

Original Article - Year 2014 - Volume 29 -
Use of polyurethane foam dressings on skin graft donor sites
Uso de espumas em áreas doadoras de enxertos
ABSTRACT
INTRODUCTION: The success of a skin graft is evaluated by not only the integration of the graft itself, but also the quality of the recovery of the donor site. Despite the fact that graft donor sites represent the best place to study wound healing, treatment regimens for donor sites have not been studied extensively.
METHOD: To evaluate the efficiency of polyurethane foam as a dressing for graft donor sites. Methods: We conducted a prospective study in which graft donor sites were treated with polyurethane foam dressing, as an alternative to a cellulose acetate film.
RESULTS: We treated 11 patients and 14 donor graft sites. Problems associated with the use of polyurethane foam included prolonged adherence (73%) and an unpleasant odor (45%). The majority of patients reported that they found the dressing to be unsatisfactory (73%).
CONCLUSIONS: The use of a polyurethane foam was shown to be ineffective as a graft donor site dressing, due to the high rate of associated complications.
Keywords: Bandages; Dressing materials; Skin healing; Skin grafts; Donor site; Epithelialization
RESUMO
INTRODUÇÃO: O sucesso de um enxerto de pele é avaliado não apenas pela integração do enxerto em si, mas também pela qualidade da recuperação da área doadora. A despeito de as áreas doadoras de enxertos representarem o melhor local para estudo de cicatrização de feridas, regimes de tratamento, de áreas doadoras, tem sido incompletamente estudados.
OBJETIVO: Avaliação da eficácia de espumas de poliuretano como curativo de áreas doadoras de enxertos.
MÉTODO: Estudo prospectivo no qual áreas doadoras de enxertos foram tratadas com espumas de poliuretano como alternativa a filmes de acetato de celulose.
RESULTADOS: Foram tratados 11 pacientes e catorze áreas doadoras de enxerto. Aderência prolongada (73%) e odor desagradável (45%) foram os problemas encontrados. Os resultados foram considerados insatisfatórios na grande maioria dos casos (73%).
CONCLUSÕES: O uso de espumas de poliuretano mostrou-se ineficaz, nesse grupo de pacientes, devido à ocorrência de alto índice de complicações.
Palavras-chave: Bandagens, Materiais de curativos, Cicatrização, Enxertos de pele, Área doadora, Epitelização
Skin grafts have been used in the treatment ofwounds for over 100 years. In contemporary practice, a variety of dressings may be used in the treatment of the donor site. The success of surgery is evaluated not only by the integration of the graft, but also the quality of the recovery of the donor site1,2. Despite the fact that graft donor sites represent the best place to study wound healing, treatment regimens for donor sites have not been extensively studied3,4.
Graft donor sites are often painful, and are prone to infection if the exudates are not retained within a dressing. The donor site may also suffer chronic complications, including hypertrophic scarring and dyschromia5.
It is known that complete re-epithelialization is achieved earlier with use of a dressing that can provide a moist environment for the wound1. With such dressings, the wound is better protected against dehydration, infection, and trauma. Moreover, an occluded wound may be associated with less pain, due to reduced stimulation of free nerve endings. However, occlusive dressings can macerate the site of healing6.
The present study was conducted to evaluate the efficiency and safety of the use of polyurethane foam as a dressing for graft donor sites, until complete re-epithelialization had occurred.
METHODS
This prospective study was conducted from April 1 to August 31, 2013. The study included adult patients (n=11) who were admitted to the Burn Treatment Center of the State General Hospital of Bahia, with 3rd degree burns affecting <20% of the body surface. Individuals with clinical and/or laboratory evidence of any condition that could compromise skin healing (advanced age (>65 years), blood dyscrasias, uncontrolled diabetes mellitus, uncontrolled hypertension, severe anemia (Hb <7mg%), alcoholism, hypoalbuminemia (albumin <2.5mg/dL), severe vitamin deficiencies, and decompensated coronary heart, kidney, or liver disease) were excluded. We also excluded patients with a documented contact allergy of any kind; chronic dermatoses; the presence of 1 or more necrotic, infected, or grossly contaminated burn site; or donor areas for thin (<0.008 in) or thick (> 0.014 in) grafts. Finally, patients were excluded if they refused to participate in the study, or refused to sign the Terms of Free and Informed Consent.
Surgical technique
Antisepsis was performed using a 0.5% chlorhexidine in alcohol solution, after which, sterile surgical drapes were placed. General anesthesia was achieved using an inhalational anesthetic. During the induction of anesthesia, antibiotic prophylaxis (1000 mg cefazolin) was administered to the graft donor site.
Surgery involved the following steps
Removal of granulation tissue and fibrotic tissue from bleeding sites.
1. Acquisition of a partial-thickness skin graft (0.012 inches) of the opposite symmetrical region or neighboring region most proximal to the burn.
2. Rigorous hemostasis applied to both the donor site and the recipient site, using compresses soaked in a vasoconstrictor solution (adrenaline, 1:250,000 IU) and very light electrocautery.
3. Fixation of the graft to the debrided wound area with simple points spaced apart with nylon 5-0.
4. Dressing of the recipient site: a non-adherent gauze was applied directly to the graft, and overlaid with a cotton gauze. These layers were covered with a crepe bandage.
5. Dressing of the donor site, using a sterile polyurethane foam with cotton gauze as secondary dressing.
Digital photographs were taken during the procedure, for surgical documentation.
Post-operative course
Patients remained in the hospital for a minimum of 7 days. On the 5th post-operative day, external dressings were completely removed so that the donor and recipient sites were covered only by the non-adherent gauze, and the foam film, respectively.
From the 7th post-operative day, patients were instructed to wet the donor site during washing, and remove the parts of the foam that were spontaneously detaching. Patients that were clinically stable, with fully integrated grafts, and clean, dry donor sites, were discharged on the 8th post-operative day. All patients remained hospitalized until these conditions were met.
All patients were monitored until full detachment of the foam, and total re-epithelialization of the donor site. At the final post-operative follow-up appointment, photographs were taken to evaluate the results. Photographs were also obtained from those patients who returned in the late post-operative period (Figures 4-7).

Figure 1. Distribution of lesions.

Figure 2. Post-operative complications.

Figure 3. Results according to patient satisfaction.
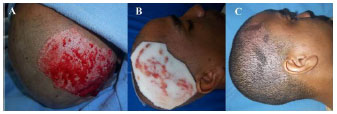
Figure 4. 29-year-old man with donor site on the scalp; (A) intra-operatively, (B) on the 5th post-operative day, with foam adherent, (C) fully re-epithelized on the 55th post-operative day.

Figure 5(A) 29-year-old man with donor site on the right forearm; (A) on the 5th post-operative day, (B) fully re-epithelized on the 46th post-operative day.

Figure 6(A) 25-year-old man with donor site on the thigh; (A) in the immediate post-operative period, (B) on the 14th post-operative day, with foam partially adherent, (C) fully re-epithelized on the 54th post-operative day.
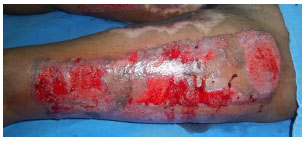
Figure 7. A 34-year-old woman with donor site on the thigh; partially re-epithelialized on the 26th post-operative day, immediately after removing the adherent foam.
Throughout the post-operative follow-up period, the patients were asked about the occurrence of the following donor site problems: pain, unpleasant odor, itching, skin reddening, and appearance of exudates (including pus). Patients also reported the adherence time of the foam.
RESULTS
The characteristics of the study population are documented in Table 1. Figures 1-7 show the results obtained, including the incidence of complications.

There were no detected cases of infection, severe pain, or contact dermatitis in the study population.
DISCUSSION
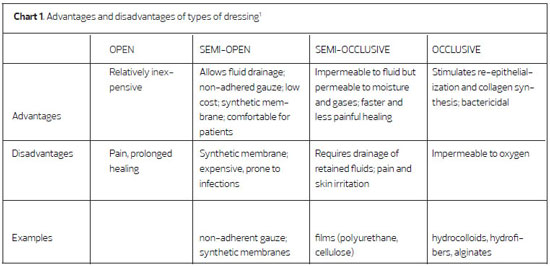
The ideal dressing for a graft donor site is one that holds moisture; is impervious to microorganisms but permeable to water vapor; is free of toxic elements; absorbs exudate; reduces contamination of, and discomfort from, the wound; facilitates re-epithelialization; promotes the quality of the cosmetic results; is easily applied; and has low cost. Dressings are applied at the time of surgery and maintained until re-epithelialization is complete, when they can easily be removed. Recently, an improved understanding of the factors involved in wound healing has stimulated the development of new dressings (chart 1). However, the ideal dressing is still being sought. The choice of the most appropriate dressing may cause confusion; there are many options, and the commercial literature provides many indications for the use of the various dressings1-9.
The most commonly used dressings for treating wounds are occlusive and semi-occlusive, including polyurethane foam films8. Non-adherent gauze (AdapticTM, Johnson & Johnson; XeroformTM, Kendall; Jelonet, Smith & Nephew) is a semi-open dressing made from fabric soaked in petrolatum, which prevents adhesion to the wound bed, and results in an exchange without pain and with tissue protection. These dressings require frequent changes and therefore cause a little more pain than other types of dressings6.
Sodium carboxymethyl cellulose, pectin, and gelatin dressings (DuodermTM, Convatec) are self-adhesive, absorb exudate, and promote a moist environment that helps in the healing of surgical wounds. Polyethylene mesh, impregnated with molecular silver (ActicoatTM, Smith & Nephew) acts as an antimicrobial barrier, and has an open structure that easily allows passage of fluid9. Polyurethane adhesive films (OpsiteTM, Smith & Nephew; TegadermTM, 3M) are transparent dressings that are permeable to water vapor. Although comfortable, these adhesive bandages are expensive, are not easy to apply, and often leak exudate from the wound, and loosen early1,6.
BiobraneTM (Smith & Nephew) is a trilaminate synthetic membrane formed of a thin silicone layer, covering a nylon mesh impregnated with porcine collagen10. Although it allows for observation of the underlying wound, it can be breached by the accumulation of fluid under the dressing, macerating the surrounding skin and becoming infected, and resulting in far more frequent dressing changes, with associated discomfort for patients9.
Carboxymethylcellulose hydrofiber impregnated with silver (Aquacel® Ag, Convatec) and calcium alginate matrix impregnated with silver (Algisite® Ag, Smith & Nephew) have been considered one of the most efficient covers for graft donor sites, notably due to the promotion of rapid re-epithelialization, a large exudate absorption capacity and, in addition to the alginate, hemostatic properties. Unfortunately, these dressings are prohibitively expensive for use in most public hospitals in the country, and it is impossible to suggest their routine use5,6.
Foams are a porous polyurethane polymer matrix. They can be classified as semi-open coverings or as passive dressings. Foams are indicated for deeper wounds, or highly exudative wounds, such as those resulting from the removal of partial-thickness grafts, as used in this study8.
Several authors have observed that most graft donor sites become re-epithelialized, with the resulting detachment of the adherent dressings, by the 14th post-operative day1-4,6; consequently, in the present study, the 14th post-operative day was considered as a cutoff point to classify foam adhesion as extended or not extended. Adherent dressings have been considered safe, easy to use, comfortable for patients, and capable of facilitating re-epithelialization. They are more suitable for use in highly exudative donor sites, due to their increased capacity for absorbing liquids4.
The use of a polyurethane foam with a non-stick film (HydrositeTM, Smith & Nephew) on donor sites of partial-thickness grafts has proved satisfactory; such foams have several advantages over hydrogel dressings, including a reduction in time to re-epithelialization, reduced accumulation of exudate, a requirement for fewer changes, and a reduced likelihood of hypertrophic scar formation. Only pain was better controlled with the hydrogel dressings.3,4 Dressings with a greater capacity for the absorption of exudates help the healing process by maintaining a moist microenvironment on the wound5.
Almost all graft donor sites, regardless of the type of dressing used, are associated with some degree of discomfort, especially during the first 7 days after surgery2,6. Consequently, in the present study, the authors were interested in the prevalence of "intense pain" in the study population. Fortunately, no patient reported this symptom. This is consistent with findings in the literature, in which only mild pain has been reported in graft donor sites, especially when the patient is mobilized, and an adhesive dressing is used2-6.
In the Burn Unit in which the authors work, cellulose films (BioFill®, Bionext®, NexfillTM, Fibrocel; Veloderm®, BTC) is the most widely used dressing for graft donor sites. When moistened, the material becomes transparent and selectively semipermeable, allowing gas exchange and water vapor transmission, but preventing fluid loss and penetration of bacteria. Cellulose films are easy to apply, allow direct observation of the wound, and are associated with reduced pain.
Post-operatively, the cellulose forms a thin blood crust that acts as a protective cover, and is released gradually as the wound re-epithelializes. The resulting film facilitates local wound hygiene, can be wetted without concern, and provides additional coverage, which results in convenience and post-surgical comfort. In the absence of exudate on the wound, the cellulose remains for approximately 7 days, without requiring changes2. The main disadvantage of the product is that it is impervious to liquids, which predisposes its early deployment in donor sites of medium- and large-thickness grafts. Furthermore, the film is expensive, and tears easily, making it difficult to handle. In the present study, the polyurethane foam was chosen with the aim of overcoming these difficulties.
In the present study, the low cost and the absorption of exudate from the graft donor sites were the most notable advantages of the use of foams. When used on smaller donor sites (<200 cm2) and the scalp, foams behaved similarly to cellulose films, detaching within 14 days of use, with the added advantage of preventing the accumulation of liquid on the wound area. In such cases, the early detachment of the foams was attributed to the lower area of adhesion to the de-epithelialized bed and, on the scalp, to the rapid re-growth of hair.
In most cases, however, the use of foam was not encouraging. Foams increased the volume, and thus the work of placing the dressing. They also tended to move from where they were initially placed, until they adhered to the graft donor site, in the first post-operative hours. When used on larger donor areas, a strong odor developed once patients began to wet the foam, presumably due to water retention and proliferation of contaminating microorganisms in the interstices.
In addition, in most patients (73-78%), the foams had become strongly adherent in the late post-operative period, which resulted in prolonged attachment (detachment of the material after the 14th post-operative day). In one case, after the 21st post-operative day, it was evident that the material would not detach; the patient required removal of the foam in the operating room, under general anesthesia (Figure 7). At this time, it was found that the foam had adhered both to the wound area and to the already healed underlying skin. This severe union was attributed to penetration of granulation tissue, such that the epidermis regenerated at the interface of the foam and the donor area. The case eventually resulted in complete re-epithelialization of the donor site, 14 days after the withdrawal of the foam, at which time the patient was discharged without complications.
Despite these problems, all graft donor sites were re-epithelialized within approximately 10-35 days. There were no cases of infection or allergic reaction. Other authors have reported the incidence of infection in adherent dressings to be in the region of 2.7-15%4,5.
Unfortunately, 73% of patients were dissatisfied with the use of foams, whether due to prolonged adherence (73%, n = 8), unpleasant odor (45%, n=5), or lack of re-epithelialization (18%, n=2). Two patients (18%) were satisfied, with particular emphasis on the padding that the foam provided the wound. One patient did not return for the final post-operative evaluation.
CONCLUSIONS
The use of polyurethane foam as a dressing for graft donor sites showed a high rate of complications and patient dissatisfaction, although results were more favorable when used on the scalp or a small donor site.
REFERENCES
1. Kelton Jr PL. Skin grafts and skin substitutes. Read Plastic Surgery. 1999;9:1-26.
2. Ferreira LM, Blanes L, Gragnani A, Veiga DF, Veiga FP, Nery GB, et al. Hemicellulose dressing versus rayon dressing in the re-epithelialization of Split-thickness skin graft donor site: a multicentre study. J Tiss Viab. 2009;18:88-94.
3. Akita S, Akino K, Imaizumi T, Tanaka K, Anraku K, Yano H, et al. A polyurethane dressing is beneficial for split-thickness skin graft donor wound healing. Burns. 2006;32:447-51.
4. Blome-Eberwein, Johnson RM, Miller SF, Caruso DM, Jordan MH, Milner S, et al. Smith. Hydrofiber dressing winth silver for the management of split-thickness donor sites: a randomized evaluation of two protocols of care. Burns. 2010;36:665-72.
5. Ding X, Shi L, Liu C, Sun B. A randomized comparison of Aquacel Ag and Alginate Silver as skin graft donor site dressing. Burns. 2013;39(8):1547-50.
6. Demirtas Y, Yagmur C, Soylemez F, Ozturk N, Demir A. Management of Split-thickness skin graft donor site: a prospective clinical trial for comparison of diferente dressing materials. Burns. 2010;36:99-105.
7. Weinzweig J. Plastic surgery secret plus. 2end. ed. Philadelphia: Mosby Elsevier, 2010.
8. Smaniotto PHS, Ferreira MC, Isaac C, Galli R. Sistematização de curativos para o tratamento clínico das feridas. Rev Soc Bras Cir Plast. 2012;27(4):623-6.
9. Grabb WC, Smith JW. Grabb and Smith's plastic surgery. 6. ed. Philadelphia: Lippincott-Ravens, 2007.
10. Haeney JA, Austin OMB, Phipps AR. Cyanoacrylate tissue glues facilitate the application of Biobrane. Burns. 2005;31:396-7.
1 - Full member of the Brazilian Society of Plastic Surgery - BA, Brazil
2 - Associate of the Brazilian Society of Plastic Surgery, Coordinator of the Burn Treatment Center of the State General Hospital, BA, Brazil
3 - Plastic Surgery Resident
Institution: State General Hospital of Bahi (Hospital Geral do Estado da Bahia).
Corresponding author:
Sandro Cilindro de Souza
Rua Conselheiro Correia de Menezes, 182, Horto Florestal
Salvador-BA, CEP: 40295-030
Tel.: (71) 9977-7303
E-mail: sandrocilin@gmail.com
Article received: November 6, 2013
Article accepted: December 8, 2013


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter