

Original Article - Year 2014 - Volume 29 -
The autologous greater omental flap as a structure for extraperitoneal surgical repair: A comparative, paired, and controlled experimental study of its adaptive properties
Retalho autólogo de grande omento como estrutura cirúrgica de reparo extraperitoneal. Estudo experimental comparativo, pareado e controlado de suas propriedades adaptativas
ABSTRACT
INTRODUCTION: The greater omentum was initially used in the repair of gastrointestinal defects in the 19th century; during the 20th century, it has been used extraperitoneally in the treatment of various disorders, in several surgical specialties. Despite the fact that the greater omentum was studies in detail in the 1960s, there are no reported comparative studies concerning the use of omental flaps extraperitoneally. The present study analyzed the adaptive features of the greater omentum in the extraperitoneal space, with the aim of identifying its surgical applicability.
METHODS: A paired, controlled comparative study was conducted using 20 tissue samples from 5 obese female Sprague-Dawley rats (Rattus norvegicus). The following specimens from each animal were analyzed and compared, macroscopically and microscopically, using the hematoxylin-eosin (HE) technique: (1) omentum without manipulation; (2) intraperitoneally manipulated omentum; (3) extraperitoneally manipulated omentum; and (4) subcutaneous adipose tissue.
RESULTS: Macroscopically, the extraperitoneal omentum exhibited a more intense yellowish color and a higher degree of contraction than the control (intraperitoneal) omentum. The extraperitoneal omentum was similar in color to the adjacent subcutaneous adipose tissue. HE staining revealed a high degree of fibrosis and an average adipocyte size, similar to that in the control omentum, but lower than that in subcutaneous adipose tissue (p< 0.001).
CONCLUSION: The results of this study indicate that the extraperitoneal omentum was not able to promote tissue regeneration, as metaplasia of the translocated flap was not observed in the histological analysis. However, this structure may be used to correct small deformities, in the treatment of ischemic areas, as a carrier structure for surgical reconstruction and as a germination platform for the development of new organs.
Keywords: Omentum; Breast; Reconstruction; Epiploon; Metaplasia; Fat.
RESUMO
INTRODUÇÃO: O grande omento vem sendo utilizado como estrutura de reparo desde o século XIX e a partir do século XX tem sido descrito, em meio extraperitoneal, para o tratamento de diversas afecções em várias especialidades cirúrgicas. Apesar de amplamente estudado a partir da década de 1960, não há descrição de estudos comparativos sobre o seu retalho em meio extra peritoneal. O objetivo do presente estudo foi analisar as características adaptativas do grande omento em meio extra peritoneal para identificar a real aplicabilidade cirúrgica desta estrutura.
MÉTODOS: Estudo experimental comparativo, pareado e controlado de 20 amostras teciduais de ratos (Rattus norvegicus) fêmeas obesas, irmãs da linhagem Sprague-Dawley. De cada animal foram analisados e comparados, macroscopicamente e microscopicamente, através das técnicas de Hematoxilina-eosina (HE) amostras de: (1) omento sem manipulação, (2) omento manipulado intraperitoneal, (3) omento manipulado extraperitoneal e (4) tecido adiposo subcutâneo.
RESULTADOS: omento extraperitoneal, macroscopicamente, apresentou uma coloração amarelado mais intenso, semelhante à gordura subcutânea adjacente, com alto grau de contração se comparado ao omento intraperitoneal de controle. Pela técnica de HE, foi identificado alto grau de fibrose e tamanho médio dos adipócitos semelhante ao omento de controle e inferior ao do subcutâneo (p<0,001).
CONCLUSÃO: O omento extraperitoneal não se mostra capaz de promover regeneração tecidual, uma vez que não foi observado metaplasia à histologia do retalho translocado. Entretanto, pode servir para a correção de pequenas deformidades, para o tratamento de áreas isquêmicas, como estrutura carreadora para a reconstrução cirúrgica e como plataforma germinadora para o desenvolvimento de novos órgãos.
Palavras-chave: Omento; Mama; Reconstrução; Epíploon; Metaplasia; Gordura.
The use of the greater omentum or caul as a surgical repair structure for intra-abdominal organ injury was initially reported in the 19th century1,2. The first successful use of the greater omentum outside the peritoneal cavity was described by Morison in 19033, for the treatment of ascites. In 1937, O'Shaughnessy4 reported the use of an omental pedicle flap in the treatment of angina, and identified the angiogenic properties of this tissue after observing the neovascularization of structures adjacent to the transposed flap. However, this was without knowledge of the mechanisms of angiogenesis.
Attention returned to the extraperitoneal surgical potential of the greater omentum during the 1960s, when successful surgical transposition of the omentum was reported by Kiricuta5 for breast reconstruction, and by Goldsmith6 for the treatment of chronic lymphedema. Since then, the study of the applicability of extraperitoneal omentum has expanded to several other surgical specialties. In orthopedics, the omentum has been used for the repair of bone defects, the treatment of osteomyelitis, prosthetic coverage, and the prevention of adhesions after tenolysis. In thoracic surgery, the omentum has been utilized in chest wall reconstruction and the reconstruction of sternal defects, coverage of esophageal anastomoses, and in the treatment of bronchial and tracheal fistulas, diaphragmatic hernias, and mediastinal infections. Vascular surgeons have utilized the omentum in the treatment of chronic lymphedema, to protect vascular and prostheses anastomoses, in the revascularization of ischemic limbs, and the treatment of gangrene. In the field of oncology, omental tissue has been used in the treatment and prevention of injury resulting from radiation therapy. Neurosurgical uses of the omentum include bypassing areas of brain ischemia, in the treatment of CSF leaks, in epilepsy and in Alzheimer's disease. The omentum is used in urology and gynecology in the treatment of urinary and gynecological fistulas, and in cardiac surgery, for coronary artery bypass grafting. In plastic and reconstructive surgery, the omentum is used in the treatment of various regions of the body5-11.
Studies of the omentum, mainly undertaken over the last 50 years, have focused on its characteristics in terms of its anatomical, physiological, metabolic, angiogenic, immunogenic, and molecular properties10,12-16. Several studies have compared and contrasted the characteristics of adipose tissue from the greater omentum with subcutaneous adipose tissue17-21. However, there are no comparative studies focusing on the adaptive properties of the greater omentum in the middle extraperitoneal region. It is established that transposed omentum vascularizes the receptive site and fills defects. However, it is unclear whether it undergoes metaplasia, conferring the ability to functionally regenerate damaged structures from its vast concentration of stem cells, or whether it undergoes a more simple adaptive process while maintaining the same tissue and function as it has intraperitoneally22. This information is of great importance, as it should indicate how best to use omental tissue, and also improve the ability to predict the results of areas repaired with this structure. Knowledge of the adaptive properties of the extraperitoneal greater omentum may also serve as a basis for future studies, in which omental tissue could be used as a structural framework and means of transportation for tissue regeneration, and even the reconstruction of new organs.
On the basis of these features of extraperitoneal omentum, and its great potential for the future, we undertook a paired comparative experimental study in rats, designed to investigate the structural, anatomical, and histological changes that occur when an omental flap is translocated into the subcutaneous tissues, and assess its potential for metaplasia. The aim of this study was therefore to analyze the adaptive features of extraperitoneal greater omental tissue, and to identify the surgical applications of this structure.
METHODS
Study design and sample size
This was a comparative experimental study, in which subjects were matched and controlled. Tissue samples of female obese Sprague-Dawley rats (n=5, Rattus norvegicus) were used. The rats were obtained at 56 days of age, from the Central Breeding Center of the State University of Campinas (UNICAMP). Obesity was induced to provide greater amount of omental tissue, and enhance the immunohistochemical identification of different adipose tissues, using anti-CD68 labeled macrophages. Obesity was achieved through the administration of the cafeteria diet for an 8-week period. This diet replaces standard rodent food with calorific foods consumed by the human population, such as chocolate, peanuts, bacon, condensed milk, cake, cream, soda, as recommended by Vanzela in 201023.
This study was approved by the Ethics Committee on Animal Experimentation (ECAE) of the Institute of Biology (IB), UNICAMP, and all rules relating to the ethical aspects established for the use of animals in research were followed. From the beginning of the fattening period until harvest for analysis materials, rats remained in the animal house of the IB UNICAMP, isolated in boxes with daily cleaning care and illumination (12hours/day) and a constant temperature (20-22ºC), and submitted to the cafeteria diet ad libitum.
Surgical procedure
All procedures for translocation of the greater omentum were performed by two researchers. When the rats were 112 days old (weight, 323.8± 4.2g), general anesthesia was induced with an intraperitoneal injection of 0.5ml ketamine (Vetaset® Fort Dodge, 1 g/10 ml) and xylazine (Rompun® Bayer, 2g/10ml) in a 3:2 ratio, as per UNICAMP IB protocol. A maintenance dose was not required, since the total procedure time was 18±5 minutes.
The procedure for translocation of the omentum was performed after trichotomy, antisepsis with polyvinyl pyrrolidone iodine (1% active iodine) and asepsis. The following procedures were conducted as shown in Figure 1: (1) a skin incision of approximately 4cm was made in the midline; (2) dissection of the subcutaneous tissues of the right hemi-abdomen; (3) laparotomy incision through the abdominal muscles, 4 cm in length; (4) identification and exteriorization of the greater omentum of the abdominal cavity; (5) division of the greater omentum into 3 parts; a left pedicle flap (20mm2) on the left gastroepiploic branch, remained in the abdominal cavity (control); a right flap (20mm2) on the right gastroepiploic branch, translocated into the subcutaneous area through the right hemi-abdomen, and attached at the distal portion to the abdominal wall with a nylon 5.0 wire, for future identification. The remainder of the omentum was photographed and collected for analysis; (6) the muscle was closed with continuous stitches using nylon 5.0 wire, with the exception of the upper 0.3 cm, which was left open for the passage of the translocated omentum pedicle; (7) the skin was closed with continuous reversed nylon 5.0 wire stitches, and covered with micropore paper tape. Following the procedure, the rats were placed in the lateral position under thermal lamps for anesthetic recovery.
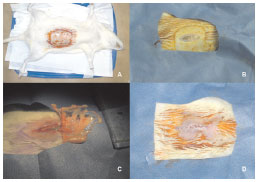
Figure 1. The translocation process. A - Rat positioned during general anesthesia; B - After antisepsis and positioning of fenestrated sterile field, an incision is made in the midline of the abdomen to access the cavity. C - The omentum is exposed and 2 flaps (2 cm each) were made. One of the flaps was transposed into the subcutaneously; the other remained in the abdominal cavity, and served as a control. The remaining omental tissue was resected and used as a sample of "unhandled omentum." D - After translocation, the incision was closed in the muscular plane; a 1-cm opening at the upper area allowed maintenance of pedicle blood flow. The skin was closed completely with continuous inverted sutures.
Preparation and macroscopic anatomical analysis
Eight weeks after surgery (168 days old; mean weight, 351 ± 26.92g), the rats were euthanized. At this time, 3 tissue samples were identified, photographed, and resected from each rat: the control omental flap, the translocated omental flap, and a sample of subcutaneous adipose from the left iliac fossa. The left iliac fossa was chosen as it is an area with abundant adipose tissue, distant from the site of omental translocation, and without local influence on inflammatory processes.
A macroscopic comparison of tissues, with reference to contraction and extent of staining, was performed by the 2 researchers who had conducted the translocation procedure, and removed the tissue samples after euthanasia. As both the translocated and control flap had a pedicle, it was not possible to accurately measure the volume during the translocation process. Quantitative measurement of macroscopic contraction of the omental flaps, and volumetric loss of these structures, could not be carried out, although all flaps were of the same initial size (20mm2).
Preparation and histological analysis
A microscopic analysis of the omental flaps was made by a pathologist, blinded to tissue origins; the obtained samples were identified only by numbers and letters. Only the researchers who performed the procedures of preparation and extraction knew the origin of the analyzed parts. Each collected sample was immediately identified and introduced in a bottle with 10% buffered formalin.
In the pathology laboratory, the tissue samples were processed; a slide was made from each sample and stained using the hematoxylin-eosin method (HE) to evaluate the following: (1) fibrosis, (2) adipocyte diameter, and (3) the presence of different cell populations to those usually present in the omentum, i.e. metaplasia.
Fibrosis was analyzed by a pathologist, using a simple optical microscope. To analyze the diameter of adipocytes, 100× images were captured using a digital camera (Leica DFC360 FX, Solms, Germany) connected to a clear field microscope (Leica DM5000 B). These images were evaluated by using image analysis software (Leica Qwin Standard V3 Imaging Microsystems), to measure the diameter of 100 random adipocytes, counted by a semi-automated procedure in the program.
Statistical analysis
Data from each rat and from each tissue sample collected were analyzed statistically. Changes in the rat's weight, and the average diameter of adipocytes, were analyzed using the Student t-test, paired in independent pairs. The significance level was 5% and the software used for analysis was SAS version 9.2.
RESULTS
Macroscopic anatomical analysis
At 8 weeks after the translocation the average weight of the rats increased from 323.8 ± 34.16 g to 351.0 ± 26.92 g (p = 0.012, 95% CI -44.68 to -9.72; Figure 2). During this period, complete healing of the surgical wound was observed, with full hair growth on the site (Figure 3A).
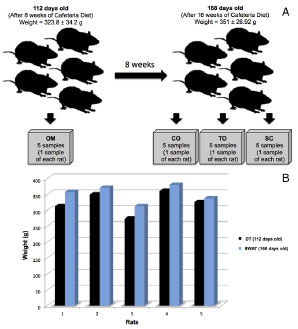
Figure 2. A - After 8 weeks of cafeteria diet, rats (112 days of age; mean weight, 323.8 ± 34.2 g) underwent surgery for omental translocation to a subcutaneous site. At this time, a sample of omentum (OM) from each mouse was identified, extracted, and photographed. At 8 weeks post-surgery, the rats were euthanized (168 days of age; mean weight, 351 ± 26.92 g). Three tissue samples were identified, photographed, and extracted: (i) postoperative omental control flaps (CO); translocated omentum (TO); and a fragment of subcutaneous adipose tissue (SC) from the left iliac fossa. B - Graph depicting weight progression of each mouse from translocation (DT) until 8 weeks post-translocation (8WAT). The average weight increased significantly during the 8-week postoperative period.
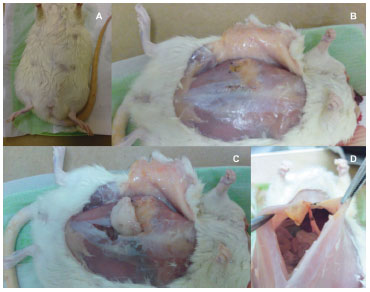
Figure 3 (A) - A wound, showing complete healing 8 weeks following surgery. B - Translocated omentum set in the right upper quadrant of the abdomen. C - Coloration of the translocated omentum, similar to that of subcutaneous adipose tissue, but different to that of control omentum. D - Confirmation of pedicle integrity. a região abdominal.
Macroscopically, omental tissue translocated into the subcutaneous environment was a more intense yellowish color, similar to adjacent subcutaneous adipose tissue (Figures 3B and C); this was in contrast to the lighter shade observed at the time of translocation, and still present in the intra-abdominal control omental flaps (Figure 3C).
Regarding the degree of contraction, the translocated omentum showed severe contraction, compared with the intra-abdominal omental control; the contracted omentum was thin, with an elastic-fibrous consistency (Figure 3C). The postoperative pedicle integrity was confirmed (Figure 3D).
to accurately measure the volume during the translocation process. Quantitative measurement of macroscopic contraction of the omental flaps, and volumetric loss of these structures, could not be carried out, although all flaps were of the same initial size (20mm2).
Preparation and histological analysis
A microscopic analysis of the omental flaps was made by a pathologist, blinded to tissue origins; the obtained samples were identified only by numbers and letters. Only the researchers who performed the procedures of preparation and extraction knew the origin of the analyzed parts. Each collected sample was immediately identified and introduced in a bottle with 10% buffered formalin.
In the pathology laboratory, the tissue samples were processed; a slide was made from each sample and stained using the hematoxylin-eosin method (HE) to evaluate the following: (1) fibrosis, (2) adipocyte diameter, and (3) the presence of different cell populations to those usually present in the omentum, i.e. metaplasia.
Fibrosis was analyzed by a pathologist, using a simple optical microscope. To analyze the diameter of adipocytes, 100× images were captured using a digital camera (Leica DFC360 FX, Solms, Germany) connected to a clear field microscope (Leica DM5000 B). These images were evaluated by using image analysis software (Leica Qwin Standard V3 Imaging Microsystems), to measure the diameter of 100 random adipocytes, counted by a semi-automated procedure in the program.
Statistical analysis
Data from each rat and from each tissue sample collected were analyzed statistically. Changes in the rat's weight, and the average diameter of adipocytes, were analyzed using the Student t-test, paired in independent pairs. The significance level was 5% and the software used for analysis was SAS version 9.2.
RESULTS
Macroscopic anatomical analysis
At 8 weeks after the translocation the average weight of the rats increased from 323.8 ± 34.16 g to 351.0 ± 26.92 g (p = 0.012, 95% CI -44.68 to -9.72; Figure 2). During this period, complete healing of the surgical wound was observed, with full hair growth on the site (Figure 3A).
Macroscopically, omental tissue translocated into the subcutaneous environment was a more intense yellowish color, similar to adjacent subcutaneous adipose tissue (Figures 3B and C); this was in contrast to the lighter shade observed at the time of translocation, and still present in the intra-abdominal control omental flaps (Figure 3C).
Histological analysis
Under the microscope, it was apparent that all samples were from viable flaps (no necrosis). In the translocated omentum, there was a higher degree of fibrosis, compared with all other samples. No tissue types, besides those customarily present in the omentum, were identified in flaps samples (Figure 4).
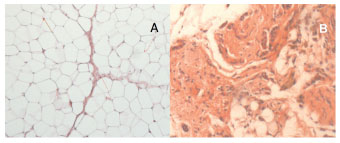
Figure 4 (A)- Microscopic analysis to measure the diameter of 100 random adipocytes, and the histological appearance of the unhandled omentum (OM) (HE, 100×). (B) - Histological appearance of the translocated omentum (TO), showing a high degree of fibrosis (HE, 100×).
Morphological assessment was used to ascertain the average diameter of omental adipocytes (0.059 µm), subcutaneous adipocytes (0.071 µm), translocated omental adipocytes (0.064 µm), and adipocytes from intra-abdominal omentum / control (0.065 µm) (Table 1). Unhandled omental adipocytes had a smaller diameter than those of subcutaneous adipose tissue (p < 0.001; 95% CI, -0.016 to -0.010). There was a statistically significant difference between the diameter of translocated omental adipocytes and the unhandled omental adipocytes (p = 0.0014; 95% CI, -0.008 to -0.003), and those from subcutaneous adipose tissue (p < 0.001; 95% CI, 0.004 to 0.010). Adipocytes from the translocated omental flap did not differ significantly from the control (p = 0.27) . (Figure 5).
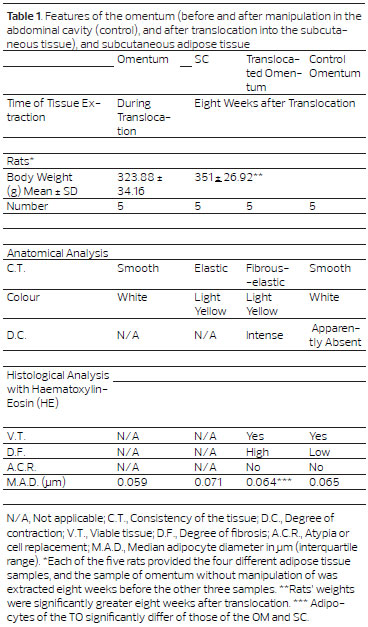
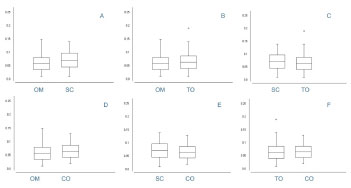
Figure 5. Average diameter of adipocytes (Student t-test for independent variables). A - Unhandled omentum vs. subcutaneous adipose tissue, p < 0.0001 B - Translocated omentum vs. unhandled omentum, p < 0.0001. C - Translocated omentum vs. subcutaneous adipose tissue, p < 0.0001. D - Unhandled omentum vs. control omentum, p < 0.0001. E - Control omentum vs. subcutaneous adipose tissue, p < 0.0001. F - Control omentum vs. translocated omentum, p = 0.27.
DISCUSSION
Despite our detailed knowledge of the greater omentum and its widespread clinical applicability, in various medical specialties, there is a lack of comparative studies focusing on the adaptive properties of extraperitoneal omentum. The majority of studies that have examined the surgical and therapeutic potential of extraperitoneal flaps of the greater omentum have focused either on the recipient tissue or on the treated anatomic structures and the mechanisms involved6-11,14,16.
The present study focused on evaluating the adaptive properties of translocated extraperitoneal omentum. It was therefore possible to compare important tissue changes, not previously reported in the literature. Regarding the postoperative time before collection of material for analysis i.e. 8 weeks after the omental translocation, the skin scar showed complete healing, with the regrowth of hair in the abdominal region (Figure 3A). This indicates that complete regeneration had occurred during the 8-week postoperative period, which is more than twice the duration of rat gestation (3 weeks), and is considered sufficient for subsidence of any inflammatory response to the procedure (including confirmed low concentration of macrophages by immunohistochemistry) and complete adaptation or metaplasia of the transposed flap.
Considering the anatomical evaluation, the translocated omentum showed the same characteristics as adjacent subcutaneous adipose tissue, with a light yellow color, a dense appearance, and an elastic-fibrous consistency; this is distinct from the control omentum, which retained the same features as the unhandled omentum. These findings suggest contraction of the greater omentum, possibly resulting in lack of peritoneal fluid, may be a contraindication to the use of this structure as a fill tissue or for volumetric gain. However, the high degree of fibrosis and the elastic-fiber consistency resulting from the extraperitoneal translocation may be useful for the treatment of deformities and repairs that require structural support or coverage without volume. The omentum can also be effectively used as a tissue or cell carrier, for tissues or organs in almost all body regions12,16.
With regard to the HE stained histological analysis, the translocated omentum showed the same tissue pattern and the same cell morphology as control adipocytes, differing only in terms of the presence of moderate fibrosis. The absence of peritoneal fluid is apparently the main factor responsible for these adaptive changes to the omentum in the subcutaneous space. The contraction of the translocated omentum, and the absence of contraction in the control omentum may be due to dehydration and greater fibrosis18-21,24.
As the subcutaneous adipose tissue is known to be different from the greater omentum17-21, it was expected that the mean diameter of adipocytes in these two tissues would differ; however, the influence of translocation on an omental flap had previously been described. Data regarding differences between adipocytes of the greater omentum and the subcutaneous adipose tissue were statistically confirmed in the present study.
Regarding the comparison between the translocated flap and the other tissue samples, it was identified that the average diameter of adipocytes in the translocated omentum was similar to that of the control omental flap, but significantly different to that of the unhandled omentum and subcutaneous adipose tissue. These results suggest that the peritoneal fluid may predominantly influence the extracellular matrix of the omentum and its population of floating cells, with little effect on adipocytes. The two handled flaps had the same average adipocyte diameter, which was significantly smaller than that of adipocytes found in the subcutaneous adipose tissue. This suggests that the observed anatomical transformation stems mainly from changes in the extracellular matrix of the omental adipose tissue, and that there is no indication of replacement in the cell population by subcutaneous adipocytes. The statistically significant difference between the diameter of omental adipocytes without manipulation that was smaller than the diameter of the adipocytes in the omental flaps (control omentum flaps and translocated flaps) may have resulted in the significant fattening of animals, which occurred during the 8 weeks between the initial procedure and euthanasia of the rats (Table 1).
The greater omentum is an omnipotent source of stem cells (cells capable of differentiating into other cell types of the same embryonic origin) and has great angiogenic potential, restoring ischemic tissues and actinic lesions 3,6,8,10,14,16. Translocated flaps are in a different location to the site of origin, and may therefore be exposed to metaplasia, by the same principles observed in Barrett's esophagus or in metaplasia of intraoral skin flaps; hence, it was important to investigate whether there is substitution of the transposed tissue cell type. The results of our histological assessment did not show the presence of cell metaplasia in the translocated omental flap; no atypical tissue type was observed, and there was no change in adipocyte diameter between paired samples, when compared with the control omentum.
In mammals, the adipose tissue of the subcutaneous tissues, bone marrow, and intra-abdominal cavity have structural and functional differences, and distinct characteristics, despite being mainly formed of adipocytes25,26. In the present study, morphological assessment indicated that the extraperitoneal omental adipocyte was smaller than the adipocytes of other samples. Consequently, it can be inferred that these adipocytes partially lost their functional capacity or energy reserve, and started to serve a fill tissue function, probably similar to adipose tissue of the bone marrow. This is likely due to tissue adaptation. Therefore, the extraperitoneal omental flap does not seem to be able to transform the recipient tissue, despite its high concentration of omnipotent stem cells and its great angiogenic potential.
The findings of this study demonstrate that the omentum, similar to subcutaneous adipose tissue, is a rich source of material that can be used in the reconstruction of various organs and body structures, as a tissue filler and support, without any apparent ability to regenerate organ or tissue function27,28. In addition, the angiogenic potential of the omentum allows the revascularization of ischemic areas, such that the translocated omentum could serve as a carrier structure for surgical reconstruction, and a germinating platform for the development of new organs16,29,30. However, omental tissue appears inappropriate, when used alone, for large reconstructions, tissue regeneration, or volume replacement.
CONCLUSION
In conclusion, in the subcutaneous environment, the translocated omentum undergoes contraction and volume loss. Although translocated omentum appears macroscopically similar to subcutaneous adipose tissue, microscopically it maintains the same structural and morphological pattern of adipocytes present in the intra-abdominal environment, with no signs of metaplasia. Therefore, the extraperitoneal omentum appears incapable of promoting tissue regeneration, although it could be used in the correction of small deformities, for the treatment of ischemic areas, as a carrier structure for surgical reconstruction and as a germinating platform for the development of new organs.
REFERENCES
1. de Lamballe AJJ. Memóire sur les plaies du canal intestinal. Paris, 1826.
2. Senn N. An experimental contribution to intestinal surgery, with special reference to the treatment of intestinal obstruction. Ann Surg. 1888;7(6):421-30.
3. Morison R. A case of ascites due to liver cirrhosis treated by operation. Ann Surg. 1903;38(3):361-6.
4. O'Shaughnessy L. Surgical treatment of cardiac ischaemia. Lancet 1937;1:185-94.
5. Kiricuta I. L'emploi du grand épiploon dans la chirurgie du sein cancereux. Presse Medicale 1963;71:15-7.
6. Goldsmith HS, De los Santos R, Beattie Jr EJ. Relief of chronic lymphedema by omental transposition. Ann Surg. 1967;166(4):573-85.
7. Liebermann-Meffert DM, Siewert JR. The role of the greater omentum in intrathoracic transposition. Neth J Surg. 1991;43(5):154-60.
8. Williams RJ, White H. Transposition of the greater omentum in the prevention and treatment of radiation injury. Neth J Surg. 1991;43(5):161-9.
9. Normington EY, Papay FA, Yetman RJ. Treatment of recurrent cerebrospinal fluid rhinorrhea with a free vascularised omental flap: A case report. Plast Reconstr Surg. 1996;98(3):514-9.
10. Liebermann-Meffert D. The greater omentum. Anatomy, embryology, and surgical applications. Surg Clin North Am. 2000;80(1):275-93.
11. Hultman CS, Carlson GW, Losken A, Jones G, Culbertson J, Mackay G, et al. Utility of the omentum in the reconstruction of complex extraperitoneal wounds and defects: donor-site complications in 135 patients from 1975 to 2000. Ann Surg. 2002;235:782-95.
12. Das SK. The size of the human omentum and methods of lengthening it for transplantation. Br J Plast Surg. 1976;29(2):170-74.
13. Agca B, Paksoy M, Polat E, Aksin E, Dirican A, Durgun V, Eren D. Influence of omentectomy on peritoneal defense mechanisms in an experimental model of intra-abdominal infection. Eur Surg Res. 2003;35(1):35-40.
14. Oloumi MM, Derakhshanfar A, Molaei M, Tayyebi M. The angiogenic potential of autogenous free omental graft in experimental tibial defects in rabbit: Short-term preliminary histopathological study. J Exp Anim Sci 2006;43(3):179-87.
15. Collins D, Hogan AM, O'Shea D, Winter DC. The omentum: anatomical, metabolic, and surgical aspects. J Gastrointest Surg. 2009;13(6):1138-46.
16. de Figueiredo JC, Naufal RR, Claro Jr F, Arias V, Bueno Pereira PR, Inaco Cirino LM. Prefabricated flap composed by skin and terminal gastromental vessels. Experimental study in rabbits. J Plast Reconstr Aesthet Surg. 2010;63(6):e525-8.
17. Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112(12):1796-808.
18. Harman-Boehm I, Blüher M, Redel H, Sion-Vardy N, Ovadia S, Avinoach E, et al. Macrophage infiltration into omental versus subcutaneous fat across different populations: effect of regional adiposity and the comorbidities of obesity. J Clin Endocrinol Metab. 2007;92:2240-47.
19. Dicker A; Astrom G; Wahlen K; Hoffstedt J; Naslund E; Wiren M; Ryden, et al. Primary differences in lipolysis between human omental and subcutaneous adipose tissue observed using in vitro differentiated adipocytes. Horm Metab Res 2009;41(5):350-5.
20. Toyoda M; Matsubara Y; Lin K; Sugimachi K; Furue M. Characterization and comparison of adipose tissue-derived cells from human subcutaneous and omental adipose tissues. Cell Biochem Funct. 2009;27(7):440-7.
21. Aron-Wisnewsky J, Tordjman J, Poitou C, Darakhshan F, Hugol D, Basdevant A, et al. Human adipose tissue macrophages: m1 and m2 cell surface markers in subcutaneous and omental depots and after weight loss. J Clin Endocrinol Metab. 2009;94(11):4619-23.
22. Kamei Y, Toriyama K, Takada T, Yagi S. Tissue-engineering bone from omentum. Nagoya J Med Sci. 2010;72(3-4):111-7.
23. Vanzela EC, Ribeiro RA, de Oliveira CA, Rodrigues FB, Bonfleur ML, Cet al. Pregnancy restores insulin secretion from pancreatic islets in cafeteria diet-induced obese rats. Am J Physiol Regul Integr Comp Physiol. 2010;298(2):320-8.
24. Bassols J; Ortega FJ; Moreno-Navarrete JM; Peral B; Ricart W; Fernandez- Real JM. Study of the proinflammatory role of human differentiated omental adipocytes. J Cell Biochem. 2009;107(6):1107-17.
25. Baglioni S, Francalanci M, Squecco R, Lombardi A, Cantini G, Angeli R, et al, Luconi M. Characterization of human adult stem-cell populations isolated from visceral and subcutaneous adipose tissue. FASEB J. 2009; 23(10):3494-505.
26. Dhanasekaran M, Indumathi S, Kanmani A, Poojitha R, Revathy KM, Rajkumar JS, et al. Surface antigenic profiling of stem cells from human omentum fat in comparison with subcutaneous fat and bone marrow.Cytotechnology. 2012;64:497-509.
27. Claro F Jr, Figueiredo JC, Zampar AG, Pinto-Neto AM. Applicability and safety of autologous fat for reconstruction of the breast. Br J Surg. 2012;99(6):768-80.
28. Illouz YG. Present results of fat injection. Aesthetic Plast Surg. 1988;12(3):175-81.
29. Kobayashi T, Aomatsu Y, Iwata H, Kin T, Kanehiro H, Hisanga M, et al. Survival of microencapsulated islets at 400 days posttransplantation in the omental pouch of NOD mice. Cell Transplant. 2006;15(4):359-65.
30. Baumert H, Simon P, Hekmati M, Fromont G, Levy M, Balaton A, et al. Development of a seeded scaffold in the great omentum: feasibility of an in vivo bioreactor for bladder tissue engineering. Eur Urol. 2007;52(3):884-90.
1- Master - Plastic Surgeon Member of the Brazilian Society of Plastic Surgery
2- Ph.D. - Pathologist at UNICAMP
3- Medical General Surgeon - Plastic Surgery Resident of Plastic Surgery Institute of Santa Cruz
4- Ph.D. - Supervisor of the Specialized Pathology Laboratory - CAISM - UNICAMP
5- Full Professor - Department of Gynecology and Obstetrics of CAISM - UNICAMP
Institution: 1 - Department of Tocogynecology of the Medicine School of the State University of Campinas (UNICAMP). 2 - Institute of Plastic Surgery of Santa Cruz (IPSSC).
Corresponding Author:
Francisco Claro Júnior
Avenida Indianópolis, 2244 - Indianópolis
São Paulo - SP - CEP.: 04062-002
E-mail: fclarojr@gmail.com
Article received: October 25, 2013
Article accepted: December 8, 2013


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter