

Original Article - Year 2015 - Volume 30 -
Wound management of partial-thickness skin-graft donor areas with a collagen and alginate dressing (Fibracol®): findings in 35 patients
Curativo da área doadora de enxerto de pele parcial com curativo de colágeno e alginato (Fibracol®): uma experiência de 35 pacientes
ABSTRACT
INTRODUCTION: The objective of this study was to report our findings with a collagen and alginate dressing (Fibracol®) used to cover donor areas of partial-thickness skin grafts.
METHOD: We retrospectively evaluated the medical records of 35 patients in whom Fibracol® was used on donor areas. The routine used to manage the donor area is as follows: The area is covered with one or more units of Fibracol®, followed by application of a waterproof film. After three or four days, the dressing is removed and the area cleaned gently with saline and gauze; the area is dressed again if necessary. The mean patient age was 25.52 years (range, 1-65 years).
RESULTS: The thigh was used as the donor area in 29 patients, the arm in 2, the leg in 4, and the trunk in 3. Two patients had more than one donor area. The mean time needed for epithelization was 4.51 days (range, 3-8 days). The Pearson correlation coefficient value correlating age and time of epithelization was -0.0755; p = 0.6685. None of the patients experienced an infection in the donor area. The ideal dressing for the donor area of split-thickness skin grafts would have multiple characteristics including low price, good patient comfort, low infection rate, and a short epithelization period.
CONCLUSION: The authors report a positive experience with the use of Fibracol® in 35 patients over a period of 22 months. The mean epithelization period was 4.51 days, shorter than that in the majority of published studies, and had no correlation with the age of the patient.
Keywords: Burns; Grafts; Dressing.
RESUMO
INTRODUÇÃO: O objetivo deste estudo é relatar a nossa experiência com curativo de colágeno e alginato (Fibracol®) para cobrir áreas doadoras de enxerto de pele de espessura parcial.
MÉTODO: Estudamos, retrospectivamente, 35 prontuários de pacientes que utilizaram o Fibracol® em áreas doadoras. Nossa rotina para cobrir a área doadora é a seguinte: cobertura da área com uma ou mais unidades de Fibracol® e, em seguida, com uma película à prova de água. Depois de três ou quatro dias, remover o curativo, limpar delicadamente com soro fisiológico e gaze e, quando julgava-se necessário, cobria-se novamente. A idade média foi de 25,52 anos (1-65).
RESULTADOS: A coxa foi usada como área doadora em 29 pacientes, o braço em 2, a perna em 4 e tronco em 3 (2 pacientes tiveram mais de uma área doadora). O tempo médio necessário para epitelização foi de 4,51 dias (3-8). O valor de R do coeficiente de correlação de Pearson correlacionando a idade e tempo de epitelização foi -0,0755, com p = 0,6685. Nenhum dos pacientes teve infecção na área doadora. O curativo ideal para a área doadora do enxerto de pele de espessura parcial teria muitas características, incluindo: preço baixo, bom conforto do paciente, baixa taxa de infecção, período curto de tempo para epitelização, etc.
CONCLUSÃO: Os autores relatam uma boa experiência usando Fibracol® em 35 pacientes, durante um período de 22 meses. O tempo para epitelização foi de 4,51 dias, mais curta do que a maioria dos trabalhos publicados, e não tinha correlação com a idade do paciente.
Palavras-chave: Queimaduras; Enxertos; Curativos.
The treatment of burn patients usually includes the application of partial-thickness skin grafts1. In this procedure, the physician creates a donor site, which can cause more postoperative morbidity than the burn itself2. There is no consensus on the most suitable dressing for this area3,4.
OBJECTIVE
The objective of this study was to report our 2-year experience with the use of a collagen and alginate dressing (Fibracol®) to cover donor areas of partial-thickness skin grafts in burn patients.
METHOD
We retrospectively studied the medical records of 35 burn patients in whom Fibracol® was used to cover donor areas of partial-thickness skin grafts. All the burn patients were operated on by the same surgeon (LSM) between May 2012 and March 2014.
The routine used to manage the donor area is as follows: The area is covered with one or more units of Fibracol®, followed by application of a waterproof film. After three or four days, the dressing is removed and the area cleaned gently with saline and gauze; if deemed necessary, the area is again covered with a dressing.
RESULT
Of the 35 patients, 26 were men. The mean age was 25.52 years (range, 1-65 years). The thigh was used as the donor area in 29 patients, the arm in 2 patients, the leg in 4 patients, and the trunk in 3 patients. Two patients had more than one donor area.
The mean time needed for epithelization was 4.51 days3-8.
The R Pearson correlation coefficient between age and time of epithelization was -0.0755, with p = 0.6685.
None of the patients experienced an infection in the donor area.
Figures 1 to 4 illustrate the care of a donor area on the arm of a patient. Figures 5 to 8 illustrate the progression of the donor area of the thigh of another patient.
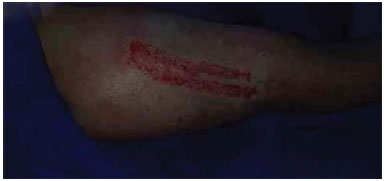
Figure 1. Donor area immediately after removal of the partial-thickness skin graft.
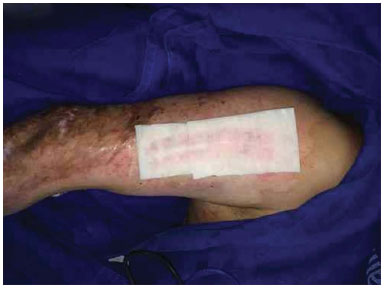
Figure 2. Donor area covered with Fibracol.
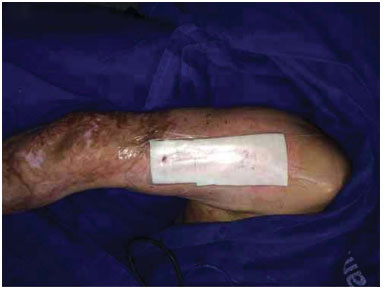
Figure 3. Donor area covered with Fibracol (c) and the film.
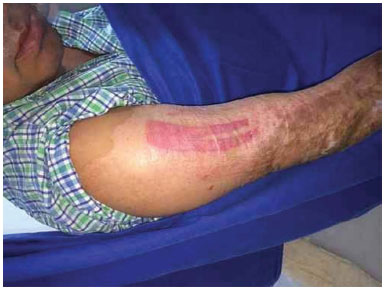
Figure 4. Donor area after 4 days of dressing.
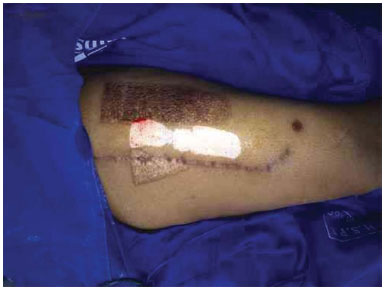
Figure 5. Donor area after removal of the partial-thickness skin graft.
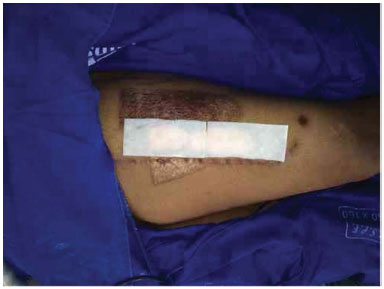
Figure 6. Donor area covered with Fibracol (c).
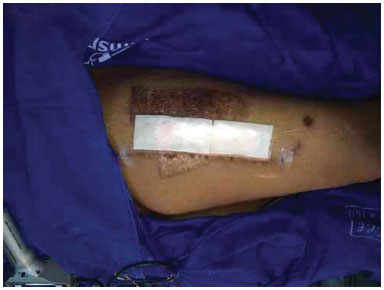
Figure 7. Donor area covered with Fibracol (c) and the film.
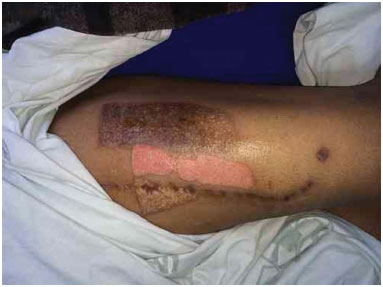
Figure 8. Donor area after 5 days of dressing.
DISCUSSION
The ideal dressing for the donor area of partial-thickness skin grafts would have multiple characteristics including low price, good patient comfort, low infection rate, and a short period for epithelization. Normally, non-adherent dressings are used, but adherent dressings5 and other materials including honey6 have been used to achieve these objectives.
Fibracol® is composed of collagen (90%) and alginate (10%). It combines the structural support of collagen and the gel-forming properties of alginate in a soft, comfortable, and absorbent dressing. A few studies have been published on the subject.
Higgins et al., in 2012, compared an alginate dressing (KaltostatR®) with a polyurethane dressing (AllevynTM) and concluded that KaltostatR® was superior7.
Kaiser et al., in 2013, evaluated an alginate dressing with polyurethane film and paraffin gauze in a well-designed prospective study. They found no difference between the two dressings in terms of pain and time of healing8. An explanation for the longer healing times in their study (mean of 18.1 days for the alginate group and
15.4 days for the paraffin gauze group) in comparison with our patients (mean, 4.5 days) is probably owing to thicker skin grafts. Uraloglu et al., in 2014, published an experimental study that showed rapid and good-quality epithelialization with alginate9.
CONCLUSION
The authors report a positive experience with the use of Fibracol® in 35 patients over 22 months. The epithelization time was 4.51 days, shorter than that in most published studies, and had no correlation with the age of the patient.
REFERENCES
1. Orgill DP, Ogawa R. Current methods of burn reconstruction. Plast Reconstr Surg. 2013;131(5):827e-36e. PMID: 23629122 DOI: http://dx.doi.org/10.1097/PRS.0b013e31828e2138
2. Läuchli S, Hafner J, Ostheeren S, Mayer D, Barysch MJ, French LE. Management of split-thickness skin graft donor sites: a randomized controlled trial of calcium alginate versus polyurethane film dressing. Dermatology. 2013;227(4):361-6. PMID: 24281776 DOI:http://dx.doi.org/10.1159/000356122
3. Karlsson M, Lindgren M, Jarnhed-Andersson I, Tarpila E. Dressing the split-thickness skin graft donor site: a randomized clinical trial. Adv Skin Wound Care. 2014;27(1):20-5. DOI:http://dx.doi.org/10.1097/01.ASW.0000437786.92529.22
4. Assadian O, Arnoldo B, Purdue G, Burris A, Skrinjar E, Duschek N, et al. A prospective, randomised study of a novel transforming methacrylate dressing compared with a silver-containing sodium carboxymethylcellulose dressing on partial-thickness skin graft donor sites in burn patients. Int Wound J. 2015;12(3):351-6. DOI: http://dx.doi.org/10.1111/iwj.12136
5. Kaartinen IS, Kuokkanen HO. Suprathel(®) causes less bleeding and scarring than Mepilex(®) Transfer in the treatment of donor sites of split-thickness skin grafts. J Plast Surg Hand Surg. 2011;45(4-5):200-3. PMID: 22150140 DOI: http://dx.doi.org/10.3109/2000656X.2011.583515
6. Misirlioglu A, Eroglu S, Karacaoglan N, Akan M, Akoz T, Yildirim S. Use of honey as an adjunct in the healing of split-thickness skin graft donor site. Dermatol Surg. 2003;29(2):168-72.
7. Higgins L, Wasiak J, Spinks A, Cleland H. Split-thickness skin graft donor site management: a randomized controlled trial comparing polyurethane with calcium alginate dressings. Int Wound J. 2012;9(2):126-31.
8. Kaiser D, Hafner J, Mayer D, French LE, Läuchli S. Alginate dressing and polyurethane film versus paraffin gauze in the treatment of split-thickness skin graft donor sites: a randomized controlled pilot study. Adv Skin Wound Care. 2013;26(2):67-73.
9. Uraloğlu M, Livaoğlu M, Agdoğan Ö, Mungan S, Alhan E, Karaçal N. An evaluation of five different dressing materials on split-thickness skin graft donor site and full-thickness cutaneous wounds: an experimental study. Int Wound J. 2014;11(1):85-92
1. Sociedadede Brasileira de Cirurgia Plática, São Paulo, SP, Brazil
2. Hospital do Servidor Público Estadual de São Paulo, São Paulo, SP, Brazil
3. Instituto do Câncer do Estado de São Paulo, São Paulo, SP, Brazil
Institution: Hospital do Servidor Público Estadual de São Paulo, São Paulo, SP, Brazil.
Corresponding author:
Lincoln Saito Millan
Rua Joaquim Floriano, 466, Itaim Bibi
São Paulo, SP, Brazil Zip code 05403908
E-mail: lincolnsaito@gmail.com
Article received: July 14, 2014.
Article accepted: March 15, 2015.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter