

Original Article - Year 2014 - Volume 29 - Issue 3
Extended resection for treatment of dermatofibrosarcoma protuberans
Ressecção alargada no tratamento do dermatofibrossarcoma protuberante
ABSTRACT
INTRODUCTION: Dermatofibrosarcoma protuberans (DFSP) is a rare skin tumor with intermediate malignancy and low metastatic potential, but a high recurrence rate after surgical treatment. The classical treatment is extended resection with varying margins. Many studies have described Mohs micrographic surgery for treatment of this disease. This retrospective study was to verify if extended resection is a reliable DFSP treatment method.
METHOD: A total of 31 lesions were resected in 30 patients with DFSP between August 1968 and September 2013. All patients underwent radical surgical excision, with removal of 3 cm of healthy tissue on the lateral margins and with deep margin including an anatomical structure without tumor infiltration. Analyzed patient characteristics included sex, age, tumor site, previous treatment, and peculiar characteristics observed during the follow-up period.
RESULTS: Nineteen (63.3%) patients were male and 11 (37.7%) female. Their average age at tumor presentation was 40.9 years. The tumors were located on the trunk (61.3%), head (226%), upper limbs (6.4%), lower limbs (6.4%), and neck (3.3%). No previous treatment was reported in 58.1% of the patients. One patient (3.3%) developed recurrence and died due to the surgical treatment; three patients (10.0%) died from other causes.
CONCLUSIONS: Extended resection with 3-cm margins and removal of healthy anatomical structures is an effective treatment for DFSP.
Keywords: Dermatofibrosarcoma protuberans; Dermatofibrosarcoma; Skin tumors.
RESUMO
INTRODUÇÃO: Dermatofibrossarcoma Protuberante (DFSP) é um tumor de pele raro e de malignidade intermediária, com baixo potencial metastático, mas alta taxa de recorrência após tratamento cirúrgico. O tratamento clássico é a ressecção alargada, com margens variáveis. Muitos trabalhos descreveram os resultados da cirurgia micrográfica de Mohs no tratamento desta afecção. O objetivo deste estudo retrospectivo é verificar se a ressecção alargada constitui um método confiável no tratamento do DFSP.
MÉTODO: Entre agosto de 1968 e setembro de 2013, 31 lesões foram ressecadas em 30 pacientes com DFSP. Todos os pacientes foram submetidos à excisão cirúrgica radical, com remoção de 3 cm de tecido sadio nas margens laterais e com a margem profunda incluindo uma estrutura anatômica não infiltrada pelo tumor. Os seguintes aspectos foram estudados: gênero, idade, local da lesão, tratamento prévio e características peculiares da proservação.
RESULTADOS: Dezenove (63,3%) pacientes eram do sexo masculino e 11 (37,7%), do feminino. A média de idade da apresentação foi de 40,9 anos. As lesões estavam localizadas em tronco (61,3%), cabeça (22,6%), membros superiores (6,4%), membros inferiores (6,4%) e pescoço (3,3%). Tratamento prévio não foi observado em 58,1% dos pacientes. Um paciente (3,3%) evoluiu com recidivas e óbito, em decorrência do tratamento cirúrgico; três (10,0%) faleceram por outras causas.
CONCLUSÕES: A ressecção alargada com margens de 3 cm, com remoção de estrutura anatômica sadia, constitui método eficiente no tratamento do DFSP.
Palavras-chave: Dermatofibrossarcoma protuberante; Dermatofibrossarcoma; Neoplasias cutâneas.
Dermatofibrosarcoma protuberans (DFSP) is a rare skin tumor. Initially described by Taylor1 in 1890, it has slow growth and low rate of metastasis, despite its aggressive local behavior. In 1924, it was commonly known as "Darier-Ferrand Tumor"2, after he reported its progressive and recurrent behavior in the context of economical resections. In 1925, the term was modified by Hoffman3 to "dermatofibrosarcoma protuberans", in an attempt to emphasize the protruding appearance of the tumor.
The tumor typically affects individuals in the third and fourth decades of life and is slightly more predominant in men4. Clinical diagnosis in early tumor stages is difficult due to its similarity to cutaneous vascular malformations4. Pain is manifested in 25% of cases5. Its evolution is initially characterized by plaque formation (Figures 1 and 2) originating from one or more nodules (Figure 3), which gives the tumor its protruding appearance (Figure 4). The tumor may develop over a long period of time and the nodules may be a reddish or brownish color4,6. The tumor often resembles a keloid6,7 and can present as an atrophic lesion similar to circumscribed scleroderma4.
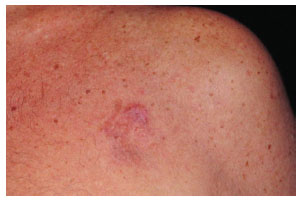
Figure 1. Case 21, male, 66 years old, presenting a plaque-shape tumor in the left pectoral region.
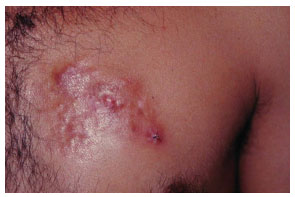
Figure 2. Case 15, male, 31 years old, plaque-shape left pectoral dermatofibrosarcoma protuberans.
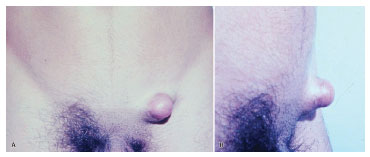
Figure 3. Case 1, male, 19 years old, tumor in the left inguinal region, with a nodular shape and protruding appearance, in front view (A) and profile view (B).
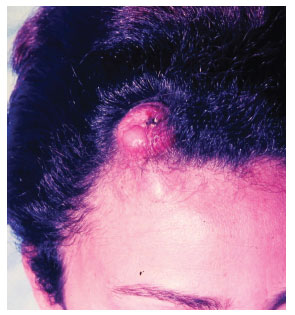
Figure 4. Case 2, female, 31 years old, frontal tumor with a protruding appearance.
The trunk is most commonly affected, followed by the proximal regions of the lower limbs, head, and neck4,6,8-10. This tumor is uncommon on hands and feet, and does not typically involve more than limited regions of the skin4. History of previous trauma and association with immunosuppressive conditions have also been reported4.
The tumor is located in the dermis and invades the subcutaneous tissue through sheath cell bundles branching parallel to the epidermis4,9,10. A subcutaneous form of this tumor, not connected to the dermis, has also been described. Epithelial ulceration may occur due to trophic changes of the skin overlying the tumor6.
DFSP is characterized by a dense and uniform fibroblast population arranged in a spiral or radial form (wind-vane, cart-wheel, or whirlwind-like aspect) around the small vessels4,6,9,10, which gives the tumor a reed mat-like, or storiform, appearance4,6 (Figure 5). In the adipose tissue, the tumor appears reticulated due to the penetration of fat lobes that presents a honeycomb appearance10 (Figure 6).
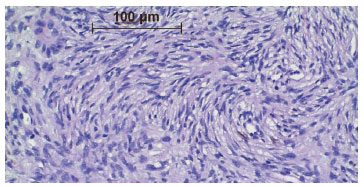
Figure 5. Photomicrograph showing tumor cells arranged around small blood vessels, with a whirlwind or storiform appearance. Medium amplification.
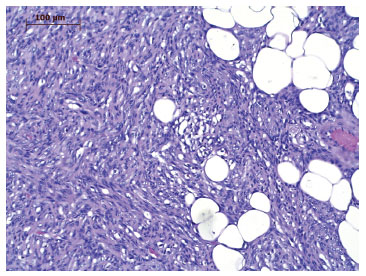
Figure 6. Photomicrography of dermatofibrosarcoma protuberans tumor tissue infiltrating the subcutaneous adipose tissue. Magnification of 200×.
CD34 is the main immunohistochemical marker of DFSP. Tumor cells positive for CD34 present a diffuse pattern. These cells are negative for other markers; e.g., XIIIa factor, S-100 protein, desmin, and actin4,9 (Figure 7). Although controversial, the tumor histogenesis of DFSP has been increasingly accepted as a fibroblastic4,9.
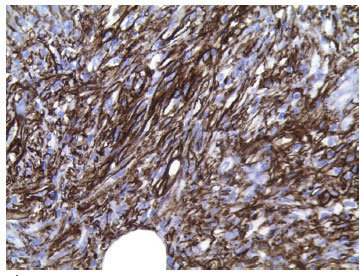
Figure 7. Immunohistochemical stain, showing CD34 positive for DFSP.
A chromosomal translocation involving the collagen type 1 alpha 1 (COL1A1) gene on chromosome 17 and PDGFB on chromosome 22 is present in more than 90% of cases. This change is characterized by a reciprocal translocation t(17;22)(q22;q13) and a supernumerary ring chromosome r(17;22) derived from the translocation, which leads to the fusion of the COL1A1 and Beta-type platelet-derived growth factor (PDGFB) genes4. PDGFB may act as a mitogenic factor for autocrine stimulation of the PDGF receptor. Imatinib mesylate, a tyrosine kinase antagonist and PDGF receptor, has been used for treatment of DFSP4 with good results, even in cases with metastasis.
Metastases are uncommon and mainly occur in cases of multiple recurrences, when DFSP evolves to fibrosarcoma. Fibrosarcomatous DFSP can induce metastases in about 20% of cases4. The most affected organs are the liver, lungs, pancreas, and bones11,12, whereas lymph node involvement is rare4,6,12. Tumors of other types in the same individual may have a genetic correlation or be sporadic6. The familial form is an exception4.
Depending on the progressive and recurrent behavior of the tumor, the previous standard DFSP treatment was radical surgical resection, with extended lateral margins of healthy tissue. The deep margin included an anatomical structure that was not infiltrated by the tumor, such as fascia, muscle, or bone external lamina. Over the past 15 years, however, Mohs micrographic surgery (MMS) has been increasingly adopted for treatment of this disease, despite the numerous biases discussed later.
Radiotherapy for therapeutic treatment of primary lesions shows poor results, although it may complement surgical treatment, especially in the presence of fibrosarcomatosis variants13.
Although rare and indolent, the tumor can affect noble areas of body; e.g., the face and limbs, and recurrences can lead to development of deformities. Therefore, it is essential to establish an ideal method for treatment of DFSP.
The aim of this study was to review clinical and anatomopathological aspects of DFSP, determine whether extended resection is a reliable method for its treatment, and analyze the biases of literature recommending MMS.
METHOD
Data were reviewed from 30 patients, corresponding to 31 DFSP tumors treated between August 1968 and September 2013. Patient information included sex, age, tumor location, previous treatment, and information regarding the follow-up period. The first ten cases in this series have been already published7. However, post-operative aspects and later observations were included in the data of the current study.
All patients were operated on and monitored at the Plastic Surgery Clinic of Felício Rocho Hospital, Belo Horizonte- Minas Gerais. The surgical technique consisted of radical surgery with resection of 3 cm of healthy tissue in the lateral margins and a deep margin that included an anatomical structure that was not invaded by the tumor; e.g., fascia, muscle, or bone external lamina. All individuals were subjected to reconstruction procedures such as grafts or skin flaps. In three cases, the flaps were previously expanded.
The patients were followed up through clinical examinations every four months for the first two years and every six months between the third and the fifth years after surgery. The patients were advised to undergo annual medical examinations for an indefinite period of time. Patients who did not attend this examination were monitored by postal mail, e-mail, or telephone contact.
All surgical specimens were examined at the Pathological Anatomy Service of the same institution. All samples were stained with hematoxylin-eosin. In some cases, immunohistochemistry findings were necessary for differential diagnosis.
This retrospective study was approved by the Research Ethics Committee of Felício Rocho Hospital.
RESULTS
Nineteen patients (63.3%) were male and 11 (37.7%) female, corresponding to a male-female ratio of 1.7 (Figure 8).
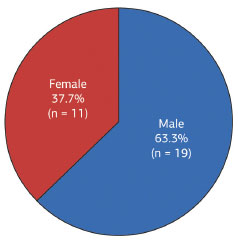
Figure 8. Distribution of dermatofibrosarcoma protuberans according to patient sex.
Patient age at the time of surgery ranged between 9 and 72 years, with an average age of 40.9 years. The tumors occurred mainly between the third and fifth decades of life, corresponding to 63.4% of cases, and were distributed in an ascendant-descendant pattern among other age groups. The peak incidence age was 30-39 years (Figure 9).
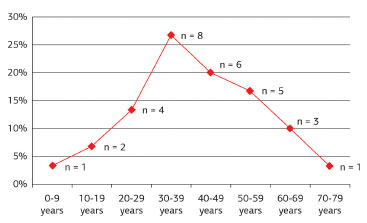
Figure 9. Distribution of dermatofibrosarcoma protuberans according to patient age group.
Of 31 lesions, 19 (61.3%) affected the trunk; seven (22.6%), the head; two (6.4%), the upper limbs; two (6.4%), the lower limbs, and one (3.3%), the neck (Figure 10).
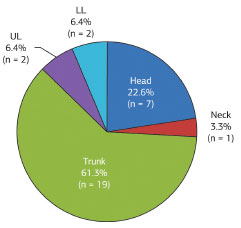
Figure 10. Distribution of dermatofibrosarcoma protuberans according to location.
Of patients who had undergone previous treatment, six (19.3%) had tumor resection, two (6.4%) received radiation therapy combined with tumor resection, and one (3.3%) was previously treated with chemotherapy combined with tumor resection. Four patients (12.9%) were misdiagnosed with keloids and were treated with intralesional injections of triamcinolone acetonide. Among misdiagnosed patients, treatment in three (9.6%) cases was combined with tumor resection, and one patient (3.3%) received only a corticosteroid infiltration. In 18 (58.1%) individuals, no previous procedures were reported (Figure 11). Immunohistochemical analysis was used to correctly diagnose nine tumors (29.0%).
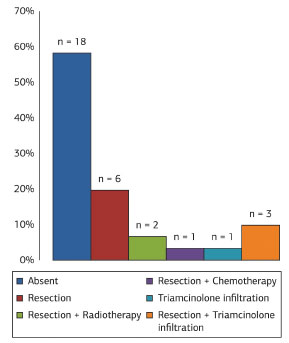
Figure 11. Distribution of dermatofibrosarcoma protuberans tumors according to previous treatment.
All lesions were addressed by extended resection, as described above. Seventeen (51.6%) were reconstructed with skin flap; 7 (21.2%) with skin grafts; three (9.1%) with pre-expanded skin flaps; one (3.0%) with skin flap and graft; one (3.0%) with skin grafts on a muscle flap, and two (6.1%), with fasciocutaneous flap and skin graft (Figure 12). One patient (3.0%) underwent transmetacarpal amputation of the index finger and flap reconstruction (Figure 13). One surgery (3.0%) consisted of a craniectomy and maxillectomy, which after two attempts was followed by relapses, extended removal and reconstruction with skin flap (Figure 14).
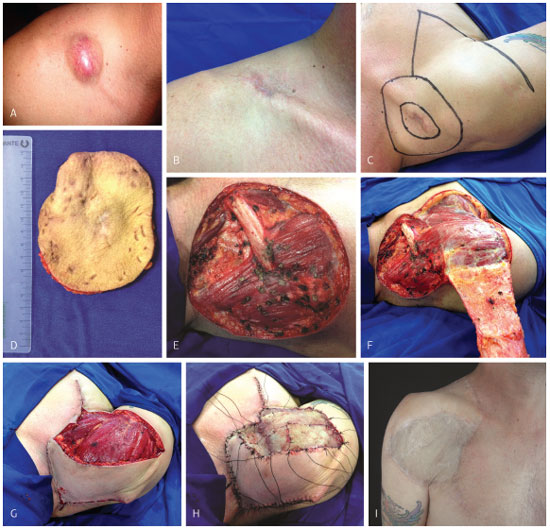
Figure 12. Case 29, male, 47 years old, right supraclavicular DFPS. (A) Nodule with a reddish-brownish color and protruding aspect. (B) Excisional biopsy performed elsewhere. (C) Surgical plan. (D) Surgical specimen. (E) Result after extensive resection, including, in the surgical specimen, the periosteum of the right clavicle. (F) Forming the fasciocutaneous flap. (G) Flap fixation in the open area. (H) Skin graft fixation in the recipient bed. (I) Late postoperative result.
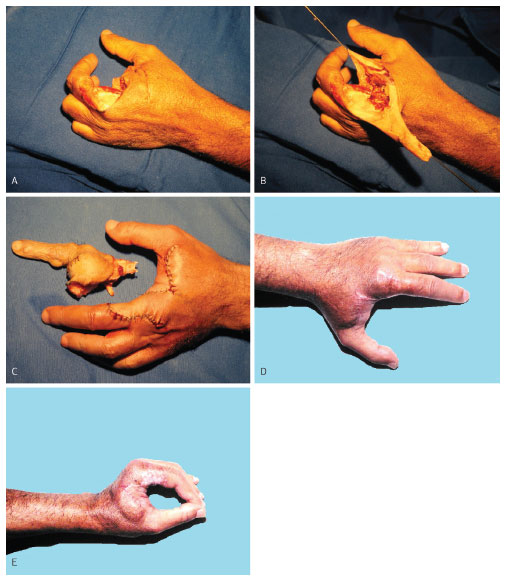
Figure 13. Case 6, male, 54 years old, DFSP affecting forefinger and second left interdigital fold. (A) Transmetacarpal amputation of the index finger and resection of the radial side of the first phalanx of the middle finger. (B) Hood pedicled flap in the dorsum of the hand. (C) Sutured flap and surgical specimen. (D) Late postoperative result. (E) Late postoperative result with tweezers movement.
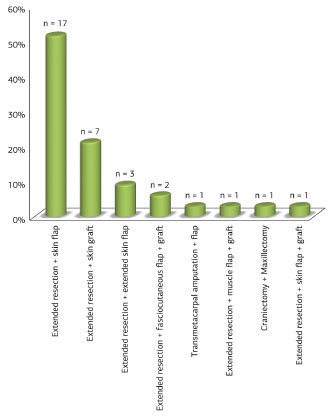
Figure 14. Distribution of dermatofibrosarcoma protuberans tumors according to surgical treatment.
The follow-up periods varied between one and 491 months, with an average of 114 months. Twenty patients (66.7%) remained alive and disease-free; one patient (3.3%) died after surgical treatment for cancer, and three (10.0%) died from other causes. Six individuals (20.0%) did not return for consultation, so recent examinations were not available (Figure 15). Only one patient (3.3%) had tumor recurrence. Finally, one case (3.3%) showed two lesions on separate occasions and one patient (3.3%) was diagnosed with a Bednar tumor.
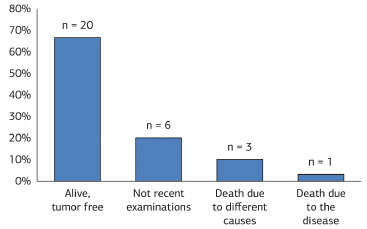
Figure 15. Distribution of patients with dermatofibrosarcoma protuberans according to outcome.
DISCUSSION
DFSP has been widely reported by Brazilian authors, with 23 published articles available through Lilacs and Medline3,7,11-18.
The tumor is reportedly most common in individuals in their third and fourth decades of life4. In this study, the lesions affected patients between 9 and 72 years old, with an average age of 40.9 years. The tumors developed between the third and fifth decades of life in 63.4% of the cases, and the peak age at tumor presentation was 30-39 years, similar to other published findings.
DFSP is slightly more prevalent in men4. In this cohort, 63.3% of patients were male and 37.7% female, resulting in a male-female ratio of 1.7, similar to previous reports.
The most affected areas include, in decreasing order of frequency, the trunk, the proximal region of the limbs, and the head and neck region4,6,8-10. This study generally agrees with previous findings, with 61.3% of the lesions in the trunk, 22.6% in the head, 6.4% in the upper limbs, 6.4% in the lower limbs, and 3.3% in the neck.
In 18 tumors, the patient had not received previous treatment. In the remaining 13 tumors, the previous treatment was incorrect, with narrow excision margins, radiotherapy, chemotherapy, and intralesional corticosteroid injections.
All patients were followed for an average of 114 months, ranging between one and 491 months. Most patients (66.7%) achieved a cure without tumor recurrence or metastasis. One patient (3.3%), who presented with an extensive tumor on the lower eyelid and cheek and was treated elsewhere with intralesional triamcinolone injections for 18 years, had tumor recurrence seven years after radical surgical treatment. This patient died due to surgical treatment for the recurrent tumors (craniectomy and extended maxillectomy).
Extended surgical resection is the standard treatment for DFSP. The importance of radical excision as the primary treatment of this condition was confirmed in 1991, when Smola reported six recurrences in cases with marginal excision in a cohort of 20 patients who received marginal, extended, and radical resections17.
Recommendations for lateral resection margins vary significantly - between one and 5 centimeters - and was classically established as being of 3 centimeters4,7,8. A progressive reduction in the recurrence rates was noted by increasing the surgical margins4. Factors that contribute to a worse prognosis include a high mitotic index, age greater than 50 years, presence of a fibrosarcomatosis variant and excision margins compromised or very small17. The use of extended resection showed satisfactory results in secondary surgeries17. Vanderweyer observed only one instance of recurrence in a study of 18 patients treated with excision but free histological margins17. Dawes and Hanke reported a series of cases in which 85% of a total of 24 patients reduced their surgical margins with an excision of 2.5 cm from the tumor. However, in both cases, a standard margin of 3 cm was not sufficient for the complete resection of the tumor. Therefore, regardless of the type of surgery performed, the surgical specimen margins should be examined for the presence of the tumor17.
MMS was developed in 1932 and numerous studies in the past 15 years have reported its superiority over extended surgical resection. The method, as chemosurgery, has been used since 1985 for treatment of DFSP. Since then, MMS has been extensively used as a therapeutic option17. The main difficulties related to MMS are: 1) patient intolerance if the procedure time is lengthy and requires maintenance of the same position on the operating table, since the operation is usually performed under local anesthesia; 2) possibility of interrupting the procedure in case of bone involvement or dangerous anatomic areas; 3) limited coverage of this type of treatment by the national health insurance companies, due to high costs and suspected fraud17. Comparative studies addressing surgical therapy for MMS and extended resection showed lower rate of relapse in the first procedures mentioned17.
MMS has been considered the first line of treatment for DFSP. However, a prospective, multicenter and randomized study -desirable for cases of rare diseases - comparing the efficacy of MMS and extended resection has not yet been performed17. An information bias was detected in these articles, thanks to the absence of description of the actual size of resection margins and the great variation in this parameter found in the literature. Moreover, the MMS series reported do not illustrate cases with metastases and bone invasion, which demonstrates a selection bias8. Finally, the monitoring period reported by different studies is not long enough, which demonstrates a detection bias. DFSP should be monitored for long time due to its recurrent behavior. Relapses after 148, 2317, and 2617 years after surgery have been described, as well as metastases 16 years after the initial surgery11. In this study, a patient who died in the immediate postoperative period after craniectomy and maxillectomy had remained cancer-free for seven years. Another study reported a 25% recurrence rate after five years17. Yu et al.19 reported no sign of recurrence after 68 months of follow-up in 25 patients with DFSP treated with extended resection, modified extended resection, MMS, and MMS followed by extended resection; the authors concluded that appropriate preoperative planning combined with satisfactory selection of surgical approach are key elements for successful therapy17,19.
This innovative study demonstrates some unique options for treatment of DFSP. The use of previously expanded flaps constitutes a new approach for treatment of this disease, since only two articles in the literature deal with this issue7. In the series, three cases (10.0%) of frontal lesions, two with bone involvement, underwent surgery with previously expanded flaps, with reportedly satisfactory results. Moreover, this study reports four cases(13.3%) previously treated with intralesional infiltration of triamcinolone, due to keloid misdiagnosis. There is only one report in the medical literature of patients with DFSP previously treated with corticosteroid injections related to misdiagnosis19,20. It is widely known that steroids are immunosuppressive and cause changes in tissue permeability. Although ineffective prior surgical treatment leads to recurrences and, therefore, a worse prognosis, the effect of intralesional triamcinolone infiltration in tumor behavior had not yet been established. The only case in this study that showed relapse and died as a result of the disease had received corticosteroid infiltration for 18 years.
The occurrence of other types of tumors in patients with DFSP may be due to a genetic correlation or be sporatic4,8. In this study, four individuals (13.3%) were affected by other neoplastic diseases. One patient with right scapular DFSP was cured after extended resection, but died 81 months later due to complications from chronic lymphoid leukemia. Another patient who presented with a tumor on the index finger and was treated with a transmetacarpal amputation, died after 127 months during the immediate postoperative period for total cystectomy due to bladder carcinoma. A third patient with frontal neoplasia underwent extended excision, but died after 17 years, during treatment for a metastatic renal carcinoma. The fourth, who presented a lesion on the trunk, developed an extensive superficial melanoma (Breslow depth of invasion 0.94 mm and Clark level II) in the left arm and remains alive and healthy after treatment.
CONCLUSION
DFSP is a rare skin tumor.
The treatment of choice is complete surgical resection. Extended resection, characterized by the removal of 3 cm of healthy tissue in the lateral margins and deep margin, including an anatomical structure not infiltrated by tumor, e.g. fascia, muscle or bone external lamina, showed effective results in terms of controlling the tumor. This fact can be confirmed by the absence of metastases and the presence of only one instance of tumor recurrence, despite the locally aggressive behavior of this disease.
This study also highlights the importance of considering DFSP for all presentations of protruding tumors, especially those on the trunk, limbs, and head, which can lead to precise diagnosis and, therefore, appropriate therapy. Finally, extended surgical resection is a reliable, secure, and appropriate option for treatment of this disease
ACKNOWLEDGMENTS
We thank Dr. Gil Patrus Mundim Pena for providing and preparing the photomicrographs and immunohistochemistry figures used in this work.
REFERENCES
1. Taylor RW. Interesting cases, dermatological and otherwise. J Cutan Genito-Urin Dis. 1890;8:379-88.
2. Darier J, Ferrand M. Dermatofibromes progressifs et récidivants ou fibrosarcomes de la peau. Ann Dermatol Syphil. 1924;5:545-62.
3. Costa OG. Progressive recurrent dermatofibroma (Darier-Ferrand); anatomoclinical study. Arch Derm Syphilol. 1946;54(4):432-54. http://dx.doi.org/10.1001/archderm.1946.01510390057005. PMid:21000203
4. McKee PH, Calonge E, Granter SR. Dermatofibrosarcoma protuberans. In: McKee PH, Calonge E, Granter SR, editores. Pathology of the skin with clinical correlations. 3. ed. Philadelphia: Elsevier Mosby; 2005. p. 1729-35.
5. Taylor HB, Helwig EB. Dermatofibrosarcoma protuberans. A study of 115 cases. Cancer. 1962;15(4):717-25. http://dx.doi.org/10.1002/1097-0142(196207/08)15:4<717::AIDCNCR2820150405>3.0.CO;2-2. PMid:13919964
6. Shapiro L, Brownstein MH. Dermatofibrosarcoma protuberans. In: Andrade R, Gumport SL, Popkin GL, Rees TD, editores. Cancer of the skin: biology, diagnosis, management. Philadelphia: WB Saunders; 1976. v. 2, p. 1069-78.
7. Silva AF Fo, Andrade JS Fo, Siqueira IMG, Saraiva PS, Magalhães GM, Alves JCRR. Dermatofibrossarcoma protuberante: aspectos clínicos e cirúrgicos. An Bras Dermatol. 1997;72(3):247-51.
8. McPeak CJ, Cruz T, Nicastri AD. Dermatofibrosarcoma protuberans: an analysis of 86 cases-five with metastasis. Ann Surg. 1967;166(5):803-16. http://dx.doi.org/10.1097/00000658-196711000-00011. PMid:4964386
9. Enzinger FM, Weiss SW. Fibrohistiocytic tumors of intermediate malignancy. In: Enzinger FM, Weiss SW, editores. Soft tissue tumors. 3. ed. St Louis: Mosby; 1995. p. 325-49.
10. Heenan PJ. Tumors of the fibrous tissue involving the skin. In: Elder D, editor. Lever's histopathology of the skin. 8. ed. Philadelphia: Lippincott-Raven; 1997. p. 847-87.
11. Fiod NJJ, Rezende JFN, Guitmann C, Meohas W, Estevan R. Dermatofibrosarcoma protuberans metastático para pulmão: relato de caso. Rev Bras Cancerol. 1994;40(3):169-73.
12. Silva RG, Paiva RKS, Santos CER, Melo ELR, Dias JA, Bravo Neto GP. Ressecção de metástase hepática de sarcomas de partes moles. Rev Col Bras Cir. 2006;33(6):380-6. http://dx.doi.org/10.1590/S0100-69912006000600010.
13. Bastos EM, Santos IDAO, Ferreira LM, Paiva GR, Brunstein F, Lima AH, et al. Radioterapia após cirurgia para dermatofibrossarcoma protuberante de grandes dimensões: revisão da literatura e relato de casos. Acta Oncol Bras. 2004;24(2):612-7.
14. Cláudio-da-Silva CS, Boghossian LC, Gonçalves LFF, Franco D, Franco T. Dermatofibrossarcoma protuberans gigante. Rev Soc Bras Cir Plast. 1997;12(2):7-14.
15. Fleury LFF Jr. Sarcomas cutâneos primários: estudo retrospectivo de casos registrados na divisão de Dermatologia do Hospital das Clínicas da FMUSP no período de 1992 a 2002 [Dissertação]. São Paulo: Universidade de São Paulo; 2007.
16. Golcman B, Bonetti EY, Friedhofer H, Filomeno LT, Monteiro AA Jr. Dermatofibrossarcoma protuberante. Rev Hosp Clin Fac Med Sao Paulo. 1984;39(4):197-200. PMid:6531616.
17. Alves JCRR. Tratamento cirúrgico radical do dermatofibrossarcoma protuberante. In: Anais da Academia Mineira de Medicina [Relação de trabalhos aprovados para ingresso como membros titulares]; 2010. Belo Horizonte, Brasil.
18. Furtado JPR, Furtado CF, Alves JCB, Morais CS, Laporte GA. Dermatofibrossarcoma protuberans: experiência e discussão numa série de casos. Rev AMRIGS. 2011;55(1):48-52.
19. Yu W, Tsoukas MM, Chapman SM, Rosen JM. Surgical treatment for dermatofibrosarcoma protuberans: the Dartmouth experience and literature review. Ann Plast Surg. 2008;60(3):288-93.
20. Manalan SS, Cohen IK, Theogaraj SD. Dermatofibrosarcoma protuberans or keloid-a warning. Case report. Plast Reconstr Surg. 1974;54(1):96-8. http://dx.doi.org/10.1097/00006534-197407000-00020. PMid:4832476
1. Doctor in Surgery at the Federal University of Minas Gerais (UFMG), Titular member of the Brazilian Society of Plastic Surgery (BSPS), Full Professor of Surgical and Anesthetic Techniques at the Faculty of Medicine of the University of Itaúna (UIT), Member of the clinical staff of the Plastic Surgery Service of Felício Rocho Hospital and of the Institute for Advanced Plastic Surgery, Belo Horizonte, MG, Brazil
2. Physician, Plastic Surgeon, Specialist Member of the Brazilian Society of Plastic Surgery (BSPS), Member of the clinical staff of the Plastic Surgery Service of Felício Rocho Hospital and of the Institute for Advanced Plastic Surgery, Belo Horizonte, MG, Brazil
3. Physician, Plastic Surgeon, Titular Member of the Brazilian Society of Plastic Surgery (BSPS), Member of the clinical staff of the Plastic Surgery Service of Felício Rocho Hospital and of the Institute for Advanced Plastic Surgery, Belo Horizonte, MG, Brazil
4. Physician, Professor of Pathology at the Faculty of Medical Sciences of Minas Gerais (FCMMG), Chief of the Pathological Anatomy Service of the Felício Rocho Hospital, Belo Horizonte, MG, Brazil
5. Physician, Aspirant Member in Training of the Brazilian Society of Plastic Surgery (BSPS), Specializing Physician at the Plastic Surgery Service of Felício Rocho Hospital, Belo Horizonte, MG, Brazil
Institution: Study performed at the Plastic Surgery Clinic of Felício Rocho Hospital, Belo Horizonte, MG, Brazil.
Corresponding author:
Rebeca Paohwa Liu da Fonseca
Rua Dionísio Cerqueira, 42/301 - Gutierrez
Belo Horizonte, MG, Brazil CEP 30430-140
E-mail: rebeca_liu@hotmail.com
Article received: June 4, 2014.
Article accepted: January 14, 2015.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter