

Original Article - Year 2015 - Volume 30 -
Influence of postoperative complications on the failure of immediate breast reconstruction with silicone implants
Influência das complicações pós-operatórias no insucesso da reconstrução de mama imediata com implante de silicone
ABSTRACT
INTRODUCTION: Immediate breast reconstruction with silicone implants following mastectomy is a simple method, but can develop complications culminating in implant removal. The aim of this study was to analyze postoperative complications and evaluate their correlation with implant removal.
METHOD: In a period of 4 years, 323 cases of immediate breast reconstruction with silicone implants following total mastectomy were retrospectively studied in the Institut Gustave-Roussy, France.
RESULTS: The most frequent complication was lymphocele (34.9%), followed by cutaneous necrosis (22.9%), infection (19.3%), and hematoma (13.3%). Implant removal was more frequent when a surgical complication occurred, and even more frequent when there was more than one type of complication. The most frequent complication leading to implant removal was infection (75.0%). The expander was the implant that had the highest correlation with implant removal. The use of implants with a volume greater than 300 ml was associated with a significantly higher risk of implant removal.
CONCLUSIONS: 1) The presence of postoperative complications was a risk factor for implant removal. 2) The risk of removal was higher when more than one complication was present. 3) Infection was the main type of complication associated with implant removal. 4) The expander presented a higher risk of complications and implant removal. 5) The use of implants with a volume greater than 300 ml had a greater risk of need for removal.
Keywords: Prosthesis; Mammoplasty; Postoperative complications; Neoplasm; Reconstruction; Breast.
RESUMO
INTRODUÇÃO: A reconstrução da mama imediata pós-mastectomia, com implante de silicone é um método simples, porém, pode evoluir com complicações e remoção do implante. O objetivo do estudo foi analisar as complicações pós-operatórias e buscar relação entre estas e a remoção do implante.
MÉTODO: No período de 4 anos, foram estudados retrospectivamente 323 casos de reconstrução de mama imediata com implante de silicone após mastectomia total realizados no Institut Gustave-Roussy, França.
RESULTADOS: A complicação mais frequente foi a linfocele (34,9%), seguida da necrose cutânea com 22,9%, da infecção com 19,3% e do hematoma, com 13,3% dos casos. A remoção do implante foi mais frequente quando ocorreu algum tipo de complicação cirúrgica e maior quando ocorreu mais de um tipo de complicação. A complicação mais frequente nos casos de remoção do implante foi a infecção (75,0%). O expansor foi o implante que mais teve relação com remoção do implante. O uso de implantes de volume acima de 300 ml teve significativamente mais risco de remoção do implante.
CONCLUSÕES: 1) A presença de complicação pós-operatória foi fator de risco para a remoção do implante. 2) O risco de remoção foi maior na presença de mais de um tipo de complicação 3) A infecção foi o principal tipo de complicação que se relacionou com a remoção 4) O expansor apresentou maior risco de complicações e de remoção do implante. 5) A utilização de implantes de volume maior do que 300 ml apresentou maior risco de remoção.
Palavras-chave: Prótese; Mamoplastia; Complicações Pós-operatórias; Neoplasia; Reconstrução; Mama.
Breast cancer is an important public health problem. It has an increasing global incidence, and constitutes the primary cause of death from cancer among women in Western industrialized countries, as well as in some industrialized regions of developing countries1.
This group of factors, along with the need to assure a good quality of life for patients affected by this disease, enabled breast reconstruction following mastectomy to become a part of the oncological therapy protocol2.
Ever since an implant made of alloplastic material was used for breast reconstruction for aesthetic and rehabilitative purposes, there have been references to early or late surgical complications3. These are directly or indirectly associated with the insertion of synthetic material4.
The occurrence of postoperative complications associated with this type of breast reconstruction has been described by several authors5,6. However, there are few specific analyses in the literature correlating the occurrence of postoperative complications and the need for implant removal.
This study aimed to analyze and correlate the postoperative complications of immediate breast reconstruction (IBR) with silicone implants and their characteristics, with the need for implant removal.
METHODS
In a period of 4 years, a group of 306 female patients between 23 and 74 years old (average, 46 years), diagnosed with breast cancer, received treatment in the Institut Gustave-Roussy (IGR), Villejuif, France.
In 17 (5.6%) patients, tumors were bilateral, resulting in 323 mastectomies, followed by IBR.
The relevant data for the study were prospectively obtained and selected from the digitized clinical records of each patient. The following information was collected: age, TNM classification, presence and type of treatment prior to IBR, histologic type of tumor, axillary lymph node involvement, presence of axillary lymphadenectomy, type of implant utilized, volume of implant, adjuvant treatment following breast reconstruction, presence and types of postoperative complications, and whether or not an implant was removed.
The term "postoperative complications" has been used to refer to those directly resulting from the surgical procedure itself, within 3 months postoperatively.
The postoperative complications identified were infection, hematoma, cutaneous necrosis, lymphocele drained by aspiration, and inflammation.
Infection was characterized by the presence of fever associated with leukocytosis, as well as a positive culture and identification of the pathogenic organism from scar exudate or surgical wounds.
Inflammation of the cutaneous graft was considered milder than infection, with self-limiting clinical symptoms, and without laboratory findings.
A lymphocele was characterized by the presence of serous or serosanguinous collections, comprised mainly of lymph originating from inflamed lymph ducts, exudates from the open area, and local drainage problems.
Cutaneous graft necrosis was diagnosed in the postoperative period by curative exchange, and was characterized by a darkened and ischemic area.
The term "complications" has been used to refer to late issues associated with the reconstruction, and may also result in the need for implant removal, due to a class III or IV7 periprosthetic capsular contracture, for instance, or to late infection.
The need for implant removal characterizes reconstruction failure.
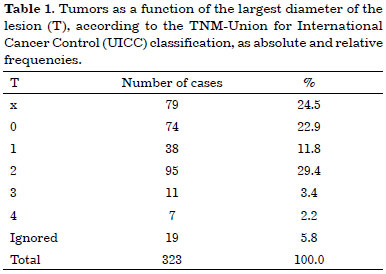
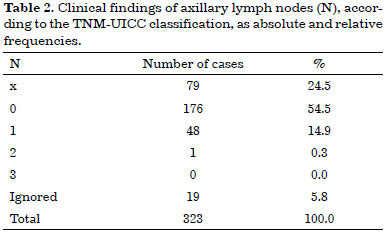
Tables 1, 2, and 3 list the patients according to tumor size as a function of the largest diameter, clinical findings of axillary lymph nodes, and presence of metastasis, respectively.
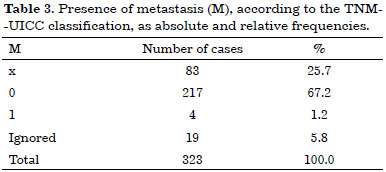
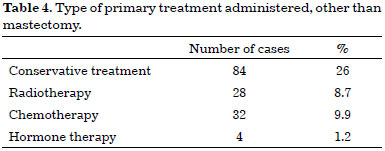
Mastectomy was the primary treatment for breast cancer in 212 cases (65.6%). Another type of treatment was performed in 111 cases (34.4%) (Table 4).
Histological analysis of the surgical tissues revealed carcinoma in 300 cases (92.9%), and another histological type of tumor in the remaining 23 (7.1%). In situ lobular carcinoma was observed in 0.9% of cases, in situ lobular carcinoma with microinvasion in 3.1%, in situ ductal carcinoma in 12.7%, in situ ductal carcinoma with microinvasion in 16.7%, invasive ductal carcinoma in 59.5%, and other types of carcinoma in 7.1%.
Axillary dissection was performed in 243 cases (100.0%), followed by a histological analysis of the axillary lymph nodes; a positive result was noted in 103 cases (42.4%) and a negative result was noted in 140 cases (57.6%).
Surgical technique
The surgeries were consecutive and performed by the team of the Breast and Reconstructive Surgery Service of the IGR Surgery Department.
The procedure performed was a modified radical mastectomy, with preservation of the pectoralis minor muscles and the medial pectoral nerve, followed by axillary lymphadenectomy.
Breast reconstruction was performed following mastectomy during the same surgical session, with inclusion of the final silicone implant8, or with the insertion of a tissue expander with the filling valve placed in the lateral thorax9. The location occupied by the implant was always separated from the mastectomy site and the axillary drainage areas, and received independent aspiration drainage.
The drains were removed as soon as the drainage was less than 50 ml in a 24-hour period.
The different types of breast implants primarily used are shown in Table 5.
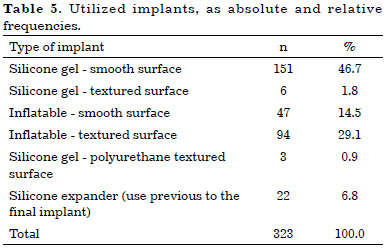
The dimensions of the final implants used varied in regards to volume, ranging from 80 to 680 ml, and were divided among 4 groups: group 1 with implants up to 150 ml (7.9%), group 2 between 151 and 300 ml (58.4%), group 3 between 301 and 450 ml (29.2%), and group 4 above 450 ml (4.3%).
The study was approved by the Ethics Committee of the IGR, Villejuif, France.
Statistical Methods
In order to evaluate the possible correlations between the analyzed variables, two contingency tables with two rows and two columns were created, using the chi-square test, represented by the symbol χ2.
When the use of the chi-square test was not possible, due to Cochrane restrictions, the contingency tables were converted into association tables with the use of Fisher's exact test.
Yule's coefficient of association was calculated only as a means of information, in cases where statistical significance was observed.
RESULTS
Eighty-three (25.7%) cases with at least one postoperative complication were observed, with only the initial or primary complication being considered here.
In these 83 cases, a total of 107 complications were diagnosed, with the occurrence of only one type of complication in 64 cases (77.1%), and of more than one type of complication in 19 (22.9%).
The complications included 26 cases of infection (8.0%), 11 of hematoma (3.4%), 24 of cutaneous necrosis (7.4%), 31 of lymphocele (9.6%), and 15 of inflammation (4.7%).
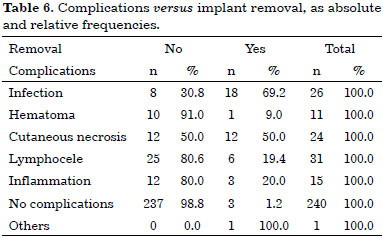
The correlation between the type of complication and the need for implant removal is presented in Table 6.
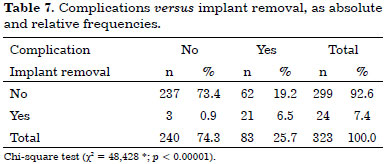
Of 323 IBR cases, 299 (92.6%) were successful and 24 (7.4%) evolved to implant removal.
Of the 299 successful reconstructions, there was some type of complication in 62 cases (19.2%), and no complication in 237 (73.4%).
Of the 24 cases of implant removal, there was some type of complication in 21 (6.5%), and no complication in 3 (0.9%). Table 7 summarizes and compares the groups that underwent a successful breast reconstruction with regard to the need for implant removal, as a function of the presence or absence of complications.
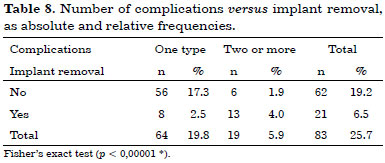
The number of complications versus implant removals, as absolute and relative frequencies, are shown in Table 8.
Infection occurred in 26 cases (8.0%), with 8 (2.5%) progressing to successful IBR, and implant removal in 18 (5.5%). In 297 cases (92.0%), no infection was observed, and 291 of these (90.1%) had a successful IBR; the implant was removed in 6 cases (1.9%) (Fisher's exact test, p < 0.00001*).
In the subgroup comprising 157 cases (48.6%) of reconstruction with silicone gel implants, there were 147 (45.5%) without any complication; among these, 142 (44.0%) received implants with a smooth surface, and 5 (1.5%) had implants with a textured surface. In this same subgroup, 10 (3.1%) complications were observed, with 9 (2.87%) involving implants with a smooth surface, and 1 (0.3%) with an implant with a textured surface.
In the subgroup of 141 cases (43.7%) of reconstruction with an inflatable implant filled with a saline solution (NaCl 0.9%), there were 132 cases (40.9%) without complications; of these, 42 (13.0%) were associated with implants with a smooth surface, and 90 (27.9%) with a textured surface. In this subgroup, 9 (2.8%) complications were observed, with 5 (1.5%) involving implants with a smooth surface, and 4 (1.2%) with implants with a textured surface.
In 22 cases (6.8%), it was necessary to insert a tissue expander in order to accommodate the final silicone implant. In the subgroup that received expanders, there were 13 (4.0%) without and 9 (2.8%) with complications (Fisher's exact test, p = 0.002%*). Infection occurred in 4 (1.2%) cases in this subgroup (Fisher's exact test, p = 0.08856; not significant).
Silicone implants coated with a layer of polyurethane foam were used in only 3 (0.9%) cases, without any complications.
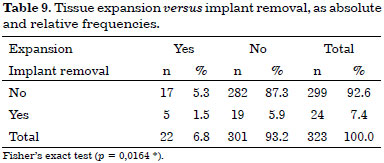
The absolute and relative frequencies of the cases with or without previous expansion, and whether or not the implant was removed, are listed in Table 9.
In the 301 cases (301 = 100%) in which the implant was final, without the need for tissue expansion, 24 (8.0%) were associated with implants with a volume smaller than 150 ml, 176 (58.5%) with volumes between 151 and 300 ml, and 13 (4.3%) with volumes greater than 450 ml.
Of the implants with volumes smaller than 150 ml, 23 cases (7.6%) evolved to a successful IBR, with implant removal in one (0.3%).
Of the implants with volumes between 151 and 300 ml, 169 cases (56.1%) evolved to a successful IBR, with implant removal in 7 (2.3%).
Of the implants with volumes between 301 and 450 ml, 82 cases (27.2%) resulted in a successful IBR, with implant removal in 6 (2.0%).
Of the implants with volumes greater than 540 ml, 8 cases (2.7%) evolved to a successful IBR, and 5 (1.7%) had implant removal.
The cases were regrouped into two subgroups according to implant volume of up to 300 ml, or above 300 ml; 200 cases (66.4%) were in the former subgroup and 101 (33.6%) in the latter.
Table 10 presents the results regarding cases of implant removal according to implant volume, in groupings either up to or above 300 ml.

DISCUSSION
Every time we perform surgery, we worry about possible complications; with the use of implants, one of the concerns is the need for implant removal.
We analyzed the reconstructions in this study, according to the presence or absence of complications, in the cases with or without implant removal, and observed that the rate of complications was 87.5% in those with implant removal, and 20.7% in those without removal; this showed a correlation between the occurrence of complications and implant removal (χ2 = 48.428*, p < 0.00001; Yule's coefficient of association).
Because more than one complication could occur simultaneously in each reconstruction case, a higher number of implant removals was observed when the number of complications was more than one (Fisher's exact test, p < 0.00001*).
The incidence of cases of implant removal (IR) among the reconstruction cases studied was 7.4%.
Of the 24 reconstructions leading to IR, 20 evolved to postoperative complications, 3 to late IR, and one had implant perforation.
Considering only the cases associated with complications referred to in the literature as the most serious (infection, necrosis, and hematoma), we observed a rate of IR of 43.5%; these complications were thus considered a risk factor for IR.
Bailey et al.10 found an incidence of inflammation of 20.0% in breast reconstructions using implants and extenders. In our study, inflammation occurred in 4.7% of those reconstructions.
In the literature, the incidence of liquid accumulation varied, with 12.0% in the study by Noone et al.11), 7.0% of seroma cases in the study by Bailey et al.10, 13.0% of seroma cases in the work by Vinton et al.12, and 20.6% of seroma cases according to Slavin13, with the use of expanders.
In the present study, the type of postoperative complication with liquid accumulation that occurred most frequently was the axillary lymphocele, with an incidence similar to that cited in the literature for breast implants for aesthetic purposes, which was 33.0%14. We observed necrosis of the cutaneous graft, infection, and hematoma with less frequency.
Even though it was the most frequent type of complication, lymphocele did not have a significant correlation with the need for IR, which occurred in 19.4% of the cases.
The incidence of cutaneous necrosis in the literature varied between 2.8% (Eberlein et al.15) and 15.0% (Noone et al.11). In the present study, necrosis of the cutaneous graft was the type of complication with the second highest correlation with the need for IR (50.0%).
The infection rate in the postoperative period also presents varying values in the literature: 2.0% in the study by Noone et al.11, 32.0% in the study by Armstrong et al.16, 4.0% in the work of Bailey et al.10 with the use of final implants and expanders, 13.0% in the work of Vinton et al.12, 9.4% in the study of Smitten & Sundell17 with the use of expanders, and 27.0% in the studies of Holley et al.6 with the use of implants and expanders.
Bailey et al.10 showed the importance of infection as a risk factor for IR following IBR, with an incidence of 57.0% in the cases leading to IR, and only 4.0% in the cases that did not require implant removal. In the present study, the correlation between one type of complication and implant removal was clear, when infection (69.2%) was the observed complication.
In an analysis of the cases of reconstruction failure, we observed that there was only one type of complication in one-third of cases, and in 75.0% of these cases, the type of complication was infection.
When we analyzed the 13 cases of failure that showed more than one type of complication, infection and necrosis were present in 92.3% and 76.9%, respectively.
At present, we tend to blame infection or necrosis as single agents resulting in reconstruction failures. However, both types of complications are associated in 76.9% of the cases, and this association could be responsible for the higher number of failures18,19.
A significant correlation between infection and lymphocele was observed in 38.5% of the cases, and between infection and hematoma in 7.7%.
In fact, we observed a correlation (p = 0.0001*) between the presence of infection, alone or combined with another type of complication, and the need for implant removal.
In the literature, the incidence of hematomas in IBR varies, and was 2.0% in the study by Vinton et al.12, 5.6% in the work of Eberlein et al.15, and 9.0% in the study by Holley et al.6 In the present study, hematoma was observed in 11 reconstructions, corresponding to 3.4% of all cases, with only one leading to implant removal.
When the presence of complications according to the type of implant used was assessed, the same incidence rate was observed in the group that received silicone gel implants (6.4%) and the group that received an inflatable implant filled with saline solution (6.4%).
The occurrence of complications after the use of expanders was 22.7%, but 6.3% when a final implant was directly used (301 cases). Expanders were used only in cases where a larger skin resection was required due to the proximity or to the volume of the tumor (22 cases). These were exceptions in this study. We observed a significant positive correlation (p = 0.0164 *)19,20 between use of implants with the use of expanders and implant removal.
In the group of reconstructions with volumes above 450 ml, the rate of failure was high (38.4%) and statistically significant. The use of implants with a volume greater than 450 ml often results in incomplete coverage of the implant, and is associated with greater tension in the skin, resulting in a higher risk of necrosis and prosthesis exposure.
According to Yule's coefficient, we verified a negative correlation between the use of implants with volumes up to 300 ml and the need for implant removal. Therefore, when the implants have a volume greater than 300 ml, the greater the probability of failure.
CONCLUSIONS
1. The presence of postoperative complications was a significant risk factor for silicone implant removal.
2. The risk of silicone implant removal was significantly higher in the presence of more than one type of postoperative complication, than with the occurrence of only one postoperative complication.
3. Infection was the main type of postoperative complication associated with silicone implant removal.
4. The use of expander implants was associated with a significantly higher risk of complications and implant removal than when the final implant was inserted from the beginning.
5. The use of implants with a volume greater than 300 ml resulted in a significantly greater risk of implant removal than the use of implants with volumes up to 300 ml.
REFERENCES
1. Zagouri F, Sergentanis TN, Tsigginou A, Dimitrakakis C, Zografos GC, Dimopoulos MA, et al. Female breast cancer in Europe: statistics, diagnosis and treatment modalities. J Thorac Dis. 2014;6(6):589-90.
2. Murray CD, Turner A, Rehan C, Kovacs T. Satisfaction following immediate breast reconstruction: Experiences in the early post-operative stage. Br J Health Psychol. 2014 Jun 20. [Epub Ahead of print] DOI: http://dx.doi.org/10.1111/bjhp.12112
3. Gurdin M, Carlin GA. Complications of breast implantations. Plast Reconstr Surg. 1967;40(6):530-3. PMID: 6077713 DOI: http://dx.doi.org/10.1097/00006534-196740060-00003
4. Cronin TD, Greenberg RL. Our experiences with the silastic gel breast prosthesis. Plast Reconstr Surg. 1970;46(1):1-7. PMID: 5430975 DOI: http://dx.doi.org/10.1097/00006534-197046010-00001
5. Nguyen KT, Hanwright PJ, Smetona JT, Hirsch EM, Seth AK, Kim JY. Body mass index as a continuous predictor of outcomes after expander-implant breast reconstruction. Ann Plast Surg. 2014;73(1):19-24. DOI: http://dx.doi.org/10.1097/SAP.0b013e318276d91d
6. Holley DT, Toursarkissian B, Vásconez HC, Wells MD, Kenady DE, Sloan DA, et al. The ramifications of immediate reconstruction in the management of breast cancer. Am Surg. 1995;61(1):60-5. PMID: 7832384
7. Spear SL, Baker JL Jr. Classification of capsular contracture after prosthetic breast reconstruction. Plast Reconstr Surg. 1995;96(5):1119-23. DOI: http://dx.doi.org/10.1097/00006534-199510000-00018
8. Little JW 3rd, Golembe EV, Fisher JB. The "living bra" in immediate and delayed reconstruction of the breast following mastectomy for malignant and nonmalignant disease. Plast Reconstr Surg. 1981;68(3):392-403. PMID: 6455688 DOI: http://dx.doi.org/10.1097/00006534-198109000-00030
9. Radovan C. Breast reconstruction after mastectomy using the temporary expander. Plast Reconstr Surg. 1982;69(2):195-208. PMID: 7054790 DOI: http://dx.doi.org/10.1097/00006534-198202000-00001
10. Bailey MH, Smith JW, Casas L, Johnson P, Serra E, de la Fuente R, et al. Immediate breast reconstruction: reducing the risks. Plast Reconstr Surg. 1989;83(5):845-51. PMID: 2710833 DOI:http://dx.doi.org/10.1097/00006534-198905000-00011
11. Noone RB, Murphy JB, Spear SL, Little JW 3rd. A 6-year experience with immediate reconstruction after mastectomy for cancer. Plast Reconstr Surg. 1985;76(2):258-69. PMID:2991957 DOI: http://dx.doi.org/10.1097/00006534-198508000-00012
12. Vinton AL, Traverso LW, Zehring RD. Immediate breast reconstruction following mastectomy is as safe as mastectomy alone. Arch Surg. 1990;125(10):1303-7. DOI:http://dx.doi.org/10.1001/archsurg.1990.01410220087012
13. Slavin SA, Colen SR. Sixty consecutive breast reconstructions with the inflatable expander: a critical appraisal. Plast Reconstr Surg. 1990;86(5):910-9. DOI: http://dx.doi.org/10.1097/00006534-199011000-00013
14. Williams JE. Experiences with a large series of silastic breast implants. Plast Reconstr Surg. 1972;49(3):253-8. PMID: 5060314 DOI: http://dx.doi.org/10.1097/00006534-197203000-00002
15. Eberlein TJ, Crespo LD, Smith BL, Hergrueter CA, Douville L, Eriksson E. Prospective evaluation of immediate reconstruction after mastectomy. Ann Surg. 1993;218(1):29-36. PMID:8328826 DOI: http://dx.doi.org/10.1097/00000658-199307000-00006
16. Armstrong RW, Berkowitz RL, Bolding F. Infection following breast reconstruction. Ann Plast Surg. 1989;23(4):284-8. PMID: 2817709 DOI: http://dx.doi.org/10.1097/00000637-198910000-00003
17. von Smitten K, Sundell B. The impact of adjuvant radiotherapy and cytotoxic chemotherapy on the outcome of immediate breast reconstruction by tissue expansion after mastectomy for breast cancer. Eur J Surg Oncol. 1992;18(2):119-23. PMID: 1582504
18. Jacombs A, Tahir S, Hu H, Deva AK, Almatroudi A, Wessels WL, et al. In vitro and in vivo investigation of the influence of implant surface on the formation of bacterial biofilm in mammary implants. Plast Reconstr Surg. 2014;133(4):471e-80e. DOI:http://dx.doi.org/10.1097/PRS.0000000000000020
19. Kato H, Nakagami G, Iwahira Y, Otani R, Nagase T, Iizaka S, et al. Risk factors and risk scoring tool for infection during tissue expansion in tissue expander and implant breast reconstruction. Breast J. 2013;19(6):618-26. DOI: http://dx.doi.org/10.1111/tbj.12175
20. Lovecchio F, Jordan SW, Lim S, Fine NA, Kim JY. Risk Factors for Complications Differ Between Stages of Tissue-Expander Breast Reconstruction. Ann Plast Surg. 2014 Mar 28. [Epub ahead of print]
1. Universidade Federal de São Paulo, São Paulo, SP, Brazil
2. Hospital São Paulo, Escola Paulista de Medicina, São Paulo, SP, Brazil
3. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
4. Universidade de São Paulo, São Paulo, SP, Brazil
5. Hospital das Clínicas, Faculdade de Medicina, Universidade de São Paulo, São Paulo, SP, Brazil
6. Escola Paulista de Medicina, São Paulo, SP, Brazil
Institution: Escola Paulista de Medicina - Universidade Federal de São Paulo, São Paulo, SP, Brazil.
Corresponding author:
Márcio Paulino Costa
Rua Gabriel dos Santos 759, 12 andar, Santa Cecília
São Paulo, SP, Brazil Zip code 01231-011
E-mail: marciopaulino@bol.com.br
Article received: July 20, 2014.
Article accepted: March 15, 2015.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter