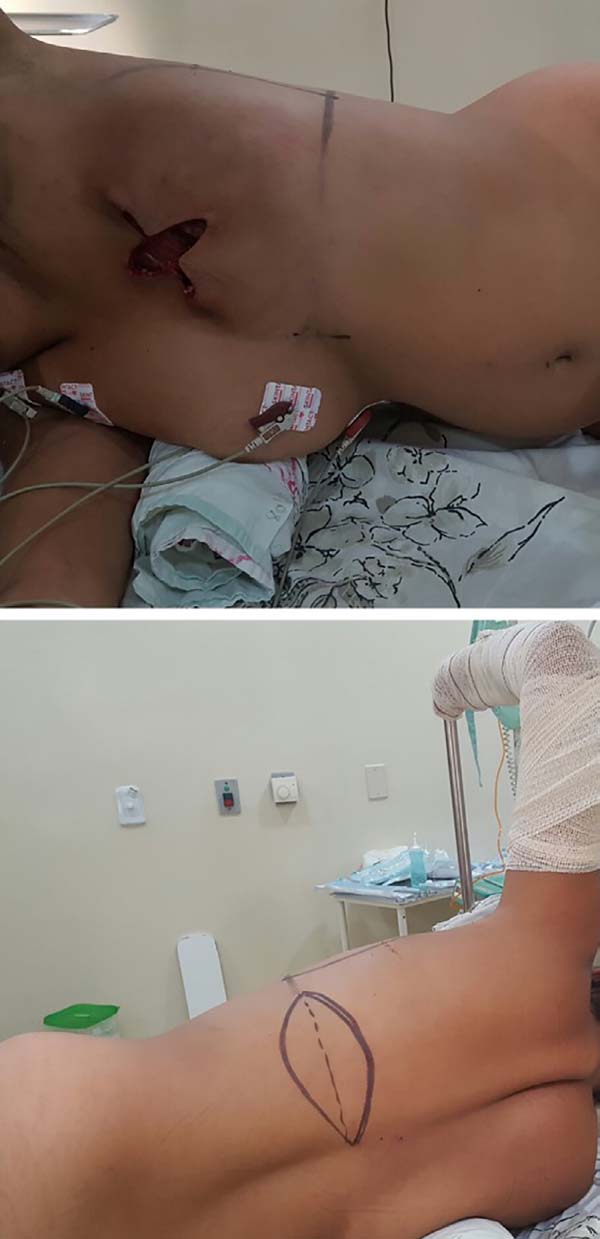
The patient should be positioned in a lateral decubitus position with the ipsilateral limb abducted at 90 degrees and attached to a fabric-coated arc through a bandage. Small cushions must be placed below the chest, between the knees, and to support the head. The hip should be stabilized on the surgical table with a wide bandage or band suitable for a lateral decubitus surgery. Thermal blankets and gradual pneumatic-compression leggings are recommended. The skin island on the dorsum is transversally marked with its axis following the projection of the corresponding inframammary sulcus, which ensures that the final scar is hidden in the posterior loop of the brassiere (Figure 1).
Further orientations of the skin island can be projected based on the location of the chest defect. The dimensions usually measure 17 × 7 cm. If the reconstruction is delayed, surgery is initiated by the removal of the scar prior to the mastectomy. This is followed by the dissection of the receptor site at a level along the fascia of the greater pectoral muscle inferiorly until the projection of the anterior inframammary sulcus, superiorly up to about 3 cm from the clavicle, medially up to 1.5 cm from the sternal border, and laterally up to the projection of the midaxillary line.
In cases of immediate reconstruction, surgery begins with a skin island incision that preserves some of Scarpa’s fascia superiorly and inferiorly to the skin island, and then dissecting to the plane closest to the latissimus dorsi muscle. The dissection should extend medially to the palpation of the vertebral transverse processes, laterally just beyond the border between the large dorsal and anterior serratus muscles, and inferiorly until the identification of the aponeurotic expansion of the latissimus dorsi muscle near the iliac crest.
The superior dissection is performed later with the aid of the Nelson forceps and an electric scalpel. The lateral insertion of the latissimus dorsi muscle is released within the limits already dissected; the muscle is released to the inferior limit and upon lifting the released muscle, it becomes easy to incise its origin along the spine. At this time, the muscle is flat and wide, with its characteristic median thickness.
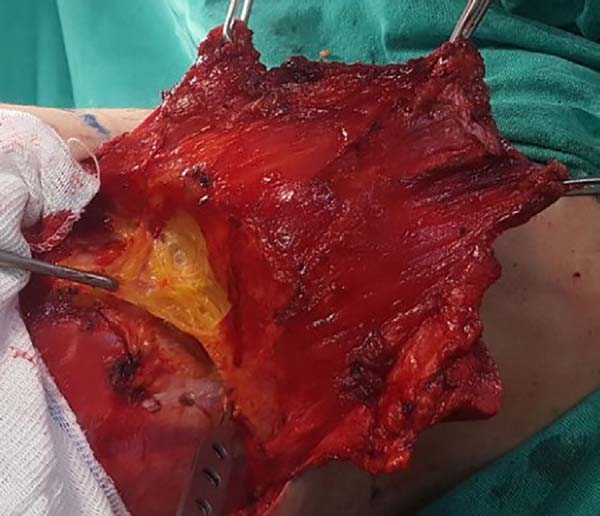
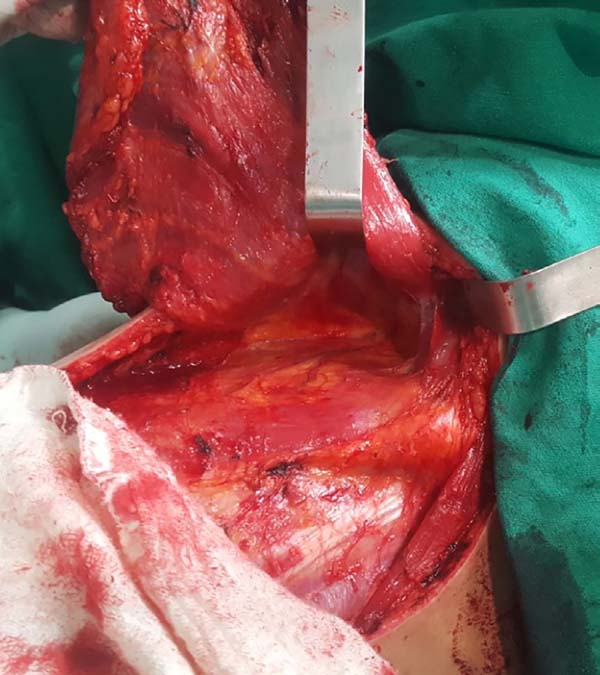
When ascending the dissection, it is important to identify and preserve the donor site structures that are not necessary for the reconstruction, including the fatty fascia behind the latissimus dorsi muscle and the portion of the trapezius muscle that overlaps medially (Figure 2). Once these structures are preserved, the dissection of the latissimus dorsi muscle must be complemented up to near its insertion in the humerus, being its release is optional, with a risk of torsion of the pedicle in certain cases; the advantage, however, is the reduced final volume of tissues in the axillary region.
At this time, the muscle should already be fully released in its inferior and medial portions, requiring only the lateral release, which should be carefully performed near the axillary region, since the pedicle with the thoracodorsal vessels is located in this region. The anatomical detail is that this release should occur over the muscle as the pedicle penetrates through its deep surface, and the dissection in this deep plane may extend a little beyond the vascular anatomical finding called the “goose foot”, which contains branches destined for the anterior serratus muscle and denotes proximity to the thoracodorsal pedicle (Figure 3).
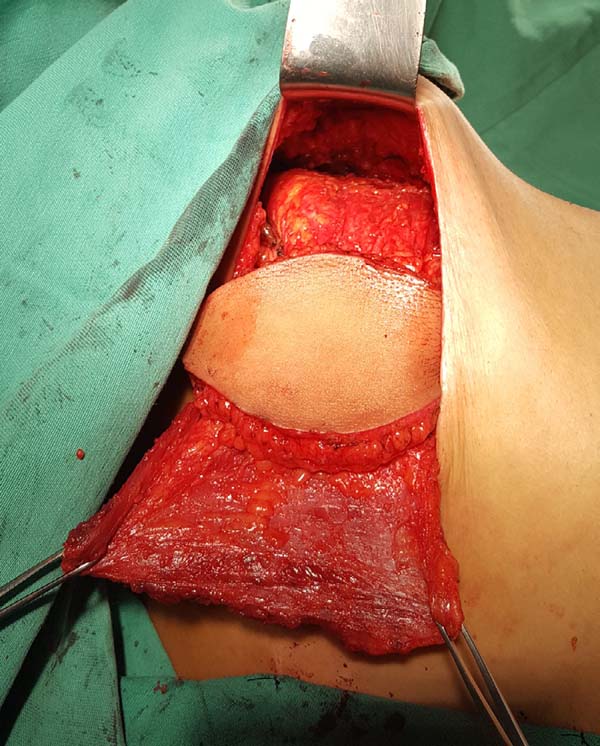
With the muscle dissected, a tunnel of approximately 6 cm width is created through the axillary fascia, approximately at the height of the line of deployment of the axillary hair (Figure 4). Taking care not to twist the muscle, the same is rotated to the receptor site through the previous traction using the Allis forceps (Figure 5).
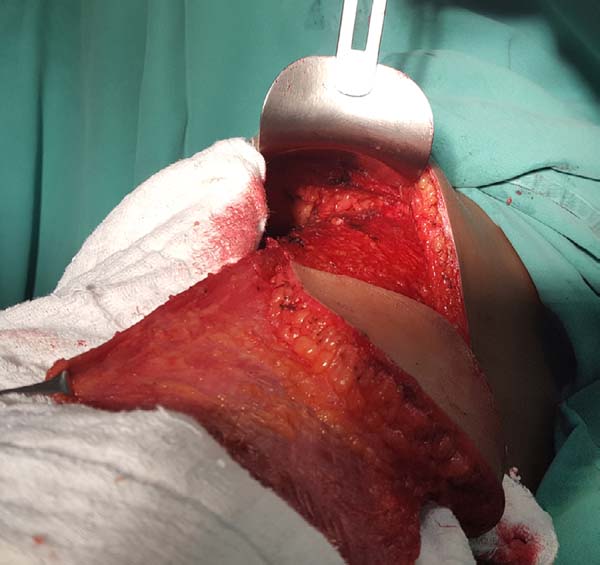
At this moment, the donor site can be synthesized with vicryl 2.0 adhesion sutures and aspiration drainage with 4.8-gauge drains. The pocket for the breast implant is created by attaching the muscle to the limits dissected using nylon 2.0 sutures.
The implant is bathed in an antibiotic solution with cefazolin (1 g) and gentamicin (80 mg) and then positioned in the pocket with a conclusion of the suture. The skin island and the remaining breast skin envelope should be customized so that the neobreast has a slight ptosis, which gives a more natural result (Figure 6). The recipient site should also be drained with a 6.4-gauge suction drain. The drains are removed postoperatively when the daily flow is less than 40 mL.
RESULTS
A total of 29 patients underwent surgery during the study period, with a minimum post-operative follow-up duration of 3 months. The mean age of the patients was 47.22 years, with the youngest patient was aged 28 years and the oldest, 76 years (Table 1).
| Variable | Maximum | Minimum | Average |
|---|---|---|---|
| Age | 76 years | 28 years | 47.22 years |
| Breast Implant | 390 cc | 280 cc | 318 cc |
None of the patients underwent neoadjuvant chemotherapy. With respect to the comorbidities detected, there were 3 patients with systemic hypertension and 1 patient with fibromyalgia (Table 2).
| Variable | Absolute number | Relative rate |
|---|---|---|
| Arterial hypertension | 3 | 10.5% |
| Fibromyalgia | 1 | 3.556% |
The timing of the reconstructive surgery was decided in conjunction with a team of mastologists and included 26 immediate reconstructions and 3 late reconstructions. There were no bilateral reconstructions. The implants used were round format in 28 patients and anatomical in 1 patient. The volumes ranged from 260 cc to 390 cc, with an average of 318 cc. The average length of surgery was 1 h 45 m (Table 1).
The period of hospitalization was uniform with discharges on the second postoperative day. The drains were removed when the daily flow fell below 40 mL, with an average of 10 postoperative days but not exceeding 14 postoperative days.
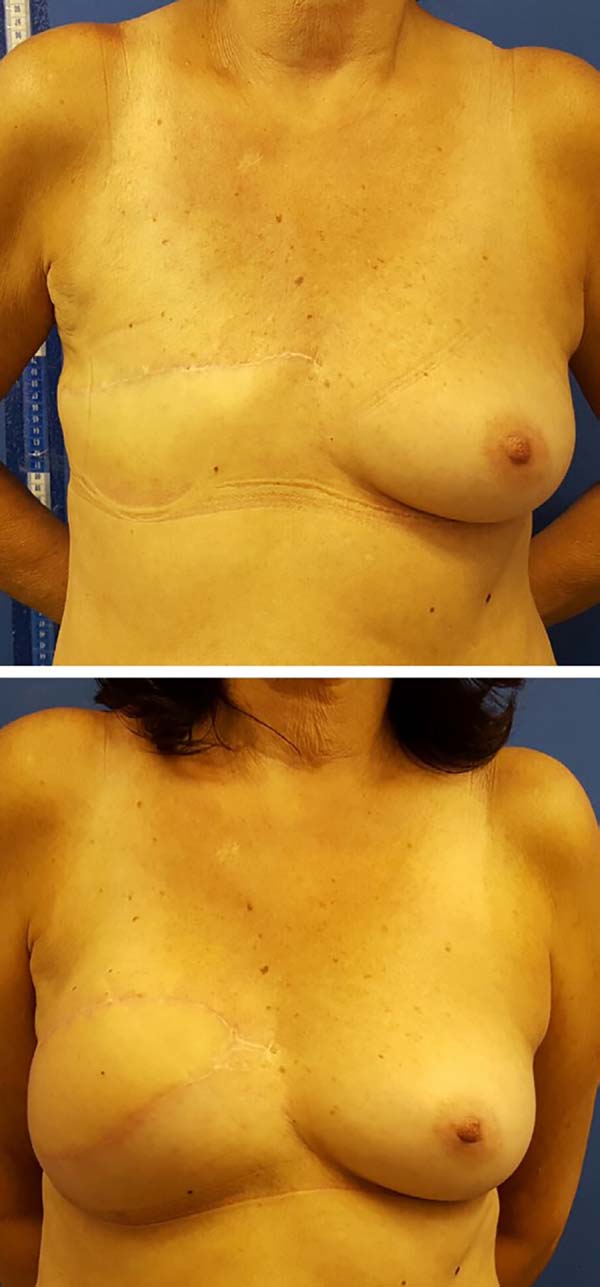
With respect to complications, there was one patient with a seroma in the receptor site (3.5%), who also had a local infection without response to antibiotic therapy, which resulted in extrusion of the breast implant.
One patient (3.5%) had partial necrosis of the skin island of the flap and local cellulitis, with resolution after debridement, antibiotic therapy, and dressings. The cause of necrosis can be attributed to the small cutaneous segment detached from the muscle to better adapt to the receptor site defect. Two patients (7.0%) suffered from the remnant cutaneous envelope of the breast without major clinical repercussions. In these 3 patients, conservative measures were sufficient since the mammary implant in this technique is completely covered by the latissimus dorsi muscle and provides both protection in cases of cutaneous necrosis and avoidance of external contamination.
One patient (3.5%) had extensive local recurrence and the implant was removed at the request of the radiotherapy team according to a therapeutic rescue protocol. One patient (3.5%), submitted to adjuvant radiotherapy, had a Baker III capsular contracture after 18 months of breast reconstruction.
Four patients (14%) were submitted to scar reviews.
On clinical examination, one patient (3.5%) had a seroma in the dorsum that was resolved with aspiration puncture. Imaging examinations were not requested routinely to evaluate this condition.
Three patients (10.5%) displayed functional abduction limitations in the articulation of the ipsilateral shoulder. One of these patients was diagnosed with a winged scapula and forwarded to follow-up with a physiotherapist (Table 3).
| Variable | Absolute number | Index on |
|---|---|---|
| Infection | 2 | 7.0% |
| Cutaneous necrosis | 3 | 10.5% |
| Implant extrusion | 1 | 3.5% |
| Removal of the implant | 1 | 3.5% |
| Seroma in receptor site | 1 | 3.5% |
| Seroma in donor site | 1 | 3.5% |
| Capsular contracture | 1 | 3.5% |
| Scar revision | 4 | 14% |
| Functional joint deficit | 3 | 10.5% |
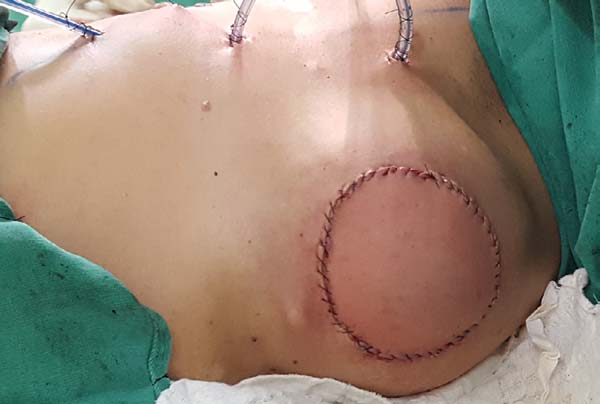
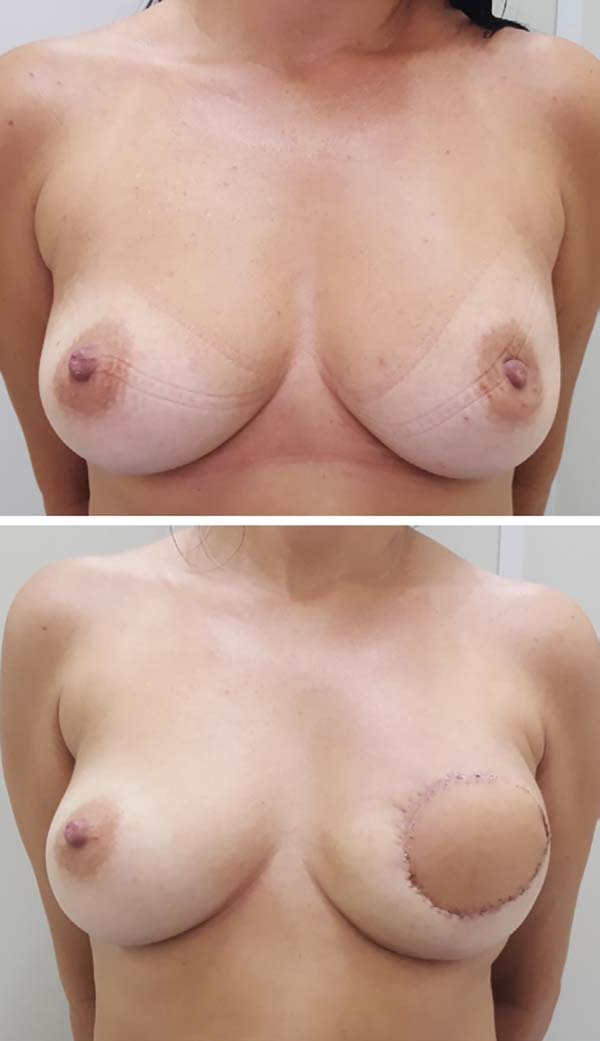
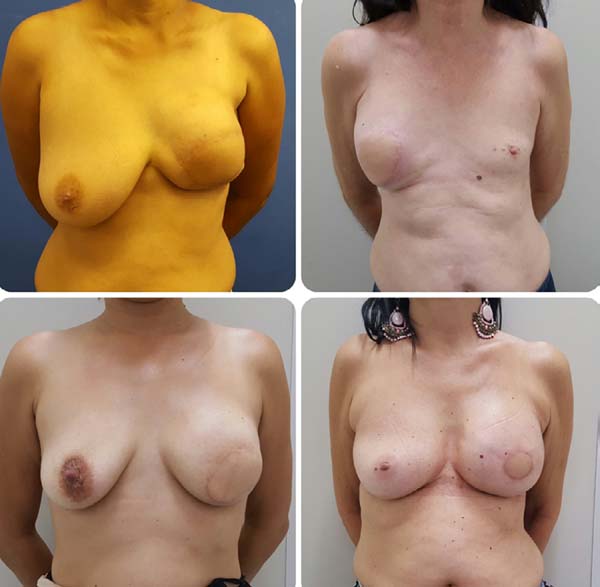
Postoperative results can be seen in Figures 7 to 9.
DISCUSSION
The surgical treatment of breast cancer has evolved over time. In 1894, Halstead described the classical radical mastectomy as the first effective treatment for breast cancer. In 1948 by comparative studies, Patey noted that the preservation of the greater pectoral muscle does not compromise the local control of the tumor, and this led to the term modified radical mastectomy.
To the extent that adjuvant systemic treatment has gained in importance, conservative surgeries of the breast that preserve lymph nodes and segments of the breast parenchyma became possible6, leading to breast reconstruction efforts.
The LDMF was historically used to cover defects in the chest wall, either to cover the defects arising from surgical breast excision or to treat sequelae caused by radiotherapy. The advent of breast implants between the 1960s and 1970s helped to regain the lost breast volume when associated with the LDMF. Techniques that rely exclusively on autologous tissues, such as the use of transverse rectus abdominis muscle (TRAM), microsurgical flaps, and others that use only alloplastic material positioned below the muscles of the anterior thoracic region, have been described by surgeons who aimed for alternatives that could be adapted for each patient.
The LDMF not only weathered time but also gained popularity and is now a mainstay of a large number of plastic surgeons. There are a few anatomical variations of the LDMF, which has reliable caliber pedicle and wide muscle width, which allows full coverage of implants, including the bulky implants. Furthermore, morbidity in the donor area is small, such as a functional deficit in the articulation of the shoulder, seroma formation, or persistent back pain.
The applicability LDMF is vast, and the LDMF may be used for coverage in cases of thin coverage of soft parts in the breast region, irradiated anterior chest wall, previous resection of the pectoralis major, ptotic or small to moderate size contralateral breast, previous unsuccessful reconstruction with exclusive breast implant, previous surgeries in the abdomen, and lack of experience in microsurgical techniques.
The skin island on the dorsum can be ample; however, the primary closure width can be 9 cm with a length of 18 cm, but 17 × 7 cm flaps are suitable. Fat compartments in the back were described in the 1990s by Delay et al.7, with the aim of incorporating a greater quantity of fatty fascia in the flap and reconstructing small to medium sized breasts without the need for implants. However, contour defects and seroma formation in the donor area decreased interest in this technique8.
Moderate amounts of fat fascia can be mobilized to the LDMF for improving both the consistency and outline of the neobreast, allowing for a smoother and less-marked transition in its upper pole, as described by Tavares-Filho et al.9. New generations of implants with high-cohesive gel confer greater stability to the breast shape and better consistency on touch, resulting in good esthetic results in reconstructions performed with the LDMF, even without an excessive fatty fascia harvest10.
Breast reconstruction with the LDMF can be both delayed and immediate (at the same time as the mastectomy). The decubitus for this surgery can vary. For surgeons who choose to harvest the LDMF in a ventral decubitus, there is a need to change the position of the patient at least once in cases of late reconstruction or even twice in cases of immediate reconstruction.
When opting for a lateral decubitus, the position change occurs only once in immediate reconstructions and becomes unnecessary in late reconstructions. Changes in the decubitus position carry the risk of joint, ligament, or even nerve lesions. In addition, they require more surgical material (new sterile fields) and an increase in the total length of surgery with all its potential morbid (infection, thromboembolic events, hypothermia, etc.) and financial implications (surgical room time and anesthetic medications). Specific studies are needed to quantify these variables.
The systematization of the surgical technique allows the surgeon to follow a certain sequence, thus, minimizing the loss of time. Systematization begins with the orientation and training of nursing assistants who will position the patient, making this dynamic moment brief and safe. The surgical procedure is then performed by following the described technique step-by-step; this ensures that the assistant surgeon and the scrub nurse know the exact sequence of presentation and the instruments used.
A disadvantage may be that some surgeons may take time to familiarize themselves with the dissection in a position different from the usual dorsal decubitus, in addition to difficulty in properly customizing the skin island and the remaining breast skin envelope, which pend slightly medially. The end result can be a satisfactory neobreast volume, but with excessive ptosis and/or markedly eccentric positioning of the skin island.
Literature shows that the rate of complications for this surgery is low. A seroma at the donor site is a fairly common complication with studies showing complication rates that vary from 16% to 79%11. The association of adhesion sutures and aspiration drains may account for the low seroma rate observed, which corroborates with the data presented by Cammarota et al. 201612.
Other complications are described with lower rates of incidence: skin infections account for 3.3% of the complications; flap necrosis, 1.3%; operative wound dehiscence, 0.6%; and clinical complications, 3.2%12. A larger sample set would show better approximation of the rate of complications observed with those previously reported.
The rates of capsular contracture are variable and range from 6% to 68%13,14. Longer follow-up duration is necessary to assess the real contracture rate. Adjuvant radiotherapy may be associated with an increase in the contracture rate resulting in actinic capsular contracture15.
An alternative for patients requiring adjuvant radiotherapy is the association of LDMF with a breast tissue expander. A silicone implant would be used only occur after complete tissue expansion and completion of radiotherapy, when it is possible to adjust the format of the neobreast and correct the possible contracture stigmas by capsulotomy or capsulectomy16,17.
Plastic surgery options have evolved; therefore, fewer morbidities are associated with these patients. The use of microsurgical flaps or implants associated with dermal matrices has been reported in recent years in specialized publications as alternatives that promote low morbidity and good results4,18.
However, the use of microsurgical flaps requires specific prolonged surgical training, and dermal matrices are still not feasible in Brazil due to the high associated costs. Consequently, the LDMF in combination with silicone implants continues to be an excellent option for plastic surgeons.
CONCLUSION
The systematization of breast reconstruction using the LDMF combined with silicone breast implants in the lateral decubitus position is a safe alternative for the plastic surgeon, which is a rapid procedure with consistent results. These advantages can be availed when the medical professionals are familiar with the surgery in this patient positioning.
REFERENCES
1. American Cancer Society. How Common Is Breast Cancer? [acesso 2017 Jun 1]. Disponível em: https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html
2. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Câncer de Mama. Estimativa de novos casos. [acesso 2017 Jun 1]. Disponível em: http://www2.inca.gov.br/wps/wcm/connect/tiposdecancer/site/home/mama/cancer_mama++
3. Secretaria de Comunicação Social do Senado Federal. Lei Garante Reconstrução da Mama em Seguida à Retirada de Câncer. [acesso 2017 Jun 1]. Disponível em: http://www12.senado.leg.br/noticias/materias/2013/05/07/lei-garante-reconstrucao-da-mama-em-seguida-a-retirada-de-cancer
4. Macadam SA, Bovill ES, Buchel EW, Lennox PA. Evidence-Based Medicine: Autologous Breast Reconstruction. Plast Reconstr Surg. 2017;139(1):204e-29e. DOI: http://dx.doi.org/10.1097/PRS.0000000000002855
5. Wei FC, Mardini S. Retalhos e cirurgia plástica reconstrutora. Rio de Janeiro: Di Livros; 2012. p. 343-64.
6. Gabka CJ, Bohmert H. Cirurgia plástica e reconstrutiva da mama. 2ª ed. Porto Alegre: Artmed; 2010.
7. Delay E, Gounot N, Bouillot A, Zlatoff P, Rivoire M. Autologous latissimus breast reconstruction: a 3-year clinical experience with 100 patients. Plast Reconstr Surg. 1998;102(5):1461-78. PMID: 9774000 DOI: http://dx.doi.org/10.1097/00006534-199810000-00020
8. Heitmann C, Pelzer M, Kuentscher M, Menke H, Germann G. The extended latissimus dorsi flap revisited. Plast Reconstr Surg. 2003;111(5):1697-701. PMID: 12655217 DOI: http://dx.doi.org/10.1097/01.PRS.0000055444.84307.75
9. Tavares-Filho JM, Franco D, Moreto L, Porchat C, Franco T. Utilização do retalho miocutâneo de grande dorsal, com extensão adiposa, nas reconstruções mamárias: uma opção para preenchimento do polo superior. Rev Bras Cir Plást. 2015;30(3):423-8.
10. D'Alessandro GS, Povedano A, Santos LKIL, Santos RA, Góes JCS. Reconstrução mamária imediata com retalho do músculo grande dorsal e implante de silicone. Rev Bras Cir Plást. 2015;30(2):163-71.
11. Roy MK, Shrotia S, Holcombe C, Webster DJ, Hughes LE, Mansel RE. Complications of latissimus dorsi myocutaneous flap breast reconstruction. Eur J Surg Oncol. 1998;24(3):162-5. PMID: 9630851 DOI: http://dx.doi.org/10.1016/S0748-7983(98)92810-4
12. Cammarota MC, Ribeiro Junior I, Lima RQ, Almeida CM, Moura LG, Daher LMC, et al. Estudo do uso de pontos de adesão para minimizar a formação de seroma após mastectomia com reconstrução imediata. Rev Bras Cir Plást. 2016;31(2):158-65.
13. Gart MS, Smetona JT, Hanwright PJ, Fine NA, Bethke KP, Khan SA, et al. Autologous options for postmastectomy breast reconstruction: a comparison of outcomes based on the American College of Surgeons National Surgical Quality Improvement Program. J Am Coll Surg. 2013;216(2):229-38.
14. Perdikis G, Koonce S, Collis G, Eck D. Latissimus dorsi myocutaneous flap for breast reconstruction: bad rap or good flap? Eplasty. 2011;11e39.
15. McCarthy CM, Pusic AL, Disa JJ, McCormick BL, Montgomery LL, Cordeiro PG. Unilateral postoperative chest wall radiotherapy in bilateral tissue expander/implant reconstruction patients: a prospective outcomes analysis. Plast Reconstr Surg. 2005;116(6):1642-7. PMID: 16267426
16. Losken A, Nicholas CS, Pineel XA, Carlson GW. Outcomes evaluation following breast reconstruction using latissimus dorsi myocutaneous flaps. Ann Plast Surg. 2010;65(1):17-22.
17. Lennox PA, Bovill ES, Macadam AS. Evidence-Based Medicine: Alloplastic Breast Reconstruction. Plast Reconstr Surg. 2017;140(1):94e-108e.
18. Qureshi AA, Broderick KP, Belz J, Funk S, Reaven N, Brandt KE, et al. Uneventful versus Successful Reconstruction and Outcome Pathways in Implant-Based Breast Reconstruction with Acellular Dermal Matrices. Plast Reconstr Surg. 2016;138(2):173e- 83e.
1. Hospital Napoleão Laureano, João Pessoa, PB,
Brazil
2. Hospital das Clínicas, Recife, PE,
Brazil.
3. Sociedade Brasileira de Cirurgia Plástica, São
Paulo, SP, Brazil.
Corresponding author: Igor Chaves Gomes Luna, Rua Reinaldo Tavares de Melo, 142 - Manaíra - João Pessoa, PB, Brazil. Zip Code 58038-300. E-mail: igorluna_med@hotmail.com
Article received: June 10, 2017.
Article accepted: September 18, 2017.
Conflicts of interest: none.





 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter