

Special Article - Year 2012 - Volume 27 -
Laboratory training program in microsurgery at the National Cancer Institute
Rotina de treinamento laboratorial em microcirurgia do Instituto Nacional do Câncer
ABSTRACT
Microsurgery is a technique in which the surgery is performed under optical magnification in vessels with a diameter of less than 3 mm. In 1960, Jacobson and Suarez first used the term "microsurgery" to describe the experimental anastomosis of vessels with a caliber between 1 and 2 mm, which is considered as the origin of the modern microvascular practice. Since then, several types of microvascular tissue transfer techniques for the repair of large body defects have been developed and published, accompanied by significant advances in optical imaging technologies and instrument design. Despite this technical progress, laboratory practice is essential and enables the surgeon to acquire the ability to master the microanastomosis technique. The present study describes the microsurgery training program of the Laboratory of Experimental Microsurgery of the National Cancer Institute (Rio de Janeiro, RJ, Brazil).
Keywords: Microsurgery. Reconstructive surgical procedures. Training. Plastic surgery.
RESUMO
A microcirurgia é uma técnica na qual se realiza cirurgia sob magnificação óptica em vasos de diâmetro < 3 mm. Jacobson e Suarez, em 1960, foram os primeiros a utilizar o termo microcirurgia para descrever anastomoses experimentais de vasos com calibre entre 1 mm e 2 mm, sendo a origem da moderna prática microvascular creditada a eles. Desde então, foram desenvolvidos e publicados diversos tipos de transferência microvascular de tecidos para reparo de grandes defeitos corporais e ocorreu intensa modernização do poder óptico e do design dos instrumentos. Apesar dessa evolução, a prática laboratorial é indispensável e permite ao cirurgião alcançar a habilidade necessária à realização da técnica de microanastomoses. Nesse contexto, este artigo apresenta a rotina de treinamento em microcirurgia realizada no Laboratório de Microcirurgia Experimental do Instituto Nacional do Câncer (Rio de Janeiro, RJ, Brasil).
Palavras-chave: Microcirurgia. Procedimentos cirúrgicos reconstrutivos. Capacitação. Cirurgia plástica.
Microsurgery is defined as surgery performed under optical magnification, commonly under the surgical microscope. One century has passed since Carrel introduced the triangulation techniques for vessel repair in 1902. In 1921, Nylen operated on rabbits' labyrinth, followed by clinical use of otology, ophthalmology and neurosurgery, in procedures that required magnification for accurate dissection1.
The origin of the modern microvascular practice is credited to Jacobson and Suarez, who used the surgical microscope of their otorhinolaryngology colleagues during the 1960s to perform the anastomosis of vessels with diameter < 2 mm. The first replantation of an arm with microvascular repair was performed in 1963 by Chen. This procedure was published only in the Chinese literature, and in 1968, Tamai and Komatsu reported the first successful thumb replantation. In 1969, Cobbet performed the first toe transference for thumb reconstruction in England2-4.
Ferreira et al.5 performed the first successful hand replantation in humans in Brazil in 19726. More elaborate microvascular tissue transference techniques were developed and published later. Current research in the field of microsurgery is aimed at developing new flap designs and refining existing techniques for application in a variety of reconstructions7-11. However, in addition to technological advances, the clinical success of microsurgical procedures also depends on the surgeon's ability to perform microvascular anastomoses.
Adherence to basic rules during vessel preparation and suturing will always be the cornerstone of a good job in microsurgery11. The diameter of anastomosed vessels has progressively become smaller, and reaching permeability rates of 98% in the anastomosis of vessels of 1 mm in diameter is now common owing to the recent advances in microscopy, the improvements in the design of microsurgery instruments and the implementation of laboratory training programs by pioneers such as Acland and Buncke.
Microsurgery is commonly used for the re-implantation of limbs or fingers after traumatic amputation, to free vascularized flaps in reconstructive plastic surgery, for the rechanneling of vas deferens and uterine tubes, and in specialized fields such as neurosurgery, ophthalmology, orthopedics and otorhinolaryngology12. In Brazil, there are no training centers or regular microsurgery courses in most of the States. In this context, one of the main obstacles is the cost of the training. However, this does not diminish the importance of existing regional microsurgery centers, because in emergency cases, such as traumatic amputation, patients often cannot be transferred to a specialized center for reimplantation within a reasonable amount of time7.
Training requires a high degree of dedication and teaching proper laboratory practices is the first step required to master the technique prior to its application in clinical practice because experimentation using materials and live animals is essential for the development of surgical skills. Several training models are currently available that involve different materials and animals6. Certain training programs were established with the aim of achieving suitable vascular permeability, and consequently to ensure the successful performance of surgery in clinical practice6,7,13-16.
The present study describes the microsurgery training program completed by trainee physicians in the Department of Plastic Surgery and Reconstructive Microsurgery at the Experimental Microsurgery Laboratory of the National Cancer Institute (INCA - Rio de Janeiro, RJ, Brazil).
STRUCTURE OF THE MICROSURGERY LABORATORY
The Experimental Microsurgery Laboratory of the INCA exists since 1981, when was created the first Reparative Microsurgery Service in Brazil, at INCA, to operate independently of the other specialties, through comanagement INAMPS-Ministério da Saúde the administration of Dr. Ary Frauzino4. It is equipped with has binocular microscopes and includes an animal laboratory with Wistar rats, in addition to the instruments required for basic training (curved needle holder without lock, straight and curved clamps for dissection, curved and straight scissors, a vessel dilating forceps and microvascular clamps). The laboratory has been approved by the Ethics and Research Committee (Figures 1 to 3).

Figure 1 - Experimental Microsurgery Laboratory of the National Cancer Institute (Rio de Janeiro, RJ, Brazil).

Figure 2 - Bench with microscopes.

Figure 3 - Animal laboratory: shelf with animals.
TRAINING PROGRAM IN MICROSURGERY
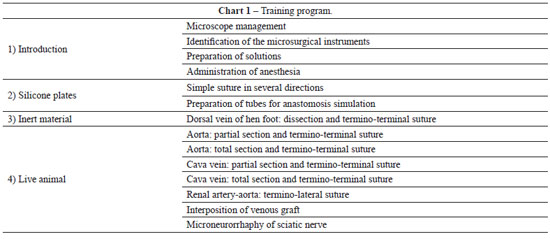
The training program is divided into several stages, which are implemented in increasing levels of difficulty in a twice weekly, 4 hours per day schedule. In the first stages, the trainee learns the handling of the microsurgical instruments and the microscope with a minimum magnification of 16x. The surgeon receives instruction on proper positioning and posture, which includes maintaining the elbows at 90 degrees of flexion with support of the forearms and wrists with the purpose of achieving two-dimensional visualization of the objects while preventing the incidence of fatigue and tremors.
The first step of the training consists of the use of silicone plates that are sutured using 8-0 and 9-0 mononylon sutures for 20 hours. The plates are sectioned and positioned in different orientations during the training, with the purpose of gradually increasing the degree of difficulty and simulating real life situations (Figure 4). Trainees first receive instruction on the continuous whipstitch suture, followed by training on the performance of simple sutures (Figure 5). Next, the plate is sectioned with two parallel cuts to generate tubes, which are used for simulation of termino-terminal anastomosis (Figure 6).

Figure 4 - Silicone plate: practice of sutures in several directions.

Figure 5 - Silicone plate: simple suture.

Figure 6 - Silicone plate: preparation of tubes for anastomosis simulation.
The next stage involves training with animals for 20 hours using hen feet, which have a dorsal medial vein with a diameter of 1 to 2 mm. The surgeons are instructed on dissection techniques and vessel preparation before performing the termino-terminal anastomosis between microvascular clamps (Figure 7).

Figure 7 - Hen foot: practice of dissection and termino-terminal suture.
The last stage, which requires 60 hours of training, involves live animals. The animal used is the Wistar rat. Prior to this stage, the trainee learns how to handle animals and anesthetize them for the procedure. Grain gloves are used for protection while handling the animals and exposing the abdomen for the intraperitoneal administration of the anesthetic solution. The anesthetic solution commonly used is chlorpromazine (1.7 mg/kg/dose) and ketamine (120 mg/kg/dose). Prior to the procedure, a heparinized solution is prepared consisting of 5 ml (5,000 UI/ml) diluted in 100 ml of Ringer's solution, which is used to irrigate the lumen of the vessels to remove impurities and blood clots. Lidocaine solution at 2% is used to irrigate the vessels and reduce vasospasm, and warm saline at 0.9% is used for irrigation of the intestinal loops and to clean the surgical site. For the anastomosis, 8, 9 and 10-0 monofilament nylon sutures are used, and the abdomen is sutured at the end with 4-0 catgut in the peritoneomuscular plane, and with 4.0 Vicryl for skin sutures. Oral Paracetamol is used as postoperative analgesia.
The materials and the solutions are prepared and positioned near the surgical site prior to the procedure (Figure 8).

Figure 8 - Prepared surgical field.
The initial procedures are performed using a midline abdominal incision, folding the peritoneum and the intestinal loops to the left, and covering them with gauze imbibed in 0.9% warm saline. The abdominal wall is retracted using a Weitlaner retractor (Figure 9) and the vessels are prepared by means of microdissection. First, the abdominal aorta is dissected the infra or suprarenal segments are prepared for the positioning of clamps. Improvement of the visual field is achieved with a green sterile piece of glove or the suture thread envelope, which are placed under the vessels and underlying tissues to expose the surgical site. The training initially includes partial section at 50% of the anterior circumference and simple sutures between the clamps (Figure 10). The needle should enter perpendicular to the vessel plane, at a distance from the edge equal to double the vessel thickness including the tunica intima. The suture should evert the edges and tied without excessive tension. After this skill has been mastered, the trainee performs a total section of the vessel (Figure 11) and termino-terminal anastomosis with simple sutures. The first suture is performed in a section of the anterior wall that is considered easy, and the second suture is placed at a third of the distance or at 120 degrees from the first17. The clamp is turned 180 degrees and another suture is made at 120 degrees from the others4,17. Through the triangulation technique, the anterior wall is sutured on both sides to reach the first sutures (Figure 12), thus entirely closing the anterior wall after returning the clamp to the initial position (Figure 13). Finally, the test of vascular patency is conducted using two clamps that are carefully retracted, releasing the flow into the interior of the vessel (Figure 14). Next, the proximal clamp in the case of arteries or the distal clamp for veins is released and blood flow through the anastomosis is confirmed (Figure 15). Venous anastomosis is performed with a termino-terminal suture using the vena cava (Figures 16 and 17) and renal (Figures 18 and 19) vein, with the same procedure described above for arterial anastomosis. After this stage, the trainee advances to the supervised clinical practice for microanastomosis training. The technical training continues at the laboratory with increasing levels of difficulty, including advanced techniques such as termino-lateral anastomosis between the renal artery and the aorta, venous graft interposition and microneurorrhaphy of the sciatic nerve.

Figure 9 - Midline abdominal incision.

Figure 10 - Partial section of the aorta and suture.

Figure 11 - Total section of the aorta.

Figure 12 - Total section of the aorta: the anterior wall is sutured after the posterior wall.

Figure 13 - Total section of the aorta: completed anastomosis.

Figure 14 - Patency test: clamps are retracted.

Figure 15 - Patency test: the flow is verified by releasing the proximal clamp in arteries (shown here) and the distal clamp in the case of veins.

Figure 16 - Suprarenal vena cava: positioning on the clamp before total section.

Figure 17 - Suprarenal vena cava: completed termino-terminal suture above the fragment of green glove used for contrast is observed.

Figure 18 - Right renal vein (with caliber of 1 mm): the underlying tissues are shown with a fragment of the green envelope of the suture thread.

Figure 19 - Right renal vein: completed termino-terminal anastomosis after total section.
The laboratory training occurs simultaneously with instruction at the surgical center, where the trainee participates in surgeries involving the use of free flaps for correction of several defects deriving from oncological resections and traumatic sequelae, among others.
RESIDENTS' PERFORMANCE
There was variation in the vascular patency indexes achieved by the residents, as assessed by the vessel filling test of Acland. These values were within the range reported in the literature (85% to 95%)14 and were considered a reflection of adequate preparation for clinical practice at the institution.
The structure of this training program, characterized by steps of increasing difficulty, adequately prepares the trainees for performing microanastomosis and free flap dissection in patients in a safe way, with supervision at the teaching hospital. Therefore, they become capable of resolving a variety of situations commonly encountered in their daily routine of reconstructive plastic surgery in which microvascular transfer of tissues is required.
DISCUSSION
The initial training in microsurgery can be long and tedious if the teaching program does not involve adequate planning. The training program is first implemented in an environment without patients, due to the complexity of the procedures being taught18. At each stage, of the training, the trainees are encouraged by the progressive gain of manual skills. Thus, the training program established in our institution enables the resident to gradually become familiar with the tactics and techniques involved in the performance of microanastomosis, becoming prepared to perform in the clinical practice.
Pessoa & Pessoa18 reported that at the beginning of the training it is important to learn certain theoretical notions on how to deal with the instruments and the microscope, as well as dissection and suturing techniques, and the administration of anesthesia in rats. The exercises should progressively increase in their level of difficulty to ensure better adaptation to the microscope and the instruments; thus, the early use of live animals in the training is not advised13. During the initial stages, it is not important to work under conditions that simulate "real life situations" with live animals because in addition to ethical and financial considerations, several animals can be saved.
The use of synthetic materials such as silicone is adequate for the initial training, although it does not accurately mimic the consistency of biological tissues, and it limits the possibilities of practicing dissection techniques. Sutures are performed in a variety of directions, as shown in Figure 4. At this stage, the trainee can practice proper positioning and learn methods of performing sutures from different directions, angles and inclinations, thus avoiding bad habits in positioning, which is often required in clinical practice when the microanastomosis site is not favorable.
The use of animal parts, which can be stored for several days in the refrigerator without losing the consistency of the tissues, is very useful19. Several inert segments can be used for training20-24, such as hen feet, which are easily obtained at places where they are offered for consumption, with the advantages of low cost and easy storage. This stage of the training is important to learn how to handle delicate microvascular structures and perform sutures without stress, which can be a limiting factor when working with live animals. In the training involving live animals, several factors such as small volume losses, prolonged surgical time and accidental lesions caused by trainees without enough experience can result in the death of the animals, which can increase the costs of the training.
Live animal work and the maintenance of an animal laboratory can be costly. In Brazil, although several renowned institutions have training programs in microsurgery, their economic resources are limited6,7. In some European and North American countries, the greatest obstacles are related to ethical issues associated with the prohibition of the use of animals for surgical training. The implementation of the training at the initial stages with inert materials and animal parts minimizes the need for the use of animals at the final stages of the training, thus reducing costs. Despite the difficulties associated with live animal work, it is essential to properly learn the surgical techniques, and the high cost is justified by the benefits associated with this training. Learning with live animals helps the resident become familiar with situations that are similar to those faced in clinical practice, as he or she learns to correctly expose, dissect and prepare the vessels in vivo, and apply vascular microsuture techniques.
In working with live animals, it is important to pay special attention to certain items before the procedure, such as separating the surgical materials and the necessary solutions. Moreover, the administration of anesthesia deserves special attention to minimize the suffering or loss of the animal. This procedure saves time and materials and also avoids accidents. To fully utilize available resources, the animals undergo surgery twice, and in cases of a third procedure, the animal is sacrificed.
The aorta and vena cava of mice range in caliber from 1 to 2.5 mm depending on the age of the animal. The aorta is used first because it has a larger caliber and greater wall thickness, which makes suturing easier. However, the enhanced elasticity of this vessel causes the vascular stumps to become distanced from each other when a complete transverse section is performed, which increases the difficulty of the suture for the beginner. Therefore, during the beginning stages, the vessel is partially sectioned, which involves a lower degree of difficulty. After the student is familiar with the procedure, the next step is the total section of the vessel followed by suturing first in the anterior wall, which is technically more difficult for the beginner. Next, the same procedure is used for the veins, which are larger in diameter but have thin and fragile walls that can be easily damaged even by the insertion of the needle. Therefore, this involves a greater degree of difficulty and increased stress during the performance of termino-terminal anastomosis or venous interposition. Microneurorrhaphy is trained using the sciatic nerve because it is located at the root of the animals' thigh, making it easily accessible for the performance of termino-terminal epi-perineural sutures25,26.
We have observed that the trainees acquire the necessary technical skills naturally and achieve adequate vascular patency indexes, as assessed by the vessel filing test described by Acland (Figures 14 and 15) and also by the animal's survival at each stage of the training program (Chart 1).
It is important that trainees do not perform microanastomosis in the clinical practice until they achieve successful results regarding vascular patency, with rates greater than 80% with vessels of similar caliber in the laboratory13. As the trainees progress in the training program and achieve patency rates that are comparable to those reported in the literature, they can begin to learn the preparation of the receptor vessels and perform vascular microanastomosis in the clinical practice, under supervision of the responsible physician. Certain courses in the United States are organized in 2 days with 7 hours of training per day. This type of training is not considered beneficial, as the training is too short and exhausting. Among beginners, the exposure to the microscope for hours is commonly associated with headaches, muscular pain, tremors and loss of concentration; moreover, microsurgery is associated with details and tricks, and there is too much information to learn in only two days27. A training program of suitable length enables the progressive training of the residents and the acquisition of manual skills in preparation for supervised clinical practice.
CONCLUSION
Training in microsurgical techniques is not commonly associated with an easy transition between the experimental and clinical practice28. The establishment of a training program in which the trainee is guided through the microanastomosis techniques in increasing levels of difficulty maximizes the learning process and keeps the students motivated. All stages of the training are important, especially the final phases consisting of work with live animals, which allow the trainee to experience a "real life" situation and to work under stress in conditions similar to those present during the performance of a microanastomosis prior to working in clinical practice. The high cost associated with the use of experimental animals is justified because training and developing the necessary skills during the first stages of the training minimizes the use of animals in the next stages, limiting the requirement to the achievement of acceptable vascular patency for performance in clinical practice.
The establishment of laboratories that include animals for the practice of surgical techniques should be promoted in Brazil and the conditions of existing laboratories should be improved, as there are excellent plastic surgeons that could learn to master the techniques of microanastomosis if provided with a basic program of supervised training. This could result in a pool of competent surgeons that could provide the users of the healthcare system with good quality care with respect to reconstructive surgery, including the performance of microvascularized flaps for repair of specific defects that are commonly performed in large centers.
Finally, mastering the microanastomosis technique is only an important cofactor in the success of tissue transference surgeries. Only years of surgical experience and facing a variety of clinical situations will enable the surgeon to achieve greater rates of success in these highly complex surgeries.
REFERENCES
1. Ferreira MC. Microcirurgia reconstrutiva: a história da microcirurgia no Brasil. In: Bijos P, Zumiotti AV, Rocha JR, Ferreira MC, eds. Microcirurgia reconstrutiva. São Paulo: Atheneu; 2005. 516 p.
2. Galvão MSL, Braga ACCR, Souza JRW. A contribuição da microcirurgia reparadora no tratamento do paciente oncológico. Rev Bras Cancerol. 1984;30(4):24-34.
3. Galvão MSL. The role of reconstructive microsurgery in cancer surgery. In: Fifth Congress of the European Section of the International Confederation for Plastic and Reconstructive Surgery. Stockholm, Sweden, 1985.
4. Galvão MSL, Cardoso MM, Köbig RN. Microcirurgia. In: Saad Jr R, Salles RARV, Carvalho WR, Maia AM, eds. Tratado de cirurgia do Colégio Brasileiro de Cirurgia. São Paulo: Atheneu; 2009. p. 1483-92.
5. Ferreira MC, Marques E, Tedesco-Marchese AJ. Microcirurgia vascular: técnica para sutura de vasos com diâmetro externo inferior a 2 mm. Rev Paul Med. 1974;38(1):67-8.
6. Dias IS, Pessoa SGP, Benevides AN, Macedo JE. Treinamento inicial em microcirurgia. Rev Bras Cir Plást. 2010;25(4):595-9.
7. Webster R, Bins-Ely P. Treinamento em microcirurgia vascular: é economicamente viável? Acta Cir Bras. 2002;17(3):194-7.
8. Cunha MS, Torre ALG, Anjos Neto JC, Monteiro LL, Meneses JV. Transplantes microcirúrgicos: experiência de 5 anos do Serviço de Cirurgia Plástica da Universidade Federal da Bahia. Rev Bras Cir Plást. 2008;23(4):305-9.
9. Souza Filho MVP, Santos CC. Microcirurgia em reconstruções complexas: análise dos resultados e complicações. Rev Bras Cir Plást. 2009;24(2):123-30.
10. Torres ALG, Milcheski DA, Nakamoto HA, Tuma Jr P, Ferreira MC. Aplicação da microcirurgia no reparo de lesões complexas. Rev Bras Cir Plást. 2009;24(2):131-7.
11. Sabapathy SR. Vessels. In: Wei FC, Mardini S, eds. Flaps and reconstructive surgery. Philadelphia: Saunders Elsevier; 2009. p. 81-92.
12. Di Cataldo A, Li Destri G, Trombatore G, Papillo B, Racalbuto A, Puleo S. Usefulness of microsurgery in the training of the general surgeon. Microsurgery. 1998;18(8):446-8.
13. Martins PN, Montero EF. Basic microsurgery training: comments and proposal. Acta Cir Bras. 2007;22(1):79-81.
14. Samaha FJ, Oliva A, Buncke GM, Buncke HJ, Siko PP. A clinical study of end-to-end versus end-to-side techniques for microvascular anastomosis. Plast Reconstr Surg. 1997;99(4):1109-11.
15. Aston SJ, Beasley RW, Thorne CHMl. Plastic surgery. Philadelphia: Lippincott-Raven Publishers; 1997.
16. Rocha JR. Manual de microcirurgia experimental. Ed. Serviço de Microcirurgia Reconstrutiva do HSE.
17. Cobbet JR. Microvascular surgery. Grabb and Smith's Plastic Surgery. Boston: Little, Brown; 1968.
18. Pessoa BBGP, Pessoa SGP. O retalho hipogástrio cutâneo no cão: modelo para o aprendizado experimental de microcirurgia. Acta Cir Bras. 2002;17(3):198-202.
19. Yasargil MG. From de microsurgical laboratory to the operation theatre. Acta Neurochir. 2005;147(5):465-8.
20. Phoon AF, Gumley GJ, Rtshiladze MA. Microsurgical training using a pulsatile membrane pump and chicken thigh: a new, realistic, practical, nonliving educational model. Plast Reconstr Surg. 2010;126(5):278e-9.
21. Schoffl H, Hager D, Hinterdorfer C, Dunst KM, Froschauer S, Steiner W, et al. Pulsatile perfused porcine coronary arteries for microvascular training. Ann Plast Surg. 2006;57(2):213-6.
22. Aboud E, Al-Mefty O, Yaşargil MG. New laboratory model for neurosurgical training that simulates live surgery. J Neurosurg. 2002;97(6):1367-72.
23. Colpan ME, Slavin KV, Amin-Hanjani S, Calderon-Arnuphi M, Charbel FT. Microvascular anastomosis training model based on a Turkey neck with perfused arteries. Neurosurgery. 2008;62(5 Suppl 2): ONS407-10.
24. Hino A. Training in microvascular surgery using a chicken wing artery. Neurosurgery. 2003;52(6):1495-7.
25. Galvão MSL. Sutura e enxerto de nervo facial. In: Freire E, ed. Trauma: a doença dos séculos. Vol. 2. São Paulo: Atheneu; 2001.
26. Galvão MSL, Sá GM, Farias T, Anlicoara R, Dias FL, Sbalchiero JC. Reconstrução tridimensional da face nos tumores avançados com invasão da fossa craniana anterior. Rev Col Bras Cir. 2004;31(2):124-31.
27. Montero EFS, Simão AFL, Chagas Neto FA, Barroso TA. Microsurgery training using a PVC rat model. In: Book of abstracts of the 8th Congress of the International Society for Experimental Microsurgery; 2006 July 16-19. Montreal, Canada.
28. Zumiotti AV, Rames Mattar Jr R, Rezende MR, Santos GB. Manual de microcirurgia. São Paulo: Atheneu; 2008.
1. Plastic surgeon, member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery) - SBCP, post-graduate student in Reconstructive Microsurgery at the Instituto Nacional do Câncer (National Cancer Institute), Rio de Janeiro, RJ, Brazil; plastic surgeon at Hospital Regional do Câncer and at Santa Casa de Misericórdia de Passos, Passos, MG, Brazil.
2. Founder and member of the Reconstructive Microsurgery Unit of the Instituto Nacional do Câncer (National Cancer Institute), head of the Reconstructive Microsurgery Unit of the Instituto Nacional do Câncer (National Cancer Institute),, Rio de Janeiro, RJ, Brazil.
3. Plastic surgeon, member of SBCP, member of the Department of Plastic Surgery and Reconstructive Microsurgery of the Instituto Nacional do Câncer (National Cancer Institute), supervisor responsible for the Experimental Microsurgery Laboratory of the National Cancer Institute Instituto Nacional do Câncer (National Cancer Institute), Rio de Janeiro, RJ, Brazil.
4. Doctor, member of the SBCP, head of the Department of Plastic Surgery and Reconstructive Microsurgery of the Instituto Nacional do Câncer (National Cancer Institute), president of SBCP - Rio de Janeiro Section, Rio de Janeiro, RJ, Brazil.
Diogo Almeida Lima
Rua Barão de Passos, 128 - ap. 101
Passos, MG, Brazil - CEP 37900-048
E-mail: drdiogolima@hotmail.com
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: January 30, 2012
Article accepted: March 29, 2012
Study conducted at the Instituto Nacional do Câncer (National Cancer Institute), Rio de Janeiro, RJ, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter