

Original Article - Year 2012 - Volume 27 -
Physical activity in women undergoing mastectomy and breast reconstruction
Nível de atividade física em mulheres mastectomizadas e submetidas a reconstrução mamária
ABSTRACT
BACKGROUND: Regular physical activity has been recommended for the prevention and treatment of coronary heart disease, diabetes mellitus, and hypertension. Besides being a protective factor against breast cancer, physical activity following the diagnosis of breast cancer has been associated with an improved quality of life and survival. The aim of this study was to assess levels of physical activity in women who had undergone mastectomy without breast reconstruction and in women who had undergone breast reconstruction after breast cancer treatment.
METHODS: Two groups, each with 18 patients, comprised the patient series. One group comprised women who had undergone mastectomy without breast reconstruction, and the other group comprised women who had undergone mastectomy and breast reconstruction. All patients were aged between 18 and 60 years. The exclusion criteria were physical disability; illiteracy; ongoing chemotherapy, radiation therapy, or psychiatric therapy; and surgery performed within the previous 12 months. Study participants completed the International Physical Activity Questionnaire. Statistical analysis was performed by applying the chi-square and Student's t tests, adopting a significance level of P < 0.05.
RESULTS: In the group of women who had undergone mastectomy without breast reconstruction, 16.7% were very active, 61.1% were active, and 22.2% were insufficiently active. In the group of women who had undergone mastectomy with breast reconstruction, 55.6% were very active, 33.3% were active, and 11.1% were insufficiently active. The difference between the groups was statistically significant (P < 0.0001).
CONCLUSIONS: In this study, the level of physical activity among women who underwent breast reconstruction was higher than that among women who underwent mastectomy without breast reconstruction.
Keywords: Mammaplasty. Breast neoplasms. Quality of life. Questionnaires.
RESUMO
INTRODUÇÃO: A prática regular de atividade física tem sido recomendada para prevenção e tratamento de doenças coronárias, diabetes melito e hipertensão arterial. Além de fator protetor, a atividade física após o diagnóstico de câncer de mama tem sido correlacionada a aumento da qualidade de vida e da sobrevida. O objetivo deste estudo é avaliar o nível de atividade física de mulheres mastectomizadas sem reconstrução mamária e de mulheres submetidas a reconstrução mamária pós-tratamento de câncer de mama.
MÉTODO: A casuística foi composta por 2 grupos, um de mulheres mastectomizadas sem reconstrução mamária e outro de mulheres mastectomizadas e submetidas a reconstrução mamária pós-mastectomia, ambos com 18 pacientes. Todas as pacientes tinham idade entre 18 anos e 60 anos. Os critérios de exclusão foram: deficiência física, analfabetismo, vigência de tratamento de quimioterapia, radioterapia ou psiquiátrico, e tratamento cirúrgico realizado em período inferior a um ano. As voluntárias responderam ao Questionário Internacional de Atividade Física (IPAQ). A análise estatística foi realizada aplicando-se o teste qui-quadrado e o teste t de Student, tendo sido adotado o nível de significância de P < 0,05.
RESULTADOS: No grupo de mulheres mastectomizadas sem reconstrução mamária, 16,7% eram muito ativas, 61,1% ativas e 22,2% insuficientemente ativas. No grupo de mulheres com a mama reconstruída, essas incidências foram de 55,6%, 33,3% e 11,1%, respectivamente. Houve diferença estatisticamente significante entre os grupos (P < 0,0001).
CONCLUSÕES: O nível de atividade física entre as mulheres submetidas a reconstrução mamária é melhor que entre as mulheres mastectomizadas sem reconstrução mamária.
Palavras-chave: Mamoplastia. Neoplasias da mama. Qualidade de vida. Questionários.
The relationship between physical activity and health is well known, and has been referred to in ancient Chinese, Indian, Greek, and Roman texts. However, a direct relationship between sedentary behavior and population mortality rates has only been established in the last 3 decades1. Epidemiological studies have indicated a strong relationship between inactivity and the presence of cardiovascular risk factors such as hypertension, insulin resistance, non-insulin dependent diabetes, dyslipidemia, and obesity2-4. Moreover, regular physical activity has been recommended not only for the prevention and treatment of the abovementioned diseases, but also for chronic diseases such as osteoporosis; certain cancers, such as colon and breast cancer; stress reduction; and to improve self-esteem5-13. This relationship becomes evident in the reduction in sequelae, reduced need for hospitalization, and fewer medications needed to control these diseases, all of which reduce the costs associated with hospital medical services. Thus, physical activity alone can be considered an excellent option for cost containment in public health.
Currently, more than 2 million deaths worldwide could be avoided with exercise14. In 2002, the Government of the State of São Paulo spent millions of dollars on sedentary lifestyle-related health problems, which equated to 3.6% of the state's total health expenditure and more than half of the total hospital expenditure ($179.9 million); 10 health problems were primarily found to be associated with inactivity15. Risk factors such as a lack of physical activity, smoking, and poor diet are directly related to lifestyle, and account for > 50% of the total risk of developing a particular chronic disease. These factors present a more decisive causal relationship than the combination of genetic and environmental factors. Among these factors, a sedentary lifestyle emerges as the most predominant risk factor in the population, regardless of sex16.
Besides being a protective factor against breast cancer, physical activity following the diagnosis of breast cancer has been strongly correlated with improvements in the quality of life17 and increased survival in woman with breast cancer18. Studies by the World Health Organization's (WHO) International Agency for Research on Cancer estimate that more active women have a 20%-40% lower risk of developing breast cancer19. This finding has been explained by the decline in circulating ovarian hormones, which are closely linked to the development of the disease20. Moreover, physical activity prevents weight gain during breast cancer treatment, which is a common adverse effect21. This is important because being overweight is a risk factor for increased morbidity. However, despite the well-established association between regular exercise and health, this relationship has been questioned. Some authors interpret this association differently, arguing that some people exercise because they would like to present an image of better health and not the reverse, which characterizes the problem of "endogeneity"22,23.
Cancer is defined as the uncontrolled growth and abnormal spread of cells in the body. Breast cancer is the most frequently diagnosed cancer in women. More than 190,000 women are diagnosed annually with breast cancer in the United States24. The most common forms of treatment include surgery (lumpectomy or mastectomy), chemotherapy, radiation therapy, or hormonal therapy. Usually, more than 1 type of treatment is used in a complementary manner. Despite the therapeutic success obtained, side effects are frequently associated with the treatment administered, leading to major declines in functionality. Fatigue is the most commonly observed side effect, affecting at least 30% of patients25. Declines in physical activity exacerbate the side effects, leading to patients experiencing recurrent adverse effects, which further aggravate the feelings of fatigue. The forced reduction in physical activity levels promotes the development of a pathological condition that, in association with other side effects, including the loss of appetite, can exacerbate physical decline and, subsequently, loss of overall muscle strength. The loss of muscle strength decreases the ability of patients with cancer to perform simple daily tasks, thus significantly compromising their quality of life26. The causes of fatigue associated with cancer treatment should be viewed as multifactorial and related to both physical and emotional deterioration27.
The diagnosis of cancer and its therapy greatly affect the psychological health of patients, especially when mutilating surgery is involved, and can even result in psychiatric sequelae28. In the United States, approximately 69,000 women undergo mastectomy annually29, and a similar rate has been reported in Great Britain30. Mastectomy affects patients psychosocially and has impacts on various aspects of their quality of life, including self-image, self-esteem, and sexual relationships31, in addition to being associated with high rates of depression32.
In patients undergoing mastectomy, the major goal of reconstructive surgery is aesthetic rehabilitation - to remove the stigma associated with cancer and mutilation. A return to the precancerous state is fundamental to this process and the removal of muscle does not have a significant functional impact33. A patient's motivation and willingness are the main indications for reconstructive surgery, which reduces the feeling of deformity that develops after mastectomy.
Immediate breast reconstruction may be considered for patients with stages 0, I, and II of the disease34. It is oncologically safe and is the most commonly indicated procedure, provided the histological results are satisfactory. Besides the obvious psychological benefit, preservation of body image is undoubtedly a good reason to encourage reconstruction. When reconstructions are performed by well-trained staff, the morbidity of such cases are not significantly greater as compared to cases that undergo mastectomy without reconstruction35.
The incidence of breast reconstruction after mastectomy differs among countries and among hospitals within the same country. In the United States, the reconstruction rate was estimated at 8.3% from 1994 to 1995 29, whereas in the United Kingdom and Ireland, it was estimated at 18% in 2000 30. In other industrialized countries, some specialized hospitals reported rates of >50%36. For women who undergo mastectomy, breast reconstruction usually improves their quality of life and the psychosocial aspects of life without diminishing their chances of survival, regardless of disease stage37.
A positive relationship exists between the quality of life and plastic surgery, even though few studies examining this association have been published. Veiga et al.38 showed that reconstruction using the rectus abdominis muscle flap (the transverse rectus abdominis myocutaneous [TRAM]) improved the quality of life of patients. Brandberg et al.39 described similar results.
The aim of this study was to evaluate levels of physical activity in women who had undergone mastectomy without breast reconstruction and in women who had undergone breast reconstruction following mastectomy.
METHODS
Data were collected from the breast reconstruction and mastology outpatient clinics of the Gynecology Department of the Federal University of São Paulo (UNIFESP). Both outpatient clinics are located within the complex of the Hospital São Paulo, a teaching hospital of the UNIFESP.
The patient series comprised 2 groups, each with 18 subjects. One group comprised women who underwent mastectomy without breast reconstruction, and the other group comprised women who underwent mastectomy with breast reconstruction. Patients were interviewed and a form pertaining to their clinical data was completed. Only those patients who completed and signed a consent form participated in the study. The following inclusion criteria were used for the 2 study groups:
Women who had undergone mastectomy without breast reconstruction who were aged between 18 and 60 years, and were not being treated with radiation or chemotherapy, or receiving psychiatric treatment. Women who had undergone mastectomy with complete breast reconstruction who were aged between 18 and 60 years, and were not receiving psychiatric treatment.
Patients who did not fit the inclusion criteria, or were illiterate, physically disabled, or had disabling sequelae preventing them from participating in physical activity, were excluded from the study.
Study participants were asked to complete the International Physical Activity Questionnaire (IPAQ) developed by the WHO in 1998. This self-administered questionnaire includes questions on the activities performed during a typical week. Activities at work, home, leisure, and during movement from one place to another are recorded. On average, the questionnaire takes 10 min to complete. This questionnaire has been translated into Portuguese and validated for use in Brazil40.
This instrument has been successfully applied to assess levels of physical activity in different populations41, including a multicenter study analyzing physical activity in individuals from 20 countries42, a study of patients with multiple sclerosis43, a comparative pre- and postoperative study of arthroplasty44 in women after the diagnosis of breast cancer45, and a study of schizophrenic patients46.
IPAQ Rating
Following the tabulation and evaluation of patient data, participants were classified according to the following IPAQ categories:
Sedentary: does not perform any physical activity for at least 10 min continuously each week; Insufficiently active: is physically active for at least 10 min continuously per week, but not in a manner that could be classified as active. To assign individuals to this category, the duration and frequency of different types of activities (walks, moderate activity, and vigorous activity) are summed. This category is divided into 2 groups: - Insufficiently active A: is continuously physically active for 10 min on 5 days of the week, or for a total duration of 150 min per week;
- Insufficiently active B: does not fulfill any of the criteria of the "insufficiently activeA" category.Active: a) > 3 days per week of vigorous physical activity for at least 20 minutes per session; b) > 5 days per week of moderate activity or walking for up to 30 minutes per session; c) any combination of moderate-intensity or vigorous activity on > 5 days of the week or for > 150 min per week; Very active: meets the following criteria a) vigorously physically active for more than 30 min per session on 5 or more days of the week, or b) vigorously physically active for more than 20 min per session on 3 days of the week, combined with moderate physical activity and/or walking for 30 min per session on 5 or more days of the week.
Statistical analysis was performed by applying the chi-square test, adopting a significance level of P < 0.05.
RESULTS
We interviewed 36 patients, including 18 who had undergone mastectomy without breast reconstruction and 18 who had undergone mastectomy with breast reconstruction.
In the group of women who had undergone mastectomy without breast reconstruction, 16.7% were very active, 61.1% were active, and 22.2% were insufficiently active. In the group of women with breast reconstruction following mastectomy, 55.6% were very active, 33.3% were active, and 11.1% were insufficiently active. The difference between the groups with respect to the levels of physical activity was statistically significant (P < 0.0001) (Figure 1).
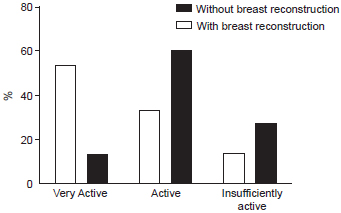
Figure 1 - Distribution of physical activity in women who underwent mastectomy with and without breast reconstruction.
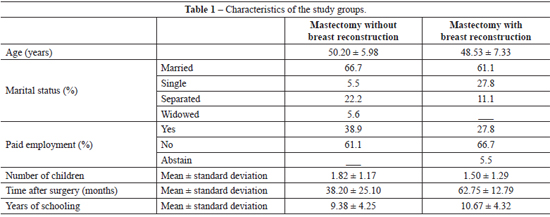
The mean age of the patients was 49.37 ± 6.63 years. The women who had undergone mastectomy without breast reconstruction had a mean age of 50.2 ± 5.98 years. The women who had undergone mastectomy with breast reconstruction had a mean age of 48.53 + 7.33 years. The difference between the mean ages of the 2 groups was not statistically significant (P = 0.5005).
The average time after surgery was 38.2 ± 25.1 months in the group that had undergone mastectomy without breast reconstruction and 62.75 ± 12.79 months in the group who had undergone breast reconstruction following mastectomy. Among the patients who underwent mastectomy without breast reconstruction, 66.7% were married, whereas, among the patients who had breast reconstruction following mastectomy, 61.1% were married.
The women who had undergone mastectomy without breast reconstruction and those who had undergone mastectomy with breast reconstruction, had a mean of 1.82 ± 1.17 children and 1.5 ± 1.29 children, respectively. In total, 38.9% of women who had undergone mastectomy without breast reconstruction were paid workers, whereas 27.8% of those who had undergone mastectomy with breast reconstruction were paid workers. The patients who had undergone mastectomy without breast reconstruction had a mean of 9.38 ± 4.25 years of schooling, whereas those who had breast reconstruction following mastectomy had a mean of 10.67 ± 4.32 years of schooling. These data are summarized in Table 1.
DISCUSSION
Historically, medical treatment outcomes have been measured primarily in terms of mortality, morbidity, or cure; however, more recent studies have correlated health outcomes with quality of life, making the latter a new goal in medical management.
The diagnosis of cancer and its treatment greatly affect the psychological health of patients, especially when treatment involves mutilating surgery47. The sense of body integrity is fundamental to humans. Wellbeing in relation to breast cancer is expressed in the manner in which each woman views herself and, thus, in the level of physical activity she pursues. Within this context, mastectomy reduces a woman's ability to perform tasks as well as her self-esteem. This causes a decline in a woman's quality of life, which also affects the levels of physical activity.
The breast symbolizes female identity. Breast reconstruction is very important if breast cancer patients are to regain self-esteem, which helps in the recovery from illness and in the restoration of social activities. Moreover, it helps women attain an acceptable quality of life, in addition to obtaining aesthetically satisfactory features48. Although few studies have explored the association between quality of life and plastic surgery, a positive relationship exists between them. For example, Veiga et al.38 and Brandberg et al.39 demonstrated that TRAM flap reconstruction positively impacts the quality of life of patients, improving their body functions and mental health. Thus, we can state that reconstruction also has repercussions on the physical activity of these patients. This is corroborated by the fact that in the current study, more women in the group who had undergone breast reconstruction following mastectomy were classified as being very active and active, compared with the group who had not undergone breast reconstruction following mastectomy. To our knowledge, none of the published studies have assessed the levels of physical activity in women who have undergone mastectomy and breast reconstruction.
The main difficulties in recruiting participants for this study were age and the amount of time that had lapsed following surgery. The upper age limit for inclusion in the study was established to avoid bias in relation to the physical activity associated with bodily limitations. Many of our patients are aged > 60 years and could not be included in the study. With regard to the length of the postoperative period, many of our patients had undergone surgery within the previous 12 months. In addition, some patients were unable to participate because they were illiterate.
Patients were carefully selected and we aimed to obtain 2 homogeneous groups. Internally, the groups were quite heterogeneous, both in relation to age and socially, which reflects the composition of the population in general.
We can infer from the results of the current study that if there was adequate educational and psychological support for patients undergoing breast cancer treatment, they would be better equipped to deal with this phase, with fewer losses in terms of quality of life and their overall health. Therefore, policies aimed at creating this type of support are needed.
CONCLUSIONS
The level of physical activity among women who underwent breast reconstruction following mastectomy is higher than that among women who underwent mastectomy without breast reconstruction.
REFERENCES
1. Stephens T. Fitness and lifestyle in Canada. Ottawa: Canadian Fitness and Lifestyle Research Institute; 1983.
2. Rennie KL, McCarthy N, Yazdgerdi S, Marmot M, Brunner E. Association of metabolic syndrome with both vigorous and moderate physical activity. Int J Epidemiol. 2003;32(4):600-6.
3. Gustat J, Srinivasan SR, Elkasabany A, Berenson GS. Relation of self-rated measures of physical activity to multiple risk factors of insulin resistance syndrome in young adults: the Bogalusa Heart Study. J Clin Epidemiol. 2002;55(10):997-1006.
4. Lakka TA, Laaksonem DE, Laaka HM, Männikkö N, Niskanen LK, Rauramaa R, et al. Sedentary lifestyle, poor cardiorespiratory fitness, and the metabolic syndrome. Med Sci Sports Exerc. 2003;35(8):1279-86.
5. Physical activity and cardiovascular health. NIH Consensus Development Panel on Physical Activity and Cardiovascular Health. JAMA. 1996;276(3):241-6.
6. Sociedade Brasileira de Cardiologia. III Diretrizes brasileiras sobre dislipidemias e diretriz sobre prevenção da aterosclerose do Departamento de Aterosclerose da Sociedade Brasileira de Cardiologia. Arq Bras Cardiol. 2001;77(Supl. III):1-48.
7. Whelton SP, Chin A, Xin X, He J. Effect of aerobic exercise on blood pressure: a meta-analysis of randomized, controlled trials. Ann Intern Med. 2002;136(7):493-503.
8. Castaneda C, Layne LE, Munoz-Orians L, Gordon PL, Walsmith J, Foldvari M, et al. A randomized controlled trial of resistance exercise training to improve glycemic control in older adults with type 2 diabetes. Diabetes Care. 2002;25(12):2335-41.
9. Durstine JL, Haskell WL. Effects of exercise training on plasma lipids and lipoproteins. Exerc Sport Sci Rev. 1994;22:477-521.
10. Slattery ML, Edwards S, Curtin K, Ma K, Edwards R, Holubkov R, et al. Physical activity and colorectal cancer. Am J Epidemiol. 2003;158(3):214-24.
11. Thune I, Brenn T, Lund E, Gaard M. Physical activity and the risk of breast cancer. N Engl J Med. 1997;336(18):1269-75.
12. Harlow SD, Matanoski GM. The association between weight, physical activity, and stress and variation in the length of the menstrual cycle. Am J Epidemiol. 1991;133(1):38-49.
13. Netz Y, Wu MJ, Becker BJ, Tenenbaum G. Physical activity and psychological well-being in advanced age: a meta-analysis of intervention studies. Psychol Aging. 2005;20(2):272-84.
14. Washburn RA, Smith KW, Jette AM, Janney CA. The Physical Activity Scale for the Elderly (PASE): development and evaluation. J Clin Epidemiol. 1993;46(2):153-62.
15. http://www1.folha.uol.com.br/folha/equilibrio/noticias/ult263u2845.shtml. Em 17/05/2009.
16. Pinto BM, Maruyama NC. Exercise in the rehabilitation of breast cancer survivors. Psychooncology. 1999;8(3):191-206.
17. Kramer JA, Curran D, Piccart M, de Haes JC, Bruning P, Klijn J, et al. Identification and interpretation of clinical and quality of life prognostic factors for survival and response to treatment in first-line chemotherapy in advanced breast cancer. Eur J Cancer. 2000;36(12):1498-506.
18. Holmes MD, Chen WY, Feskanich D, Kroenke CH, Coldtiz GA. Physical activity and survival after breast cancer diagnosis. JAMA. 2005;293(20):2479-86.
19. Bianchini F, Kaaks R, Vainio H. Weight control and physical activity in cancer prevention. Obes Rev. 2002;3(1):5-8.
20. Broocks A, Pirke KM, Schweiger U, Tuschl RJ, Laessle RG, Strowitzki T, et al. Cyclic ovarian function in recreational athletes. J Appl Physiol. 1990;68(5):2083-6.
21. Demark-Wahnefried W, Peterson BL, Winer EP, Marks L,Aziz N, Marcom PK, et al. Changes in weight, body composition, and factors influencing energy balance among premenopausal breast cancer patients receiving adjuvant chemotherapy. J Clin Oncol. 2001;19(9):2381-9.
22. Blair SN, Kohl HW 3rd, Barlow CE, Paffenbarger RS Jr, Gibbons LW, Macera CA. Changes in physical fitness and all-cause mortality. A prospective study of healthy and unhealthy men. JAMA. 1995;273(14):1093-8.
23. Williams PT. The illusion of improved physical fitness and reduced mortality. Med Sci Sports Exerc. 2003;35(5):736-40.
24. Lee JH, Glick HA, Hayman JA, Solin LJ. Decision-analytic model and cost-effectiveness evaluation of postmastectomy radiation therapy in high-risk premenopausal breast cancer patients. J Clin Oncol. 2002;20(11):2713-25.
25. Dimeo F, Fetscher S, Lange W, Mertelsmann R, Keul J. Effects of aerobic exercise on the physical performance and incidence of treatment-related complications after high-dose chemotherapy. Blood. 1997;90(9):3390-4.
26. Dimeo FC, Stieglitz RD, Novelli-Fischer U, Fetscher S, Keul J. Effects of physical activity on fatigue and psychologic status of cancer patients during chemotherapy. Cancer. 1999;85(10):2273-7.
27. Mock V, Pickett M, Ropka ME, Muscari Lin E, Stewart KJ, Rhodes VA, et al. Fatigue and quality of life outcomes of exercise during cancer treatment. Cancer Pract. 2001;9(3):119-27.
28. Frierson GM, Thiel DL, Andersen BL. Body change stress for women with breast cancer: the Breast-Impact of Treatment Scale. Ann Behav Med. 2006;32(1):77-81.
29. Morrow M, Scott SK, Menck HR, Mustoe TA, Winchester DP. Factors influencing the use of breast reconstruction postmastectomy: a National Cancer Database study. J Am Coll Surg. 2001;192(1):1-8.
30. Callaghan CJ, Couto E, Kerin MJ, Rainsbury RM, George WD, Purushotham AD. Breast reconstruction in the United Kingdom and Ireland. Br J Surg. 2002;89(3):335-40.
31. Rowland JH, Desmond KA, Meyerowitz BE, Belin TR, Wyatt GE, Ganz PA. Role of breast reconstructive surgery in physical and emotional outcomes among breast cancer survivors. J Natl Cancer Inst. 2000;92(17):1422-9.
32. Lasry JC, Margolese RG, Poisson R, Shibata H, Fleischer D, Lafleur D, et al. Depression andbody imagefollowingmastectomy and lumpectomy. J Chronic Dis. 1987;40(6):529-34.
33. Kokuba EM, Sabino Neto M, Garcia EB, Bastos EM, Aihara AY, Ferreira LM. Functional capacity after pedicled TRAM flap delayed breast reconstruction. J Plast Reconstr Aesthet Surg. 2008;61(11):1394-6.
34. Malata CM, McIntosh AS, Purushotham AD. Immediate breast reconstruction after mastectomy for cancer. Br J Surg. 2007;87(11):1455-72.
35. Leal PR, Cammarota MC, Palma L, Sbalchiero J. Reconstrução imediata de mama: avaliação das pacientes operadas no Instituto Nacional de Câncer no período de junho de 2001 a junho de 2002. Rev Bras Mastol. 2003;13(4):149-58.
36. Rosenqvist S, Sandelin K, Wickman M. Patients' psychological and cosmetic experience after immediate breast reconstruction. Eur J Surg Oncol. 1996;22(3):262-6.
37. Harcourt D, Rumsey N. Psychological aspects of breast reconstruction: a review of the literature. J Adv Nurs. 2001;35(4):477-87.
38. Veiga DF, Sabino Neto M, Ferreira LM, Garcia EB, Veiga Filho J, Novo NF, et al. Quality of life outcomes after pedicled TRAM flap delayed breast reconstruction. Br J Plast Surg. 2004;57(3):252-7.
39. Brandberg Y, Malm M, Blomqvist L. A prospective and randomized study, 'SVEA', comparing effects of three methods for delayed breast reconstruction on quality of life, patient-defined problem areas of life, and cosmetic result. Plast Reconstr Surg. 2000;105(1):66-74.
40. Pardini R, Matsudo SM, Araújo T, Matsudo V, Andrade E, Braggion G, et al. Validação do questionário internacional de nível de atividade física (IPAQ -versão 6): estudo piloto em adultos jovens brasileiros. Rev Bras Ciên Mov. 2001;9(3):45-51.
41. Jurakić D, Pedisić Z, Andrijasević M. Physical activity of Croatian population: cross-sectional study using International Physical Activity Questionnaire. Croat Med J. 2009;50(2):165-73.
42. Bauman A, Bull F, Chey T, Craig CL, Ainsworth BE, Sallis JF, et al; IPS Group. The International Prevalence Study on physical activity: results from 20 countries. Int J Behav Nutr Phys Act. 2009;6:21.
43. Wynia K, Middel B, Ruiter H, van Dijk JP, Keyser JH, Reijneveld SA. Stability and relative validity of the Multiple Sclerosis Impact Profile (MSIP). Disabil Rehabil. 2008;30(14):1027-38.
44. Naal FD, Impellizzeri FM, Loibl M, Huber M, Rippstein PF. Habitual physical activity and sports participation after total ankle arthroplasty. Am J Sports Med. 2009;37(1):95-102.
45. Johnson-Kozlow M, Sallis JF, Gilpin EA, Rock CL, Pierce JP. Comparative validation of the IPAQ and the 7-day PAR among women diagnosed with breast cancer. Int J Behav Nutr Phys Act. 2006;3:7.
46. Faulkner G, Cohn T, Remington G. Validation of a physical activity assessment tool for individuals with schizophrenia. Schizophr Res. 2006;82(2-3):225-31.
47. Rivera HR. Depression symptoms in cancer caregivers. Clin J Oncol Nurs. 2009;(2):195-202.
48. Veiga DF, Sabino Neto M, Garcia EB, Veiga Filho J, Juliano Y, Ferreira LM, et al. Evaluations of the aesthetic results and patient satisfaction with the late pedicled TRAM flap late breast reconstruction. Ann Plast Surg. 2002;48(5):515-20.
1. Associate professor, Department of Plastic Surgery, Escola Paulista de Medicina, Federal University of São Paulo (EPM-UNIFESP), full member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery - SBCP), São Paulo, SP, Brazil.
2. Resident physician EPM-UNIFESP, São Paulo, SP, Brazil.
3. MSc student, EPM-UNIFESP, São Paulo, SP, Brazil.
4. Full professor, Department of Plastic Surgery, EPM-UNIFESP, full member of the SBCP, São Paulo, SP, Brazil.
Correspondence to:
Miguel Sabino Neto
Rua Napoleão de Barros, 715 - 4º andar - Vila Clementino
São Paulo, SP, Brazil - CEP 04024-002
E-mail: msabino@uol.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: October 9, 2012
Article accepted: December 13, 2012
This study was performed at the Escola Paulista de Medicina, Universidade Federal de São Paulo (Federal University of São Paulo), São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter