

Original Article - Year 2013 - Volume 28 -
Choosing the augmentation mammaplasty technique: a method for preventing medical disputes
Escolha da técnica de mastoplastia de aumento: uma ferramenta na prevenção de litígio médico
ABSTRACT
BACKGROUND: Plastic surgery is a medical specialty that is particularly at risk of unwarranted claims. Most complaints regarding plastic surgery are not a consequence of technical failures but rather of inadequate criteria of patient selection, failure in surgical indication, and lack of effective communication between the patient and the surgeon. Training, careful use of techniques, adopting precautions, compliance with safety regulations, and the informed consent document are fundamental principles of defensive medical practice. Moreover, documenting objective information about the surgery in the medical records validates the work of the plastic surgeon and ensures that procedures are productive and technical. The aim of this study was to develop a tool for preventing medical litigations in augmentation mammaplasty based on data expressed on the scale from I to IV.
METHODS: Forty patients with an indication for augmentation mammaplasty were evaluated. All the patients were subjected to anatomical measurements, which were photographically documented, and the hereditary and congenital diseases of the patients presenting for breast augmentation were recorded. In addition, all the patients were informed about the indication and surgical procedure to be performed. They agreed with the surgical indication and provided informed consent.
RESULTS: The follow-up period was 6 months. All the patients expressed a high degree of satisfaction with the results obtained after the procedure, and no cases of complaints or disputes were encountered.
CONCLUSIONS: The authors propose the regular use of tables and classifications for the selection of implants, with the aim of establishing surgical indication as an objective parameter, facilitating patients' understanding of the procedure, and preventing litigation.
Keywords: Breast implantation. Mammaplasty. Jurisprudence.
RESUMO
INTRODUÇÃO: A cirurgia plástica é uma das especialidades médicas expostas ao risco de reivindicações abusivas. A maioria das queixas em cirurgia plástica não decorre de falhas técnicas, mas de inadequados critérios de seleção de pacientes, falha na indicação cirúrgica e falta de comunicação adequada entre o paciente e o cirurgião. O treinamento, a utilização cuidadosa de técnicas, a adoção de precauções, o cumprimento de normas de segurança e o formulário de consentimento informado são princípios fundamentais na prática da medicina defensiva. Ademais, ter nos registros dos pacientes informações objetivas sobre a cirurgia realizada avaliza o trabalho do cirurgião plástico, tornando os procedimentos profícuos e técnicos. O objetivo deste estudo é descrever uma ferramenta de prevenção de litígios médicos na mastoplastia de aumento baseada em dados de escala em graus I a IV.
MÉTODO: Foram avaliadas 40 pacientes com indicação de mastoplastia de aumento. Todas as pacientes foram submetidas a mensurações anatômicas, documentação fotográfica e registro de doenças hereditárias e congênitas encontradas nas pacientes candidatas a aumento mamário. Além disso, todas as pacientes receberam explicação da indicação e do procedimento cirúrgico a ser realizado.As pacientes concordaram com a indicação cirúrgica e assinaram o termo de consentimento.
RESULTADOS: O tempo de seguimento das pacientes foi de 6 meses. Todas as pacientes apresentaram alto grau de satisfação com o resultado obtido após o procedimento e não houve caso de queixa ou litígio.
CONCLUSÕES: Os autores propõem o uso regular de tabelas e classificações para escolha prévia dos implantes, com o objetivo de tornar a indicação cirúrgica objetiva, facilitar o entendimento da paciente e prevenir o litígio.
Palavras-chave: Implante mamário. Mamoplastia. Jurisprudência.
Breast plastic surgery is probably the medical specialty that has been most affected by the social changes that occurred during the last 20 years. This important fact led plastic surgeons to review their statistical data and rethink the concepts regarding patient expectations and needs1,2. In addition, modifications to the surgical indications for breast augmentation, reduction, and mastopexy were introduced.
In the early 1980s, mastopexy procedures made up approximately 60% of breast plastic surgeries; when considering reduction mammaplasties, the percentage reached 75%, which demonstrates the sociocultural need and desire on the part of women of that decade to undergo breast reduction or augmention1,2. Meanwhile, changes in social habits, combined with the globalization of information, popularization of computers and the Internet, and access to safer breast implants, led to changes in the expectations regarding the appearance of body shape. Patients then began primarily wanting breast augmentation with implants or in combination with mastopexy.
These data can be confirmed by analyzing mammaplasty-related publications in the last 45 years in the PubMed database. From 1966 to 1992, 15 to 20 articles were published per year; this number increased to 260 in 1992 and reached 650 publications at present.
In general, aesthetic surgical procedures are increasing. In particular, breast augmentation with silicone implants is currently a widely performed surgical procedure because it is simple, quick, and much sought after. However, as a result, medicolegal requests and litigation cases have also increased3. Similarly, mammaplasty has become an important part of aesthetic surgery but has led to an increase in the number of cases of surgical error. Many of these cases led to judicial proceedings, from which plastic surgeons were rarely dismissed. These increases in the risks of complications, errors, and lawsuits are probably due to the inexperience of many surgeons who succumbed to the pressures and demands of the media and patients, the insufficient training that new plastic surgeons receive, the invasion of other medical specialties that now perform the procedure, or inadequate patient counseling3. Although implants have evolved, results are still not considered ideal in many cases because of the lack of definition of objective parameters for surgical indication and the large number of manufacturers offering a variety of guidelines regarding the shape and volume of implants.
In the technical description of breast implants presented by Mathes4, measurements of the native breast are provided - namely the thoracic diameter, and interaxillary and midsternal distances. In addition, the width, projection, height, and volume of the implant are evaluated. An implant slightly smaller than the native breast is usually selected. Bozola et al.5 performed a geometrical evaluation of the shape of an ideal breast and that of an implant based on Phi proportion. The authors marked the midsternal vertical line to the breast meridian line (BML) for each breast, the horizontal inferior breast line (HIBL), and the horizontal superior breast line (HSBL), as well as the right and left I points, at the intersection between BML and HIBL. The base of the resultant breast was placed between the two horizontal lines (in frontal view) and the vertical lateral breast line (VLBL) e vertical medial breast line (VMBL). The implants were selected according to the vertical measurement of the midsternal line and the hypomastia classification proposed in the study. According to the authors, not many articles with sufficient information on implant shape have been published.
According to Paula6, the choice of breast implant volume should be based on patient preference, area limitations, and - most importantly - on the measurement of the base of the breast subtracted by the pinch test value in the superior pole. The author assessed the indications for anatomical profile when the patients exhibited a distance of 18 cm to 20 cm from the clavicle to the nipple and for natural profile when the patient requested a larger projection in the mid lower area of the breast. In the forum on augmentation mammaplasty that was published in Revista Brasileira de Cirurgia Plástica (Brazilian Journal of Plastic Surgery), the indications observed were mainly for round polyurethane-covered implants7. In addition, the choice of an implant depended on the existing breast volume and on patient preference; however, the indication was not standardized and the evaluation was subjective.
The issue for these authors and for plastic surgeons in general is to perform assessments and surgical indications that are more objective with regard to the shape and volume of the implant. The aim is to interpret the liability of plastic surgeons regarding aesthetic medical procedures as an obligation of means and to elevate the specialty in an equivalent level as that of the other fields of medicine to avoid the judicial understanding of the procedure as an obligation of results8.
The aim of the present prospective study was to describe a series of cases with surgical indication for augmentation mammaplasty and/or mastopexy based on the objective assessment performed using a numerical scale from I to IV, as well as to evaluate the final results based on patients' degree of satisfaction and absence of medical litigation. Moreover, the study aimed to provide data that can support the requirement for plastic surgeons to inform patients and describe the preoperative evaluation of the anatomical findings and the surgical indication in the patients' medical records and informed consent document.
METHODS
In this study, we preoperatively determined the size of silicone breast implants for 40 women, whose ages ranged from 20 and 45 years. The patients provided informed consent and authorized the publication of the images herein.
The marking technique was performed as described by Mathes4, with optimization by the authors. The measures were presented in a table, which permitted the quick and concise visualization of each case and the selection of the implant profile and diameter. The patients were divided into groups I to IV, according to the breast deformity.
All the patients who participated in this study underwent clinical examination and laboratory tests such as complete blood counts; coagulogram; and fasting glucose, glutamicoxalacetic transaminase, glutamic-pyruvic transaminase, alkaline phosphatase, urea, and creatinine levels. In addition, imaging studies were performed, such as resting echocardiography, thoracic radiography in the posteroanterior and lateral (right) positions, and breast ultrasonography.
The following were the exclusion criteria: women with abnormal results in the above-mentioned tests, presence of a nodule or cyst, and increased thickness of breast tissue that suggests previous disease.
The inclusion criteria were as follows: all women with normal clinical examination, laboratory, and imaging test results, aged between 20 and 45 years.
After a clinical history review, a clinical evaluation, and laboratory and imaging tests, some thoracic measurements were obtained using a measuring tape as follows:
1A - diameter of the thorax passing over the nipples (Figure 1);
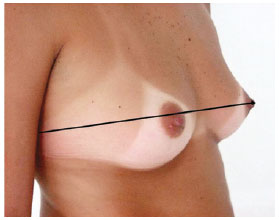
Figure 1 - Measurement 1A, which corresponds to the diameter of the thorax.
1B - distance between the 2 anterior axillary lines (Figure 2);
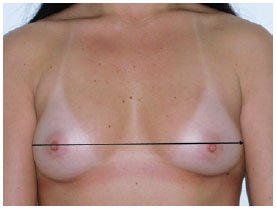
Figure 2 - Measurement 1B, which corresponds to the distance between the 2 anterior axillary lines.
1C - sternal length (Figure 3).
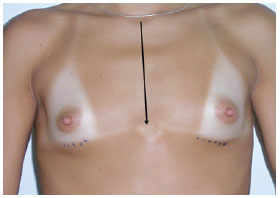
Figure 3 - Measurement 1C, which corresponds to the distance from the notch to the sternal manubrium.
These measures were documented in the patients' medical records and were helpful in the selection of the profile and diameter of the base of the implants.
The following measurements described by Mathes4 were also obtained:
Measurement A - determined using a pachymeter (Figure 4); corresponds to the width of the base of the breasts; useful for selecting the diameter and height of the implant to be used (Figure 5). The manufacturers of breast implants supply the numerical value of the diameter of the implant, depending on the profile type. When calculating breast volume, there may be a discrepancy between the sides of the patient's thorax as a result of the difference in breast size. The recommended size of the base of the implant is a least 1 cm smaller than the base of the breast;
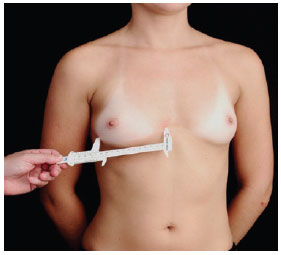
Figure 4 - Measurement performed using a pachymeter.
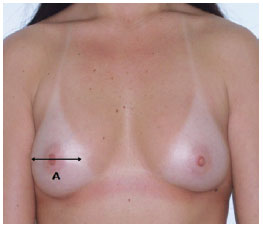
Figure 5 - Measurement A, which corresponds to the base of the breast, is useful for selecting the diameter and height of the implant. The recommended diameter of the implant is 1 cm smaller than the base of the breast.
Measurement B - determined using a flexible ruler; corresponds to the distance between the suprasternal notch and the superior margin of the areola (Figure 6). This information is important for selecting the profile of the implant to be used and for deciding whether there is a need to combine breast mastopexy with the procedure of implant inclusion;

Figure 6 - Measurement B, which corresponds to the distance from the sternal notch to the superior margin of the areola, is useful for selecting the height of the implant.
Measurement C - determined using a flexible ruler; corresponds to the distance between the inferior margin of the areola and the inframammary fold (Figure 7).

Figure 7 - Measurement C, which corresponds to the distance from the inferior margin of the areola to the inframammary fold.
The authors performed the measurements (in cm) based on the assessed parameters and the implant sizes proposed by the manufacturer of the selected brand (low, moderate, high, and extra-high projections).
All the patients received round, textured silicone implants, with profiles and diameters in accordance with each patient's need and based on the dimensions of the thorax, breasts, distance between the nipple-areola complex (NAC) and the midsternal line and midclavicular line.
Six months after the surgery, the patients evaluated their degree of satisfaction with the results of the surgery on a scale from 0 to 2, which corresponded to unsatisfactory, normal, and good results, respectively.
RESULTS
Measurement A varied between 9 and 12 cm; measurement B ranged from 23 cm to 17 cm, and measurement C varied between > 6 cm and 4 cm. Based on these markings, the patients were assigned to groups I to IV.
In group I, measurements A, B, and C were 12 cm, 17 cm, and 4 cm, respectively, and the indicated implants had moderate or low profiles (Figure 8).
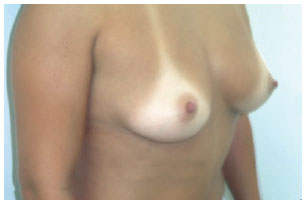
Figure 8 - Patient from group I.
In group II, measurements A, B, and C were 10 cm, 18 cm, and 5 cm, respectively, and the indicated implants had moderate or high profiles (Figure 9).
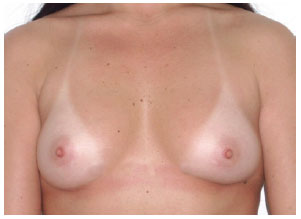
Figure 9 - Patient from group II.
In group III, measurements A, B, and C were 9 cm, 21 cm, and 6 cm, respectively, and the indicated implants had high or extra-high profiles (Figure 10).
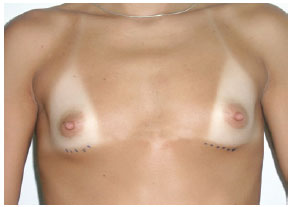
Figure 10 - Patient from group III.
Finally, in group IV, measurement A was variable, measurement B was 23 cm, and measurement C was > 6 cm, and mastopexy combined with implantation was indicated (Figure 11).
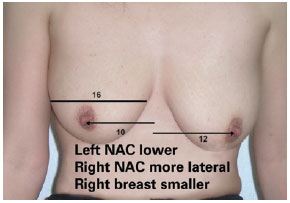
Figure 11 - Patient from group IV. The preoperative degree of ptosis and breast asymmetry can be observed. NAC = nipple-areola complex.
These data were transferred to a classification table with the scale and surgical indication, which was generated for each patient (Table 1). After a quick analysis of the parameters included in the table, the patients were assigned to groups I to IV, and subsequently, the ideal size of the implants to be used was defined. Moreover, the need for a mastopexy was evaluated.
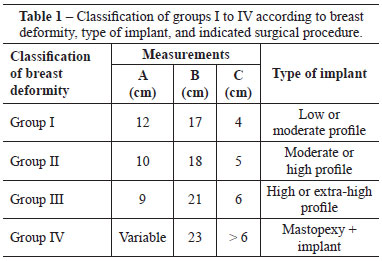
Figure 12 shows the case of a patient who desired a breast augmentation. She was assigned to group IV and was indicated to undergo augmentation combined with mastopexy. The data of the parameters were shown to the patient, and the appropriate procedure was proposed and performed after the patient provided consent. All the data were transcribed into the informed consent document, which was signed by the patient, and also into the medical records.

Figure 12 - In A, breasts requiring mastopexy; the breasts exhibit a higher degree of ptosis on the left. In B, measurement C, confirming the need for mastopexy. In C, postoperative results using similar measurements 1 year after mastopexy and inclusion of 280-cc silicone implants.
All the patients described the final surgery results 6 months after the surgery as good.
DISCUSSION
To perform augmentation mammaplasty, the incision site (periareolar, transareolar, inframammary, or axillary), positioning of the implant (retroglandular, submuscular, or dual plane), and texture, shape, and volume of the implant to be used need to be defined9.
The most important parameters for the clinical determination of implant size are the width of the base or the diameter of the patient's native breast.
In the past, it used to be more important to increase breast volume than to obtain a satisfactory breast shape2; the incidence of implant change, purely due to inadequate size selection, reported in the literature varies between 2% and 20.6%10.
Several authors have tried to evaluate the efficacy, degree of satisfaction, and quality of life after mammaplasty. Carty et al.11 studied the relationship between the degree of satisfaction and the surgeon's experience; they observed that younger patients who had complications exhibited lower levels of satisfaction, regardless of the surgeon's experience. Adams12 proposed 4 sequential steps to improve results based on educating the patient, informed consent, preoperative planning, surgical technique, preoperative instructions, and care and follow-up by the medical team. In 2007, Pusic et al.9 proposed the creation of the BREAST Q questionnaire to assess the satisfaction and quality of life of patients who had undergone aesthetic and reconstructive breast surgery. The items of this instrument are the following: reconstruction; reduction; augmentation; mastectomy; scales of satisfaction regarding the results; psychosocial and physical well-being; symptoms related to the thorax, upper limbs, trunk, and abdomen; sexual well-being; satisfaction regarding the abdominal wall and appearance of the areola; and information, in addition to parameters associated with the plastic surgeon and the medical team. We emphasize that the evaluation of the results will mostly depend on the appropriate surgical indication.
The above-mentioned authors aimed to obtain important parameters to evaluate the results of augmentation mammaplasty procedures; however, these may be insufficient in a judicial context. For further clarifications in the legal context, deformity is defined as an irregular shape - that is, alterations in shape are considered as a defect. When a breast deformity is established, the repair and overall medical care are the responsibility of the plastic surgeon; in the impossibility of predicting the result or providing a cure, the surgeon will nevertheless seek to find the best solution - this is the definition of obligation of means. This article addresses the anatomical evaluation and determination of the degrees of breast deformity that require reconstruction or repair procedures with the use of breast implants. Using the measurements obtained in this study, we were able to classify and document the condition and/or state of the breast that had lost its original shape and quantify the degree of deformity. This is a useful, practical, and objective way to select the most adequate breast implants. With this method, 100% of the patients graded the postoperative results as good and no litigations occurred during the study. The evaluation and documentation of parameters such as breast shape, angle of divergence, degree of ptosis, NAC position, asymmetries, and skin changes are important.
The European Committee on Quality Assurance and Medical Devices in Plastic Surgery13 and the competent agencies of each country have established specific requirements regarding the production and distribution of implants, including performance, material, design, manufacture, sterilization, packaging, and product information. However, surgeons did not have access to the records and to the actual number of complications related to the implants. Some initiatives have been presented, namely by the Danish Registry for Plastic Surgery of the Breast14 and the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery), the National Registry for Breast Implants15. These instruments allow the documentation of the number of implants and occurrences and demonstrate the existing concern for the safety of procedures. The use of a classification and the creation of the scale proposed in this study facilitated the combination of the litigation prevention tools already described in the pertinent and well-founded literature, with the appropriate training, doctor-patient relationship, informed consent, and objective evaluation of anatomical data for the indication of augmentation mammoplasty, among other parameters. Based on objective and easy-to-assess anatomical data, which should be documented in the medical records, it is possible to obtain results that are close to the natural, balanced beauty of the breast shape, to the satisfaction of both the surgeon and the patient. Moreover, the plastic surgeon holds objective and technical arguments to clearly explain to the patients the motives behind the choice of a specific size or projection of breast implant and to demonstrate the motives for performing a simultaneous mastopexy. Many surgeons make use of computer images; however, this tool may be questioned from a legal point of view, particularly when an image of the expected result is presented and that same result has not been achieved. Thus, we believe that the computer images of the result should be used with caution.
CONCLUSIONS
In the present study, there was a 100% satisfaction with the surgical results obtained in the 40 patients with various degrees of breast deformity, ranging from grades I to IV. Objective parameters were applied for the selection of breast implant size and profile and for the indication for augmentation mammaplasty with/without mastopexy. These data are an important tool for patient guidance with regard to the surgical indication parameters, for documentation in the consent form and medical records, for the evaluation of breast deformity, and for the proposed correction based on numerical data. These steps are important in case there is a need to be accountable before the judicial system, when the procedure involved is reconstructive breast surgery.
REFERENCES
1. Rees TD. Aesthetic plastic surgery. Philadelphia: W. B. Saunders; 1980.
2. Champaneria MC, Wong WW, Hill ME, Gupta SC. The evolution of breast reconstruction: a historical perspective. World J Surg. 2012;36(4):730-42.
3. Marchesi A, Marchesi M, Fasulo FC, Morini O, Vaienti L. Mammaplasties and medicolegal issues: 50 cases of litigation in aesthetic surgery of the breast. Aesthetic Plast Surg. 2012;36(1):122-7.
4. Mathes SJ. Plastic surgery. 2nd ed. Philadelphia: Saunders Elsevier; 2006.
5. Bozola AR, Longato FM, Bozola AP. Análise geométrica da forma da beleza da mama e da forma de prótese baseado na proporção Phi: aplicação prática. Rev Bras Cir Plást. 2011;26(1):94-103.
6. Paula PRS. Estudo descritivo e grau de satisfação de pacientes submetidos a implantes mamários de menor projeção em pólo superior. Rev Bras Cir Plást. 2010;25(1):168-78.
7. Baroudi R, Pitanguy I, Pontes R, Rebello C, Biggs T, Hochberg J, et al. Fórum: mamaplastia de aumento. Rev Soc Bras Cir Plást. 2000;15(3):7-20.
8. Queiroz RCFCL. Responsabilidade civil subjetiva nos procedimentos médicos estéticos: cirurgia plástica e obrigação de meio. Âmbito Jurídico. Maio 2012;XV(100). Acesso em: 5/7/2012. Disponível em: http://www.ambito-juridico.com.br/site/?n_link=revista_artigos_leitura&artigo_id=11602.
9. Pusic AL, Chen CM, Cano S, Klassen A, McCarthy C, Collins ED, et al. Measuring quality of life in cosmetic and reconstructive breast surgery: a systematic review of patient-reported outcomes instruments. Plast Reconstr Surg. 2007;120(4):823-39.
10. Spear SL, Dayan JH, West J. The anatomy of revisions after primary breast augmentation: one surgeon's perspective. Clin Plast Surg. 2009;36(1):157-65.
11. Carty MJ, Duclos A, Gu X, Elele N, Orgill D. Patient satisfaction and surgeon experience: a follow-up to the reduction mammaplasty learning curve study. Eplasty. 2012;12:e22.
12. Adams WP Jr. The process of breast augmentation: four sequential steps for optimizing outcomes for patients. Plast Reconstr Surg. 2008;122(6):1892-900.
13. European Committee on Quality Assurance and Medical Devices in Plastic Surgery. Consensus Declaration on Breast Implants. 4th Consensus Declaration; 2000. Acesso em: 5/7/2012. Disponível em: http://www.breastimplantanswers.com/downloads/EQUAM_European_Union_Report_on_SBI_Saf
ety_2000.pdf
14. Henriksen TF, Hölmich LR, Friis S, McLaughlin JK, Fryzek JP, Pernille Høyer A, et al. The Danish Registry for Plastic Surgery of the Breast: establishment of a nationwide registry for prospective follow-up, quality assessment, and investigation of breast surgery. Plast Reconstr Surg. 2003;111(7):2182-9.
15. Sociedade Brasileira de Cirurgia Plástica. Cadastro Nacional de Implantes Mamários. Acesso em: 20/6/2012. Disponível em: www.cirurgiaplastica.org.br
1. Plastic surgeon; full member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery, SBCP); voluntary professor of the Plastic Surgery Course at the Santa Casa de Misericórdia of São Paulo; judicial expert of the Fórum Central of the District of São Paulo, trusted by the Court and the Conselho Regional de Medicina do Estado de São Paulo (Regional Council of Medicine of the State of São Paulo); master in bioengineering and LASER by the University of Vale do Paraíba (UNIVAP); first vice president of the Brazilian Society of LASER in Medicine and Surgery; clinical director of the Clínica Lucano de Cirurgia Plástica, São Paulo, SP, Brazil
2. Plastic surgeon at the Hospital Sarah Brasília; full member of the SBCP; president of the SBCP Federal District Regional, Brasília, DF, Brazil
3. Aspiring member of the Colégio Brasileiro de Cirurgiões (Brazilian College of Surgeons); aspiring member of the SBCP; resident physician in plastic surgery at the Department of Professor Ewaldo Bolivar de Souza Pinto, University of Santa Cecília (UNISANTA), Santos, SP, Brazil
Correspondence to:
Kátia Torres Batista
SQN, 115 - Bloco I - ap. 205 - Asa Norte
Brasília, DF, Brazil - CEP 70772-090
E-mail: katiatb@terra.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: July 22, 2012
Article accepted: June 29, 2013
This study was performed at the Clínica Lucano de Cirurgia Plástica, São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter