

Original Article - Year 2016 - Volume 31 -
Breast reconstruction with Becker expanders: an analysis of 116 cases
Reconstrução de mama com expansor de Becker: uma análise de 116 casos
ABSTRACT
INTRODUCTION: Breast expanders/implants were developed in the 1980s as a breast reconstruction alternative to pre-existing techniques. They were created to provide adequate shape and volume to the breast after one-stage surgery and they do not require replacement. To evaluate the long-term results of breast reconstruction performed with Becker expanders.
METHODS: This retrospective study evaluated patients who underwent breast reconstruction with implants/expanders between 2004 and 2012.
RESULTS: A total of 161 expanders were implanted. Major complications (those that led to the removal or replacement of the prosthesis) comprised 22.98% of the enrolled subjects. The expander was removed and replaced by another type of reconstruction implant in 21.74% of the cases. The main reasons for the replacement were capsular contracture (31.43%), patients' desire (31.43%), and implant failure (20%). The surgical alternatives for the replaced expanders included prosthesis (91.43%), latissimus dorsi flap (5.71%), and TRAM flap (2.86%). Only few studies have evaluated the long-term effects of Becker expanders. In this study, the replacement rate was 28.57%. The study questions the use of Becker expanders as an adequate, one-stage reconstruction technique. In spite of a high rate of optimal outcomes in the new surgical procedures (37%), its use is limited because of the high costs. The expanders are still useful for patients with comorbidities or other factors that limit the performance of other surgical procedures.
CONCLUSION: The use of Becker expanders as an alternative for one-stage breast reconstruction technique is very limited. Therefore, these devices should only be used in selected cases.
Keywords: Mammoplasty; Mammoplasty/adverse effects; Breast neoplasms; Prostheses and implants; Devices for tissue expansion.
RESUMO
INTRODUÇÃO: Os implantes-expansores de mama surgiram na década de 80 como uma alternativa de reconstrução de mama às técnicas pré-existentes. Foram criados com a intenção de produzir um bom resultado de forma e volume de mama após uma única operação, não necessitando ser substituído. Avaliar os resultados a longo prazo de reconstruções de mama com uso de expansores de Becker.
MÉTODOS: Estudo retrospectivo de pacientes submetidas à reconstrução de mama com implantes-expansores entre 2004 e 2012.
RESULTADOS: Foram implantados 161 expansores. As complicações maiores (aquelas que levaram à retirada ou troca da prótese) totalizaram 22,98%. O expansor foi retirado e substituído por outro tipo de reconstrução em 21,74% dos casos. As principais razões que levaram à troca foram: contratura capsular (31,43%), vontade da paciente (31,43%); falhas no implante (20%). As opções cirúrgicas substitutivas para os expansores trocados foram prótese (91,43%), grande dorsal (5,71%) e TRAM (2,86%). Existem poucos trabalhos avaliando os expansores de Becker a longo prazo. Neste trabalho a taxa de substituição foi 28,57%. O estudo levanta questionamentos sobre o uso dessa técnica como reconstrução em tempo único. A taxa de cerca de 37% de novos procedimentos cirúrgicos para atingir o resultado ideal torna muito difícil o seu uso em detrimento do alto custo. Os expansores ainda permanecem úteis em casos de pacientes com comorbidades ou qualquer outro fator que limite a realização de outros tempos cirúrgicos.
CONCLUSÃO: O uso de expansores de Becker como alternativa técnica de reconstrução de mama em tempo único é muito limitado. Sua indicação deve ser para casos selecionados.
Palavras-chave: Mamoplastia; Mamoplastia/efeitos adversos; Neoplasias da mama; Próteses e implantes; Dispositivos para expansão de tecidos.
The first study on the use of expanders was published in 1957 by Neumann1 and involved a case of a subauricular defect. Although Neumann's study has demonstrated the feasibility of the procedure, a greater interest in tissue expansion only occurred 20 years later, when Cledomir Radovan developed silicone tissue expanders2. In this context, breast expanders emerged as an alternative to autologous flaps, with the possibility of increasing skin coverage. Therefore, the purpose of the expanders' use in breast reconstruction was to allow the accommodation of a permanent implant, i.e., to provide tissue expansion that would accommodate the volume of a future prosthesis.
Breast expanders/implants were created to provide adequate shape and volume to the breast after one-stage surgery, no longer needing to be replaced by prosthesis because it allows for volume adjustment. These expanders were initially described by Hilton Becker3-5 in the 1980s and could be left as permanent implants, once the desired volume was reached.
Therefore, it became an attractive alternative for the treatment of cases of breast reconstruction, particularly in patients with average volume breasts who underwent bilateral mastectomies, as well as in cases of breast asymmetry and congenital deformities. It presents several advantages5: the procedure is relatively simple and can be performed in a single-stage surgery, with the possibility of breast size adjustment, with reduced scarring, and without deformities and/or morbidity in the donor area, in addition to the shortened recovery period.
Despite these advantages, some studies6-8 have shown that in 30% to 68% of the patients who undergo this procedure, the implants need to be replaced. Therefore, its main advantage of being a one-stage and simple procedure is no longer a real argument.
OBJECTIVE
This study aimed to evaluate the procedures for breast reconstruction with Becker expanders performed in the last eight years and to assess their long-term outcome.
METHODS
This retrospective study involved the review of medical records available between January 1st 2004 and December 31st 2012. The patients enrolled in this study included those who underwent total mastectomy with breast reconstruction (immediate or delayed) using expanders/implants. Those who underwent reconstruction with temporary expanders, local flaps, autologous tissue (TRAM or latissimus dorsi), and those with partial mastectomy were excluded.
The indications for the use of expanders included the patient's desire to undergo a simpler procedure, impossibility of performing other types of reconstruction, and high surgical risk for major surgeries (comorbidities and/or old age).
The data collected included age, postoperative complications, reasons for substitution of the expander and, in cases of expander removal, the new recommended therapy.
All reconstructions were performed by the same surgeon, closely following the same surgical technique.
In all cases included in this study, the Becker expander chosen was the Round BECKERTM 50 Expander/Breast Implant (50% gel) with a remote valve. Expander/implants present a textured surface with two inner shells made with layers of silicone, a gel-filled outer lumen and an adjustable saline-fillable inner lumen. The outer lumen can account to up to 50% of the final volume, and the inner lumen is empty and can be expanded with normal saline until it reaches the desired breast volume. All patients underwent general anesthesia and the expander was placed in the submuscular pocket constructed with pectoralis major muscle flaps (on the upper and lower part), serratus anterior or its fascia (laterally), and the sheath of the rectus abdominis muscle (lower part).
The dome was placed subcutaneously in the axillary line at the height of the breast crease. The first expansion was made intraoperatively, with a volume dependent on the flap conditions at the mastectomy. However, in all cases, the expansion did not exceed 20% of the final volume.
The subsequent expansions were performed on an outpatient basis, after the wound was completely healed, after approximately 2-3 weeks, and every 2-3 weeks until the final volume was reached.
The dome was removed in a second surgical procedure, simultaneously with the reconstruction of the nipple-areola complex and with additional procedures of homograft and contralateral mammoplasty, when necessary.
Patients were monitored weekly during the first month, and monthly for up to 4 months, and as needed until the removal of the medical valve or hospital discharge.
This study followed the criteria defined by the Declaration of Helsinki and its ethical principles, and no conflicts of interest are associated with this publication.
RESULTS
During the study period, 116 patients underwent permanent breast reconstruction using Becker expanders50. In 40 patients, the reconstruction was bilateral, resulting in 161 implanted expanders. The average age was 50.87 years (with a variation between 20 and 85 years).
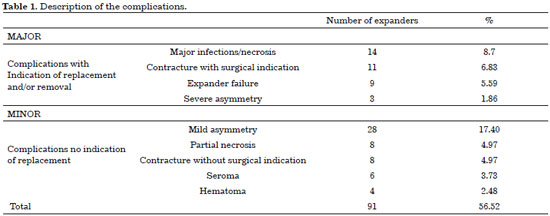
The postoperative complications were divided into minor and major (Figure 1). Major complications were considered those that led to the removal or replacement of the expanders/implants, including major infections or necrosis with exposure of the expander, severe capsular contracture requiring implant replacement, expander failure, or major asymmetries that required the replacement of the expander. These complications totaled 22.98%. Minor complications (33.54%) were defined as seroma, hematoma, partial necrosis, contractures, and minor asymmetries (Table 1).
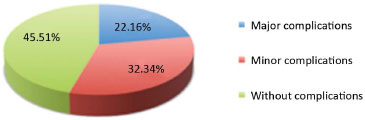
Figure 1. Postoperative complications - Graph showing the main complications. Major complications were defined as those that required replacement and/or removal of the expander.
Of the 28 patients with minor asymmetries, in 16 cases (9.93% of the total number of expanders used), a new surgical procedure was required for replacement of the expander; however, the expander was maintained and resulted in favorable esthetic outcomes. In the remaining cases of minor asymmetry, its correction was possible via fat grafting (Figure 2).
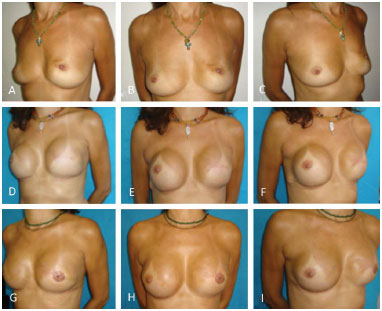
Figure 2. Asymmetry correction - A, B, C: Preoperative left mastectomy. D, E, F: Postoperative period after implantation of the left expander. Significant asymmetry was observed. G, H, I: Late post-operative period after correction of the left breast using fat grafting.
The expander was removed and replaced by another type of reconstruction in 21.74% cases, which comprised 35 expanders (Figure 3). The main reasons that led to the replacement of these expanders were: severe capsular contracture in 11 cases (31.43% of the replaced implants), patient's desire to improve the symmetry or rigidity of the new breast in 11 cases (31.43%), implant failure in 7 cases (20%), severe asymmetry in 3 cases (8.57%), and infection in 3 cases (8.57%) (Figures 4 and 5).
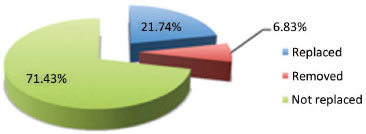
Figure 3. Eight-year evaluation of expanders. Graph showing the longevity of the expander. A rate of replacement of 21.74% was observed.
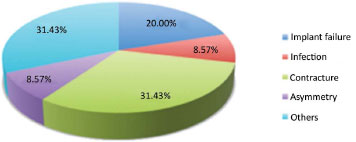
Figure 4. Reasons for replacing expanders. Among all expanders replaced and/or removed (n = 35), contractures and other factors (patient's desire) were the main reasons.
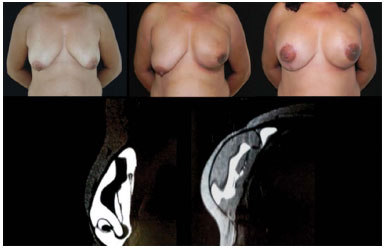
Figure 5. Failure of expanders. MRI showing the "component mixture" (mixture of silicone gel with the filling solution).
Infection led to the removal of the expanders in 8.69% cases (14 implants) and, in most of these cases, it was associated with flap necrosis. In 3 cases, after controlling the infection, the prosthesis was placed without complications.
The surgical alternatives for the 35 replaced expanders were prosthesis (32 patients, 91.43% cases), latissimus dorsi flap (2 patients, 5.71% cases), and TRAM flaps (1 patient, 2.86% cases) (Figure 6).
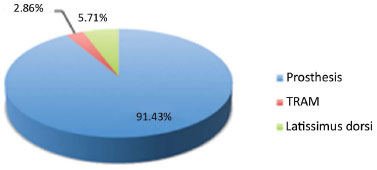
Figure 6. Replacement alternatives. The main alternative for replacement after the removal of the expander was reconstruction with prosthesis (in 91.43% cases). TRAM: Transverse Rectus Abdominis Myocutaneous.
Of the 161 Becker expanders placed, only 25 units (in 18 patients) required a single intervention to complete the reconstruction.
All patients who had their expanders replaced did so in less than 5 years from the reconstruction.
The follow-up period varied between 3 and 60 months.
Figure 7 shows the postoperative outcome in one case.
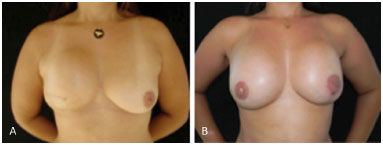
Figure 7. Reconstruction with prosthesis - A: Preoperative period. B: Late postoperative period. Reconstruction with removal of the expander and implantation of bilateral prosthesis.
DISCUSSION
Several studies3,4,9-16 have reported favorable outcomes after the use of expanders/implants in breast reconstruction surgeries, although most of these studies only report initial results because these devices had not been used long enough for a long-term evaluation by the subjects included in the studies. For this reason, to date, the actual longevity of the implant, which was designed to be permanent, has not been evaluated. Recently, Chew et al.7 reported rates of replacement/removal of expanders/implants of more than 68% in a 5-year follow-up. This rate confirms our suspicion that most of these implants were being replaced. In our case-by-case analysis, the replacement rate was 28.57%.
According to our analysis, and in agreement with the results from previous studies, expanders/implants are subject to two main types of complications6,17. The first is related to the usual reasons for removal of implants: infections, capsular contracture, extrusion, and inadequate esthetic results, among others. The second reason is related to problems inherent to the expanders: breakage, leakage, changes in their compartments, dome displacement, and valve obstruction, among others.
We found no studies in the literature on the main reason of the implant failure: rupture of the inner bag with mixture of the components (Figure 8). These cases were discovered by chance during the follow-up on patients with cancer. It is not related to any symptomatology, but causes a lot of psychological discomfort in patients after they acknowledge that they have a ruptured implant.
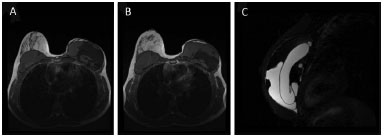
Figure 8. Component mixture of Becker expanders. MRI showing the component mixture with rupture of the internal capsule of the expander in the left breast, with mixture of the saline component with the silicone gel.
This study questions the use of expanders/implants as a strategy for one-stage breast reconstruction, as well as about their longevity. The rate of new surgical procedures for repositioning of the implant of 9.9% combined with the rate of replacement and/or removal of approximately 28% in an 8-year period reinforces the hypothesis by Chew et al.7, that the use of these devices is severely limited because of their high cost.
In addition, nowadays, there is great pressure from health insurers, which restrict the authorization for high-cost procedures and materials, therefore commonly questioning the request for this material, with the justification of conducting a one-stage reconstruction.
In our analysis, of the 161 expanders implanted, only 25 implants (in 18 patients) underwent a one-stage intervention to achieve adequate esthetic results, and those units did not need any type of adjustment except the removal of the valve.
Fernandes et al.18 evaluated the costs and resources associated with breast reconstruction surgery by comparing temporary expanders with permanent expanders/implants. For two-stage reconstruction, the medical fees were the main component of the final cost of treatment, corresponding to 28% of the total, followed by the temporary expander and the permanent implant used (13%). In one-stage reconstructions, the cost of the expanders/implants used corresponded to 46% of the total, followed by medical fees (19%). The budget impact analysis indicated significant savings from the perspective of the payer for reconstructions using Becker expanders, demonstrating that the higher unit costs of the expander would be offset by clear economic benefits, particularly with the lower utilization of funds due to the need to perform one-stage surgeries.
However, in 2012, Eriksen et al.8 compared one-stage with two-stage reconstructions and observed that the patients who underwent two-stage reconstructions had a lower surgical revision rate compared with those who underwent one-stage reconstructions, demonstrating that this technique is limited in achieving the desirable result as the single option.
The removal of the Becker expander and its replacement by breast implants does not cause any additional problems compared with the use of other models of expanders or implants6. The evaluation of patients with replaced expanders revealed that they also achieved favorable outcomes. The ultimate goal of all breast reconstructions should be to achieve the best possible aesthetic result, with the lowest number of surgeries. However, psychological stress and frustration caused by a new preoperative and postoperative routine and another period of absence from work in patients who believed they were undergoing a single procedure cannot be overlooked.
The need to perform a second procedure using temporary expanders or even prostheses is well accepted by patients because they have understood that this is part of a process aimed at achieving the best possible aesthetic outcome19-21.
It is important to note that Becker expanders continue to be a useful tool in some cases of breast reconstruction22. The evaluation of this study results allowed for a redirection of the indications concerning the use of these devices by our team. We restricted this technique to patients with comorbidities or other factors that limit the performance of other surgical procedures. In such cases, we assume the risk of proceeding concomitantly with mastectomy and symmetrization. The possibility of adjustment of the implant size becomes a favorable factor in achieving symmetry.
CONCLUSION
The use of Becker expanders as an alternative, one-stage technique for breast reconstruction is very limited. The increased need for replacement (approximately 28%) and/or associated corrections (approximately 85%) makes it costly and less efficient when compared with other, already established techniques. Therefore, these devices should be used in selected cases.
REFERENCES
1. Neumann CG. The expansion of an area of skin by progressive distention of a subcutaneous balloon; use of the method for securing skin for subtotal reconstruction of the ear. Plast Reconstr Surg (1946). 1957;19(2):124-30. DOI: http://dx.doi.org/10.1097/00006534-195702000-00004
2. Randovan C. Breast reconstruction after mastectomy using the temporary expander. Plast Reconstr Surg. 1982;69(2):195-208. DOI: http://dx.doi.org/10.1097/00006534-198202000-00001
3. Becker H. Breast reconstruction using an inflatable breast implant with detachable reservoir. Plast Reconstr Surg. 1984;73(4):678-83. PMID: 6709750 DOI: http://dx.doi.org/10.1097/00006534-198404000-00031
4. Becker H. The expandable mammary implant. Plast Reconstr Surg. 1987;79(4):631-7. PMID: 3823256 DOI: http://dx.doi.org/10.1097/00006534-198704000-00023
5. Becker H. The permanent tissue expander. Clin Plast Surg. 1987;14(3):519-27. PMID: 3608362
6. Farace F, Faenza M, Bulla A, Rubino C, Campus GV. Is mammary reconstruction with the anatomical Becker expander a simple procedure? Complications and hidden problems leading to secondary surgical procedures: a follow-up study. J Plast Reconstr Aesthet Surg. 2013;66(6):741-6. DOI: http://dx.doi.org/10.1016/j.bjps.2013.02.004
7. Chew BK, Yip C, Malyon AD. Becker expander implants: truly a long term single stage reconstruction? J Plast Reconstr Aesthet Surg. 2010;63(8):1300-4. DOI: http://dx.doi.org/10.1016/j.bjps.2009.07.004
8. Eriksen C, Lindgren EN, Frisell J, Stark B. A prospective randomized study comparing two different expander approaches in implant-based breast reconstruction: one stage versus two stages. Plast Reconstr Surg. 2012;130(2):254e-64e. DOI: http://dx.doi.org/10.1097/PRS.0b013e3182589ba6
9. Gibney J. Use of a permanent tissue expander for breast reconstruction. Plast Reconstr Surg. 1989;84(4):607-17. DOI: http://dx.doi.org/10.1097/00006534-198984040-00008
10. Berry MG, al-Mufti RA, Jenkinson AD, Denton S, Sullivan M, Vaus A, et al. An audit of outcome including patient satisfaction with immediate breast reconstruction performed by breast surgeons. Ann R Coll Surg Engl. 1998;80(3):173-7.
11. Camilleri IG, Malata CM, Stavrianos S, McLean NR. A review of 120 Becker permanent tissue expanders in reconstruction of the breast. Br J Plast Surg. 1996;49(6):346-51. PMID: 8881779 DOI: http://dx.doi.org/10.1016/S0007-1226(96)90001-6
12. Woods JE, Mangan MA. Breast reconstruction with tissue expanders: obtaining an optimal result. Ann Plast Surg. 1992;28(4):390-6. DOI: http://dx.doi.org/10.1097/00000637-199204000-00019
13. Scuderi N, Alfano C, Campus GV, Rubino C, Chiummariello S, Puddu A, et al. Multicenter study on breast reconstruction outcome using Becker implants. Aesthetic Plast Surg. 2011;35(1):66-72. DOI: http://dx.doi.org/10.1007/s00266-010-9559-x
14. Goh SC, Thorne AL, Williams G, Laws SA, Rainsbury RM. Breast reconstruction using permanent Becker expander implants: an 18 year experience. Breast. 2012;21(6):764-8. DOI: http://dx.doi.org/10.1016/j.breast.2012.03.007
15. Hsieh F, Shah A, Malata CM. Experience with the Mentor Contour Profile Becker-35 expandable implants in reconstructive breast surgery. J Plast Reconstr Aesthet Surg. 2010;63(7):1124-30. DOI: http://dx.doi.org/10.1016/j.bjps.2009.05.043
16. Munhoz AM, Aldrighi C, Montag E, Arruda EG, Aldrighi JM, Filassi JR, et al. Periareolar skin-sparing mastectomy and latissimus dorsi flap with biodimensional expander implant reconstruction: surgical planning, outcome, and complications. Plast Reconstr Surg. 2007;119(6):1637-49. DOI: http://dx.doi.org/10.1097/01.prs.0000246406.68739.e4
17. Farace F, Faenza M, Sanna M, Campus GV, Rubino C. Filling-port complications in Becker expanders. Plast Reconstr Surg. 2012;129(2):386e-387e. DOI: http://dx.doi.org/10.1097/PRS.0b013e31823aef0c
18. Fernandes RA, Takemoto MLS, Matthes AGZ, Cammarota MC, Arcoverde MM, Fraga MFP. Utilização de recursos e custos associados às cirurgias de reconstrução de mama pós-mastectomia com expansores temporários versus expansores permanentes sob a perspectiva do Sistema de Saúde Suplementar. J Bras Econ Saúde. 2011;3(2):211-6.
19. Settembrini F, Bonomi S, Gregorelli C. Patient selection and aesthetic outcomes in one-stage versus two-stage implant breast reconstruction. Plast Reconstr Surg. 2012;129(2):364e-366e. PMID: 22286461 DOI: http://dx.doi.org/10.1097/PRS.0b013e318230be38
20. Strock LL. Two-stage expander implant reconstruction: recent experience. Plast Reconstr Surg. 2009;124(5):1429-36. PMID: 19342993 DOI: http://dx.doi.org/10.1097/PRS.0b013e31818b89da
21. Castelló JR, Garro L, Nájera A, Mirelis E, Sánchez-Olaso A, Barros J. Immediate breast reconstruction in two stages using anatomical tissue expansion. Scand J Plast Reconstr Surg Hand Surg. 2000;34(2):167-71. PMID: 10900634 DOI: http://dx.doi.org/10.1080/02844310050160042
22. Sindali K, Davis M, Mughal M, Orkar KS. The natural history of Becker expandable breast implants: a single-center 10-year experience. Plast Reconstr Surg. 2013;132(3):345e-351e. PMID: 23985646 DOI: http://dx.doi.org/10.1097/PRS.0b013e31829ace7a
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil.
2. Hospital Daher Lago Sul, Brasília, DF, Brazil
Institution: Hospital Daher Lago Sul, Brasília, DF, Brazil.
Corresponding author:
Rafael Quaresma de Lima
SHIS QI 7 conjunto F, Área Especial, Lago Sul
Brasília, DF, Brazil Zip Code 717615-570
E-mail: rafaquaresma@gmail.com
Article received: January 19, 2015.
Article accepted: November 20, 2015.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter