

Original Article - Year 2012 - Volume 27 -
Extremity reconstruction using a free fibula flap after oncological resection
Reconstrução de extremidades com retalho livre de fíbula após ressecções oncológicas
ABSTRACT
BACKGROUND: Primary tumors of the long bones are rare, accounting for 0.2-1% of malignant tumors. In the past, amputation was the standard treatment and had a large impact on patient morbidity and mortality. With advances in surgical techniques and multidisciplinary involvement, conservative surgery of the limbs has become the treatment of choice, and reconstruction using a microsurgical fibula flap is the most commonly used technique. In this study, we aimed to present the experience of the National Cancer Institute (INCA) with limb reconstruction using a microsurgical fibula flap following tumor resection from the long bones.
METHODS: We retrospectively analyzed 7 cases of free fibular flap surgery at the INCA from 1997 to 2009 for the reconstruction of defects of the extremities after bone tumor resection. We evaluated the following parameters: gender, age, diagnosis, tumor location, resection size and type, reconstruction size and type, vessels used for the anastomosis, postoperative complications, disease status at the last visit, follow-up, and time to ambulation.
RESULTS: Seven patients with a mean age of 11.8 years (range, 5-14 years) underwent extremity reconstruction with a free fibula flap with 100% bone viability. The lesions were located within the femur, tibia, or humerus. Osteosarcoma was the most common tumor type. The average return to ambulation was 14.7 months.
CONCLUSIONS: The use of a free fibula flap is an excellent alternative for limb reconstruction and features a high bone healing rate, early ambulation, good functionality, and a low complication rate.
Keywords: Fibula. Extremities. Transplantation, homologous. Reconstructive surgical procedures.
RESUMO
INTRODUÇÃO: O tumor primário de ossos longos é raro, correspondendo de 0,2% a 1% dos tumores malignos. No passado, a amputação era o tratamento padrão, ocasionando grande impacto na morbidade e na mortalidade desses pacientes. Com o avanço das técnicas cirúrgicas e o envolvimento multidisciplinar, a cirurgia conservadora dos membros tornou-se o tratamento de escolha, sendo a reconstrução com retalho microcirúrgico de fíbula a mais utilizada. Este trabalho tem como objetivo apresentar a experiência do Instituto Nacional de Câncer (INCA) nas reconstruções de membros com retalho microcirúrgico de fíbula após ressecções de tumores de ossos longos.
MÉTODO: Foi realizada análise retrospectiva de 7 casos de retalho livre de fíbula operados no INCA, no período de 1997 a 2009, para reconstrução de defeitos de extremidades após ressecções de tumores ósseos. Foram avaliados os seguintes parâmetros: sexo, idade, diagnóstico, localização do tumor, tipo e tamanho da ressecção, tipo e tamanho da reconstrução, vasos utilizados para anastomose, complicações pós-operatórias, estado da doença na última consulta, seguimento e tempo até deambulação.
RESULTADOS: No total, 7 pacientes com média de idade de 11,8 anos (variando de 5 anos a 14 anos) foram submetidos a reconstrução de extremidades com retalho livre de fíbula, com 100% de viabilidade e consolidação óssea. As lesões eram localizadas em fêmur, tíbia ou úmero. O tumor mais comum foi o osteossarcoma. O tempo médio de retorno à deambulação foi de 14,7 meses.
CONCLUSÕES: O uso do retalho livre de fíbula é uma excelente alternativa para reconstrução de membros, apresentando alta taxa de consolidação óssea, deambulação precoce, boa funcionalidade e baixa taxa de complicações.
Palavras-chave: Fíbula. Extremidades. Transplante homólogo. Procedimentos cirúrgicos reconstrutivos.
Primary malignant tumors of the long bones are rare, accounting for 0.2-1% of malignant tumors; osteosarcoma and Ewing's sarcoma are the most frequent of these types of tumors1-3.
Amputation, the classical treatment of such tumors, provided excellent local disease control but had a great impact on patient morbidity and mortality.
Conservative limb surgery has emerged as an alternative, and recent studies have shown a similar efficacy of this procedure to amputation after neoadjuvant chemotherapy1,2. Conservative limb surgery is appropriate for approximately 90% of patients1. Contraindications include the involvement of key neurovascular trunks, large muscle involvement, infection, and lack of patient motivation for rehabilitation and other surgical procedures4.
With advances in surgical techniques and multidisciplinary involvement of plastic surgery, orthopedics, pathology, medical oncology, and physiotherapy, conservative limb surgery became the treatment of choice for tumors of the long bones. Surgeons performing this procedure should always respect 3 principles: 1. the chance of recurrence should not be greater than amputation; 2. it should not affect overall patient survival; and 3. the rehabilitation period should not postpone further treatment1,3,4.
There are many techniques for conservative limb surgery. Microsurgical fibular and iliac crest flaps are more commonly used than autologous bone grafts and allografts. These latter techniques do not require microsurgery and present with high infection rates, pseudoarthrosis, and fracture, which leads to graft failure in up to 50% of cases1-5. Thus, based on the experience gained with the use of free flaps in the reconstruction of limbs in trauma and jaw reconstructions, free fibula transplantation is becoming increasingly common in orthopedic reconstructions after oncological resections since they are preferable to iliac crest free flaps for creating straight and long cortical bones and because a skin island can be included when necessary5-7.
To date, few studies have evaluated the efficacy of extremity reconstruction using a free fibula flap after oncological resection1,8,9. Therefore, in this study, we aim to present the experience of the Instituto Nacional de Câncer (INCA) in the reconstruction of limbs using this flap after the resection of long bone tumors.
METHODS
We retrospectively analyzed 7 patients who received a free fibular flap at the INCA between 1997 and 2009 for the reconstruction of defects of the extremities after bone tumor resection.
Hospital records were evaluated for patient gender, age, diagnosis, tumor location, resection size, reconstruction size, blood vessels used for the anastomosis, postoperative complications, disease status at the last visit, follow-up, and time to ambulation.
RESULTS
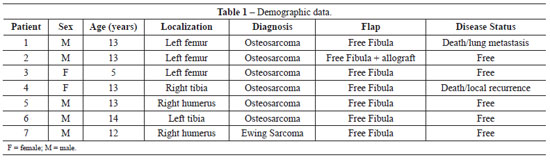
Between 1997 and 2009, 6 patients underwent extremity reconstruction at the INCA using a free fibula flap, whereas 1 patient received a fibular free flap and an allograft (Table 1). The lesions were located in the femur (n = 3), tibia (n = 2), and humerus (n = 2), and 5 patients were male. The mean age was 11.8 years (range, 5-14 years). The most common tumor was osteosarcoma (n = 6).
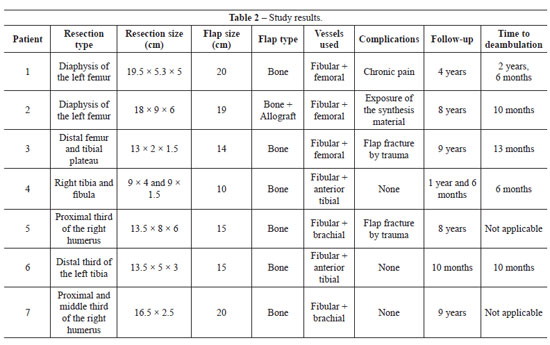
All reconstructions were performed using a bone flap without a skin island that had an average size of 15.8 cm (range, 10-20 cm) (Table 2). All patients underwent neoadjuvant and adjuvant chemotherapy.
Two patients developed a flap fracture in the late postoperative period due to trauma and both experienced consolidation after treatment. One patient had chronic pain and another presented with exposure of the synthesis material after 7 months, which was removed without flap damage. Two patients died - one as a result of metastatic disease and the other due to local recurrence.
The average return to ambulation was 14.2 months (range, 6-30 months). Four patients were free of disease at the end of this study.
Figures 1 and 2 illustrate some cases in this series.
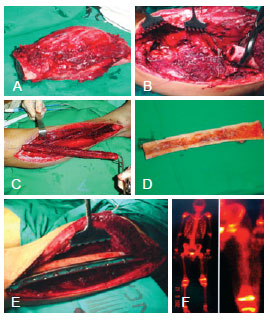
Figure 1 - Case 2. In A, osteosarcoma of the left femur. In B, the receiving area. In C, the fibula flap. In D, allograft. In E, free fibula flap + allograft fixed by the receiving area. In F, perfusion scintigraphy performed on postoperative day 20 showing good perfusion of the flap and the allograft.
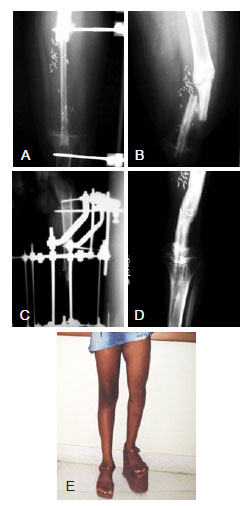
Figure 2 - Case 3. In A, radiography performed on immediate postoperative reconstruction of defect of the left femur and tibial plateau with a fibular free flap. In B, radiograph showing flap fracture in the late postoperative period. In C, radiograph after fracture reduction and external fixation. In D, radiograph showing flap consolidation and hypertrophy. In E, late postoperative appearance.
DISCUSSION
The essential goal of treating virtually all long bone tumors is appropriate surgical resection combined with chemotherapy along with lower morbidity and improved survival. With the progress of such treatment modalities, there has been a significant reduction in mortality over recent decades. Currently, up to 75% of patients without metastases at the time of admission and 50% of those with isolated pulmonary metastases are cured2. Therefore, oncological resection-related reconstruction is mandatory and requires appropriate integration between the orthopedics and plastic surgery department4,10-12. In this series, all patients received neoadjuvant and adjuvant chemotherapy.
Several long bone reconstruction techniques, such as cancellous bone graft, allografts, the use of prostheses, and external fixation techniques, have been used with this purpose in mind13-15. Cancellous bone graft reconstruction may be suitable for small defects surrounded by good amounts of soft tissue. However, it has the disadvantage of a long period required for revascularization between the graft and the recipient site and may cause pathological fractures, pseudoarthrosis, and nonconsolidation, similar to that observed when using allografts15,16.
The external fixation technique takes a long time to treat extensive bone defects and produces long-term complications in patients undergoing bone resections for osteosarcoma due to post-chemotherapy immunosuppression17.
Vascularized bone transplantation is the best reconstruction method. This technique differs from the others because revascularization of the flap occurs immediately on completion of the anastomosis, thereby retaining osteoblastic and osteoclastic activity with the same potential consolidation of a simple fracture, and is thus associated with a remodeling process that leads to rapid flap incorporation18. Use of the free fibula flap is more appropriate for this type of reconstruction, with the advantages being the involvement of a long tubular bone (25 cm), easy dissection, and the low associated morbidity of the donor area.
Chang and Weber19 demonstrated that the use of a free flap associated with an allograft improves this integration, preventing the incidence of nonconsolidation and shortening the functional limb rehabilitation. In this series, we used the allograft in one case, in which ambulation occurred 10 months after surgery.
Clemens et al.20 recently demonstrated a higher rate of consolidation and therefore earlier rehabilitation when only the free flap was used compared to the allograft/free flap (100% vs. 82.4%) and recommended the use of this kind of reconstruction in cancer patients. In this series, 100% of the flaps were viable, there was excellent integration of the flap to the receptor site, and there were no cases of pseudoarthrosis or nonconsolidation. There were 2 cases of delayed flap fracture caused by external trauma, both of which presented with normal consolidation after orthopedic fixation. The mean time to ambulation was 14.2 months, similar to those of global reports2,19,20.
The mean flap size in this study was 15.8 cm (range, 10-20 cm). No evidence of functional impairment was present in the donor area. The mean age of patients was lower than those in more recent studies, which may be most likely due to the small sample size.
Despite the small number of cases in this study, our results are in agreement with reported data showing that the free flap is the method of choice for limb reconstruction in patients with cancer since it provides better consolidation and greater resistance to infection compared to allograft and endoprosthesis, thus promoting earlier rehabilitation and facilitating further treatment of the underlying disease.
CONCLUSIONS
The use of a free fibula flap is an excellent alternative in the therapeutic armamentarium for limb reconstruction after orthopedic resection of oncological long bones since it displays a high bone healing rate, early ambulation, good functionality, and a low complication rate while favoring adjuvant treatment follow-up. The patients in this series had excellent functional and oncological results.
REFERENCES
1. Bach AD, Kopp J, Stark GB, Horch RE. The versatility of the free osteocutaneous fibula flap in the reconstruction of extremities after sarcoma resection. World J Surg Oncol. 2004;2:22.
2. Chen CM, Disa JJ, Lee HY, Mehrara BJ, Hu QY, Nathan S, et al. Reconstruction of extremity long bone defects after sarcoma resection with vascularized fibula flaps: a 10-year review. Plast Reconstr Surg. 2007;119(3):915-24.
3. Weiland AJ, Daniel RK, Riley LH Jr. Application of the free vascularized bone graft in the treatment of malignant or aggressive bone tumors. Johns Hopkins Med J. 1977;140(3):85-96.
4. Evans GRD, Goldberg DP. The management and reconstructive concerns of extremity malignances. Principles of extremity microvascular reconstruction. In: Schusterman MA, ed. Microsurgical reconstruction of the cancer patient. Philadelphia: Lippincott-Raven Publishers; 1997. p.233-47.
5. Donati D, Di Liddo M, Zavatta M, Manfrini M, Bacci G, Picci P, et al. Massive bone allograft reconstruction in high-grade osteosarcoma. Clin Orthop Relat Res. 2000;(377):186-94.
6. Helmstedter CS, Goebel M, Zlotecki R, Scarborough MT. Pathologic fractures after surgery and radiation for soft tissue tumors. Clin Orthop Relat Res. 2001;(389):165-72.
7. Lee KS, Han SB, Baek JR. Free vascularized osteocutaneous fibular graft to the tibia in 51 consecutive cases. J Reconstr Microsurg. 2004;20(4):277-84.
8. Minami A, Usui M, Ogino T, Minami M. Simultaneous reconstruction of bone and skin defects by free fibular graft with a skin flap. Microsurgery. 1986;7(1):38-45.
9. Hsu RW, Wood MB, Sim FH, Chao EY. Free vascularised fibular grafting for reconstruction after tumour resection. J Bone Joint Surg Br. 1997;79(1):36-42.
10. Steinau HU, Buttemeyer R, Vogt P, Hussmann J, Hebebrand D. Limb salvage and reconstructive procedures in soft tissue sarcomas of the extremities. Recent Results Cancer Res. 1995;138:31-9.
11. Arndt CA, Crist WM. Common musculoskeletal tumors of childhood and adolescence. N Engl J Med. 1999;341(5):342-52.
12. Drake DB. Reconstruction for limb-sparing procedures in soft tissue sarcomas of the extremities. Clin Plast Surg. 1995;22(1):123-8.
13. Choong PF, Sim FH. Limb-sparing surgery for bone tumors: new developments. Semin Surg Oncol. 1997;13(1):64-9.
14. Aberg M, Rydholm A, Holmberg J, Wieslander JB. Reconstruction with a free vascularized fibular graft for malignant bone tumor. Acta Orthop Scand. 1988;59(4):430-7.
15. Enneking WF, Morris JL. Human autologous cortical bone transplants. Clin Orthop Relat Res. 1972;87:28-35.
16. Mankin HJ, Gebhardt MC, Tomford WW. The use of frozen cadaveric allografts in the management of patients with bone tumors of the extremities. Orthop Clin North Am. 1987;18(2):275-89.
17. Ozaki T, Nakatsuka Y, Kunisada T, Kawai A, Dan'ura T, Naito N, et al. High complication rate of reconstruction using Ilizarov bone transport method in patients with bone sarcomas. Arch Orthop Trauma Surg. 1998;118(3):136-9.
18. Ozaki T, Hillmann A, Wuisman P, Winkelmann W. Reconstruction of tibia by ipsilateral vascularized fibula and allograft. 12 cases with malignant bone tumors. Acta Orthop Scand. 1997;68(3):298-301.
19. Chang DW, Weber KL. Use of a vascularized fibula bone flap and intercalary allograft for diaphyseal reconstruction after resection of primary extremity bone sarcomas. Plast Reconstr Surg. 2005;116(7):1918-25.
20. Clemens MW, Chang EI, Selber JC, Lewis VO, Oates SD, Chang DW. Composite extremity and trunk reconstruction with vascularized fibula flap in postoncologic bone defects: a 10-year experience. Plast Reconstr Surg. 2012;129(1):170-8.
1. Resident Physician, Plastic Surgery and Microsurgery Department of the Instituto Nacional de Câncer, Rio de Janeiro, RJ, Brazil.
2. Plastic Surgeon, full member of the Associação Brasileira de Cirurgia Craniomaxilofacial/Brazilian Association of Craniomaxillofacial Surgery, associate member of the Sociedade Brasileira de Cirurgia Plástica/Brazilian Society of Plastic Surgery (SBCP), Specialist in Reconstructive Microsurgery in Oncology at the Instituto Nacional de Câncer, Master from the Faculdade de Medicina (FAMED), Preceptor of the Plastic Surgery Residency and Postgraduate Course in Craniomaxillofacial Surgery at the Hospital de Clínicas de Porto Alegre, Porto Alegre, RS, Brazil.
3. Orthopedist, Connective Bone Tissue Service of the Instituto Nacional de Câncer and Instituto Nacional de Traumatologia e Ortopedia, Rio de Janeiro, RJ, Brazil.
4. Orthopedist, Connective Bone Tissue Service of the Instituto Nacional de Câncer and Chief of Orthopedic Oncology Center of the Instituto Nacional de Traumatologia e Ortopedia, Rio de Janeiro, RJ, Brazil.
5. Full member of the SBCP, Master of Medicine, Universidade Estadual de Campinas, Plastic Surgeon at the University Hospital of the Universidade Federal do Rio de Janeiro, Program Coordinator, Residency in Plastic Surgery at the Instituto Nacional de Câncer, Rio de Janeiro, RJ, Brazil.
6. PhD, Full Member of the SBCP, Plastic Surgeon, Plastic Surgery and Microsurgery Service of the Instituto Nacional de Câncer, President of the SBCP - Regional Rio de Janeiro, Rio de Janeiro, RJ, Brazil.
Correspondence to:
Eduardo Ravasio Machado
Rua Antonio Parreiras, 148 - ap. 302 - Ipanema
Rio de Janeiro, RJ, Brazil - CEP 22411-020
E-mail: eduravasio@gmail.com
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: May 12, 2012 Article
accepted: August 7, 2012
This study was performed at the Instituto Nacional de Câncer, Rio de Janeiro, RJ, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter