

Original Article - Year 2017 - Volume 32 -
Aggressive fibromatosis (desmoid tumor) associated with breast implant: literature review and presentation of three new cases
Fibromatose agressiva (tumor desmoide) associada ao implante mamário: revisão da literatura e apresentação de três novos casos
ABSTRACT
INTRODUCTION: Fibromatoses consists of the proliferation of fibrous tissue, in the form of bands or nodules, associated with fasciae and aponeuroses. The aggressive variety, previously denominated desmoid tumor, behaves similarly to malignant neoplasm, with local destruction of tissues, without, however, producing metastases.
METHODS: A literature review was carried out from 1979 to January 2017. Data from three patients, operated between May 2010 and August 2015, were reviewed. Age, implant characteristics, route of introduction, time elapsed between implantation and fibromatosis, surgical treatment, clinical follow-up and results were observed.
RESULTS: Twenty-four papers were found in the literature reporting aggressive fibromatosis of the chest wall associated with silicone breast implant. In these studies, 34 cases were reported. Three new cases of aggressive fibromatosis associated with silicone breast implant are now presented. These cases were successfully treated by extensive resection of the chest wall, including skin, musculature, ribs, endothoracic fascia, and parietal pleura. The reconstruction was successful, performed with alloplastic mesh (Prolene®) covered by muscular flap in two cases and local skin flap in one case.
CONCLUSION: The association of aggressive fibromatosis and breast implant is rare. The treatment should consist of extensive surgery, removing the breast implant and the entire area of the capsule around it, part of breast, together with the underlying ribs, intercostal muscles, endothoracic fascia and parietal pleura. The reconstruction should be made with an alloplastic mesh, covered by muscular flaps or local skin flap.
Keywords: Fibromatosis aggressive; Breast implantation; Mammaplasty.
RESUMO
INTRODUÇÃO: As fibromatoses consistem na proliferação de tecido fibroso, na forma de faixas ou nódulos, associadas às fáscias e aponeuroses. Sua variedade agressiva, denominada previamente tumor desmoide, se comporta de forma semelhante a uma neoplasia maligna, com destruição local de tecidos, sem, entretanto, originar metástases.
MÉTODOS: Fez-se revisão da literatura de1979 a janeiro de 2017. Dados de três pacientes, operados entre maio de 2010 e agosto de 2015, foram revistos, com proservação até março de 2017. Observaram-se idade, características do implante, via de introdução do mesmo, tempo decorrido entre o implante e o aparecimento da fibromatose, tratamento cirúrgico instituído, acompanhamento clínico e resultados.
RESULTADOS: Foram encontrados 24 trabalhos na literatura disponível, versando sobre fibromatose agressiva da parede torácica associada ao implante mamário de silicone, nos quais foram relatados 34 casos. São apresentados três casos de fibromatose agressiva associados ao implante mamário de silicone. Os casos foram tratados com sucesso por ressecção alargada da parede torácica, incluindo pele, musculatura, costelas, fáscia endotorácica e pleura parietal. A reconstrução foi bem-sucedida, realizada com tela aloplástica (Prolene®) recoberta por retalho muscular em dois casos e retalho cutâneo local em um caso.
CONCLUSÃO: A associação de fibromatose agressiva e implante mamário é rara. O tratamento deve consistir em cirurgia alargada, removendo-se o implante e toda a área da cápsula adjacente, em conjunto com parte da mama, costelas subjacentes, musculatura intercostal, fáscia endotorácica e pleura parietal. A reconstrução deve ser feita com tela aloplástica associada a retalho muscular ou retalho tegumentar local.
Palavras-chave: Fibromatose agressiva; Implante mamário; Mamoplastia.
The first case of desmoid tumor (aggressive fibromatosis) was described by McFarlane (1832)1, who reported the disease occurring in the anterior abdominal wall in a young woman after childbirth. According to Dorland's Illustrated Medical Dictionary2, the word "desmoid" is derived from Greek (desmos = ligament + eidos = form), i.e., the tumor has a ligament form.
The term "desmoid" was coined by Müller, in 18383. According to Kallam et al.4, desmoid tumors are classified in three types according to their location. The first one is the abdominal, which affects the anterior abdominal wall; the second type is the intra-abdominal, that affects the mesentery and the pelvis, in intra or retroperitoneal situation; the third type is the extra-abdominal, that affects the chest, extremities, head and neck.
From and Assaad5 studied aggressive fibromatoses (AF) or desmoid tumors, noting that the disease may affect several regions of the body, more often the anterior wall of the abdomen and the shoulder girdle. It is more common in puerperae and in places of abdominal scarring. Desmoid tumors grow relentlessly, invade surrounding tissues, and can reach vital organs. Retroperitoneal desmoid tumors occur in Gardner's syndrome, especially after intra-abdominal operations.
Gardner's syndrome6, (familial adenomatous polyposis FAP), was described in 1951, initially called familial polyposis coli (FPC). It consists of polyposis of the cervix subject to malignization, osteomas of the skull and mandible, epidermoid cysts, desmoid tumors, tumors of the ovary, small intestine, thyroid and adrenal. The syndrome is associated with the mutation of an autosomal dominant tumor suppressor gene called APC (adenomatous polyposis coli) located on the long arm of chromosome 56.
According to Weiss7, the desmoid tumor is known as "deep or muscle-aponeurotic fibromatosis". The disease mainly affects the abdominal wall and abdominal cavity, the scapular girdle and the anterior and posterior thoracic walls. About 1% of aggressive fibromatoses are familial, although unrelated to Gardner's syndrome7. Lesions that appear in childhood often occur in the head and neck.
The histopathological picture shows large fascicles of dense fibrous tissue with abundance of collagen (Figure 1). Among the collagen fibers, there are spindle-shaped cells with thin, conical nuclei at their extremities. Nuclei have no atypia and have a vesicular pattern of chromatin. The mitotic index is low. Immunohistochemistry reveals that spindle cells are positive for intracellular betacatenin, muscle-specific actin, and the smooth muscle alpha isoform7.
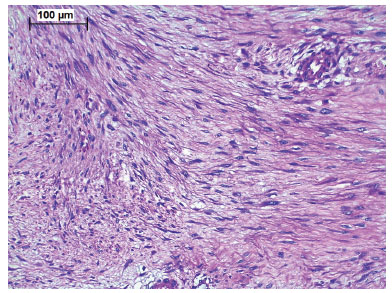
Figure 1. Photomicrography in hematoxylin-eosin: fibromatosis invading muscle tissue.
Many cases show receptors for estrogen and progesterone7. Hormone therapy has been indicated to control the neoplasia8-11. In the differential diagnosis, nodular fasciitis, neurofibroma, fibrosarcoma and low grade fibromyxoid sarcoma should be considered. Intranuclear betacatenin expression is highly specific for aggressive fibromatosis and is not observed in other fusiform cell lesions7 (Figure 2).
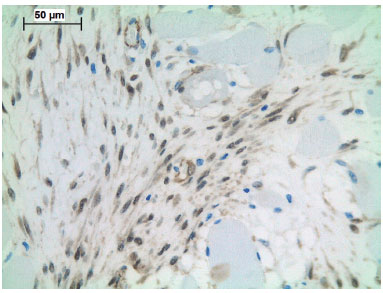
Figure 2. Photomicrography of immunohistochemistry: β-catenin expression.
In Brazil, the first report of desmoid tumor associated with aesthetic surgery was made by Caldeira et al (2006)12, who described a case of neoplasia in the abdominal wall of a woman in the right hypochondrium region, after liposuction. The first case of desmoid tumor of the chest wall associated with silicone breast implant was reported by Jewett Jr and Mead (1979)13. In the consulted literature, 24 studies report the association of breast implant and aggressive fibromatosis. According to MeSH (medical subject headings), a database that supports research in PubMed, the US National Library of Medicine, the descriptor "desmoid tumor" was replaced by "aggressive fibromatosis" (AF) in 1994.
OBJECTIVE
To review the literature from the first case published in 1979 until January 2017. Present three new cases of aggressive fibromatosis (AF) associated with silicone breast implants.
METHODS
A literature review was carried out from 1979 to January 2017. Twenty-four published papers with the association FA and breast implant were found, describing 34 cases. The following key-words were investigated: fibromatosis, aggressive; desmoid, breast implants, mammoplasty, breast implantation. The main data for all 34 patients reported were summarized in Chart 18,9,11,13-33.
Data from three patients, operated on from May 2010 to August 2015, two of them treated by the Plastic Surgery and Thoracic Surgery clinics at Hospital Felício Rocho, and one treated at Hospital Júlia Kubitschek (Fundação Hospitalar do Estado de Minas Gerais), in Belo Horizonte, MG, were reviewed retrospectively. Age, implant characteristics, route of introduction, time elapsed between implantation and the onset of fibromatosis, surgical treatment, clinical follow-up and results were observed.
There were no conflicts of interest and no funding sources. The principles of the Declaration of Helsinki revised in 2000 and Resolution 196/96 of the National Health Council were followed. All patients received the Informed Consent Form.
RESULTS
Twenty-four papers reporting 34 cases of FA associated with silicone breast implant were found. Chart 1 summarizes data regarding age, characteristics of the implant, route of introduction, time elapsed between the introduction of the implant and the appearance of AF, surgical treatment, clinical follow-up and results.
The three new cases reported in the present study were operated with the removal of the prosthesis and enlarged resection of the affected thoracic wall, comprising the capsule formed around the implants, breast tissue, underlying muscles, intercostal muscles, rib and sternum segments, endothoracic fascia and parietal pleura. The lateral resection margin was 5 cm. In these cases the parietal pleura had not been reached.
Thoracic wall reconstruction was performed by using the Prolene® alloplastic mesh, latissimus dorsi flaps in two cases and skin flap in one case. In case 1, the patient presented 82 months of disease-free follow-up. At 4 years and 8 months of follow-up, the patient received new silicone breast implants. In case 2 the patient developed mild paradoxical breathing and is waiting for reinforcement of the chest wall to treat the complication.
The mean follow-up of the three patients was 45 months.
Cases report
Case 1
Female, born on 09/04/1980. On May 21, 2008, the patient was received textured silicone gel implants submuscularly (Sebbin®, 245 ml) for the treatment of mammary hypoplasia. Previously, on 12/12/2003, she had undergone treatment of ulcerated extensive superficial melanoma (Breslow 1 mm, Clark 3) in the right parasternal pectoral region, with biopsy of a negative right axillary sentinel lymph node.
Twenty-three months later, aged 29 years and five months, she presented a tumor in the left mammary region, lateral sector, measuring 4.7 x 2.3 cm, as determined by magnetic resonance imaging. The biopsy revealed aggressive fibromatosis. The enlarged resection of the lesion was performed on 05/06/2010, which consisted of thoracectomy including skin, subcutaneous tissue, breast tissue, major pectoralis muscle segments, rib segments (2nd, 3rd and 4th), removal of the implant and the entire surrounding capsule, intercostal muscles, endothoracic fascia, and parietal pleura.
The lateral resection margin was 5 cm. The entire lesion was excised in monoblock. The tumor occupied the lateral portion of the implant capsule. Reconstruction was performed using alloplastic mesh (Prolene®) and latissimus dorsi muscular flap. Conventional thoracic drainage was used for four days. Anatomopathological examination showed the lesion measured 7cm in diameter. The surgical specimen presented disease-free margins both laterally and in depth, confirming the diagnosis of aggressive fibromatosis. The patient presented seroma in the operated area that was treated through successive aspirations.
The patient became pregnant and, on 02/09/2012, she gave birth to a healthy, female child through cesarean section and breastfed in both breasts for two months. On 01/13/2015, the implant was replaced on the right side and new textured Eurosilicone® implants were placed (240 ml left and 220 ml right). On January 23, 2017 the patient was disease free after enlarged resection treatment, presenting 80 months follow-up (Figures 3 to 10).
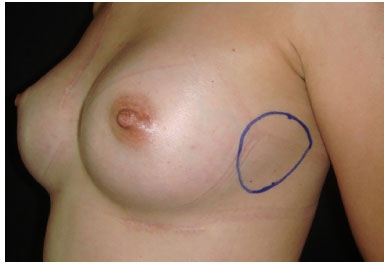
Figure 3. Location of neoplasia on the lateral side of the chest.
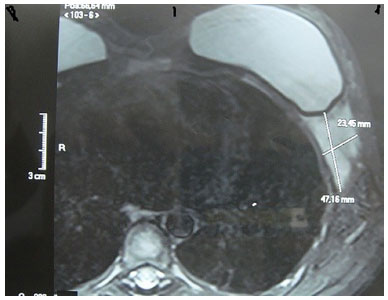
Figure 4. Tumor imaging by magnetic nuclear resonance.
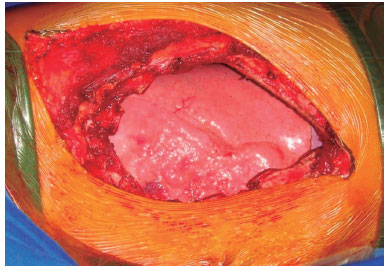
Figure 5. Loss of substance from the chest wall.
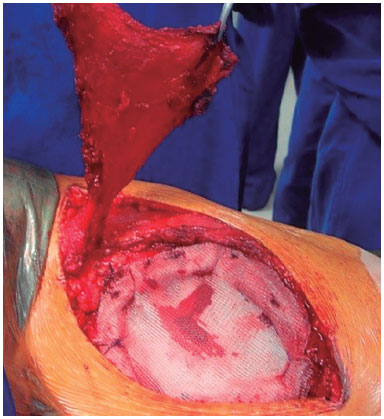
Figure 6. Latissimus dorsi muscle flap.
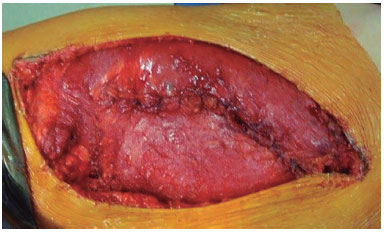
Figure 7. Muscle flap covering the alloplastic mesh.
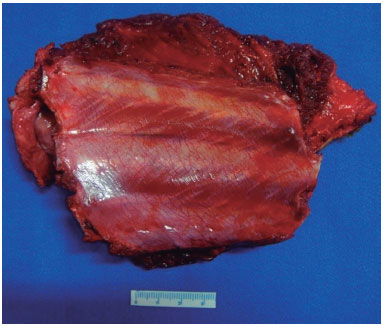
Figure 8. Resected ribs.
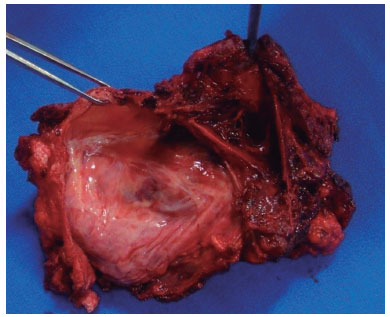
Figure 9. Implants capsule presenting AF.
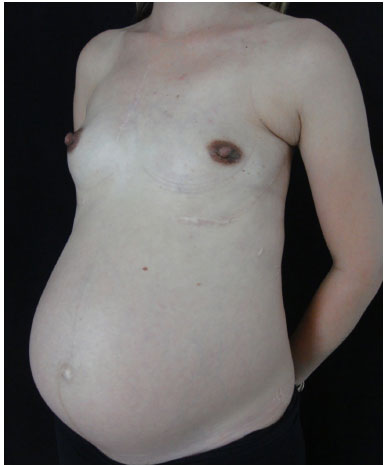
Figure 10. Pregnant patient, cured from aggressive fibromatosis.
Case 2
Female, 22 years and 5 months, presented in April 2008 with breast hypertrophy and asymmetry (greater breast on the right). She was submitted to reduction mammoplasty through perioareolar incision and subglandular textured implants (PIP silicone gel, 220 ml). On 08/10/2010, she underwent mastopexy with change of implants to the submuscular position. On 12/02/2011, she presented a right inframammary tumor.
On January 17, 2012, the diagnosis of aggressive fibromatosis was made through biopsy. The patient underwent thoracectomy, the surgical specimen measuring 13 x11 x 6 cm and neoplasia infiltrating intercostal spaces measuring 6 x 5x 3.8 cm. Extensive monobloc resection included skin, breast tissue, major pectoralis muscle, anterior segments of three ribs, intercostal muscles, endothoracic fascia and parietal pleura, sternum margin and implant capsule. The lateral resection margin was 5 cm. The implants were removed.
In the reconstruction of the thoracic wall, a folded Prolene® mesh was used, forming two layers. Conventional thoracic drainage was performed for four days. The lesion infiltrates the posterior face of the capsule. The surgical specimen presented disease-free lateral and deep margins, confirming the diagnosis of aggressive fibromatosis totally removed. The last examination was performed at 51 months of follow-up, on 04/14/2016, the patient presenting paradoxical breathing in the area of reconstruction of the ribs, without recurrence of AF (Figures 11 to 16).
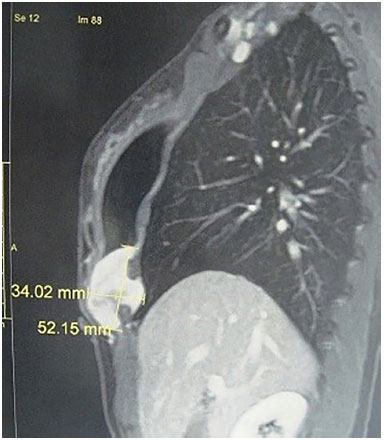
Figure 11. Computed tomography of lesions showing invation in two intercostal spaces.
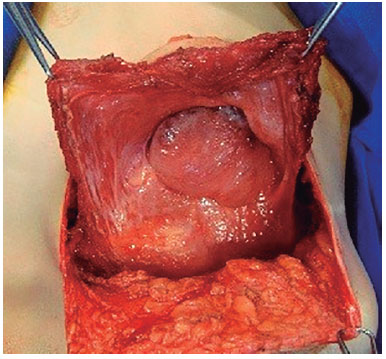
Figure 12. Open capsule showing aggressive fibromatosis.
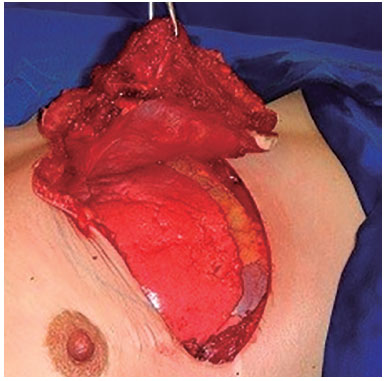
Figure 13. Enlarged thoracectomy.
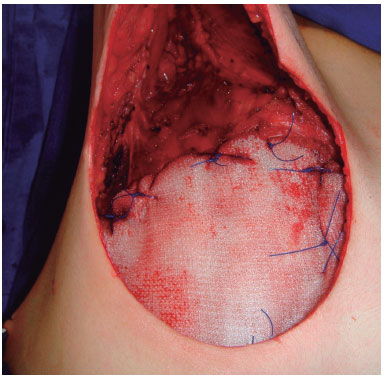
Figure 14. Alloplastic mesh.
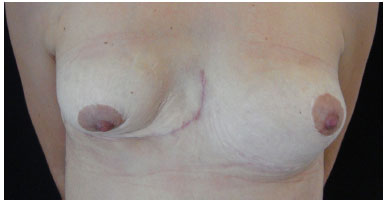
Figure 15. Postoperative aspect.
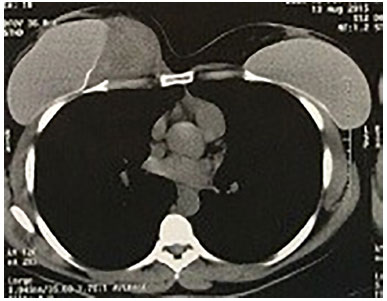
Figure 16. Computed tomography showing aggressive fibromatosis displacing right breast implant.
Case 3
Female, 38 years presented at the Hospital Júlia Kubitschek in Belo Horizonte, Minas Gerais, Brazil, on 08/18/2015, with a tumor in the right thoracic wall, measuring 9.3 x 4.8 x 4.5cm by CT-scan. She had undergone a biopsy with the diagnosis of aggressive fibromatosis. She reported receiving 265 ml textured silicone gel implants of unknown brand, submuscular, approximately 12 years before.
About five years ago, she the prostheses replaced. The submuscular route was maintained, with periareolar access, using 320 ml silicone gel implants, Shangai Winner®. In July 2014, during pregnancy, she noticed a small nodule in the inferomedial quadrant of the right breast, which had appeared about four years before and had grown rapidly.
On 9/11/2015, she the implant was removed with an enlarged monobloc resection that included skin, breast tissue, pectoralis major muscle, right lateral segment of the sternum, anterior segments of four ribs (4th, 5th, 6th, and 7th), intercostal muscles, endothoracic fascia and parietal pleura as well as the entire implant capsule. The lateral resection margin was 5 cm. The reconstruction was made using polypropylene mesh (Prolene®) and skin flap. The patient was reviewed on 01/25/2016, at four months of follow-up and was disease-free (Figures 17 to 19).
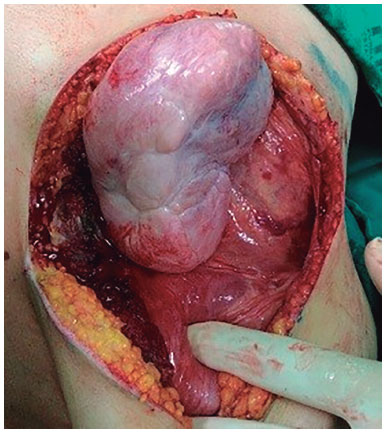
Figure 17. AF taking the form of a large tumor.
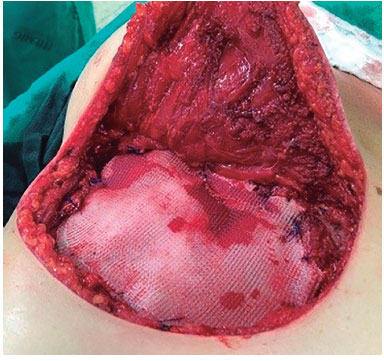
Figure 18. Reconstruction with alloplastic mesh.
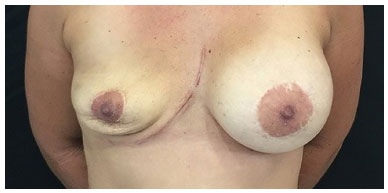
Figure 19. Postoperative aspect.
DISCUSSION
Although classified as a benign tumor, AF is locally aggressive and has high rates of recurrence, destroying the surrounding tissues. Recurrence may lead to very difficult to treat cases because of the greater amount of tissue needed to be resected to achieve cure. The neoplasm may invade joints, thoracic and abdominal cavities, large vessels and nerves, presenting high local morbidity.
As economic resections lead to high rates of recurrence5,8-10,17,26,27,33, the lateral surgical margins should be at least 5 cm and the deep margin should include an unaffected anatomical structure, such as muscle, bone or fascia6. It is impossible to know if in the cases described the tumor is a consequence of the surgical trauma caused by the augmentation mammoplasty or by the continuous trauma of the foreign body (breast implants) on the local tissues13. Because they are rare, these complications should be published in order to alert surgeons and patients about the nature of the disease.
The first authors to publish desmoid tumors related to the breast implant were Jewett Jr and Mead, in 197913. There is a Brazilian article about AF related to cosmetic plastic surgery published by Caldeira et al.12, in 2006, describing a case in the right hypochondrium diagnosed one year after an abdominal liposuction.
Chart 1 summarizes the characteristics of the 34 cases described in the 24 published articles. There is no evidence if implant biomaterial and the route of introduction interfere with the onset of AF, which may occur spontaneously or after traumatic injury to the fascia9,25.
CONCLUSION
A specific cause-effect relationship between breast implants and aggressive fibromatosis cannot yet be proven. It is accepted that any trauma of the fascia, surgical or otherwise, may be a potential inducer of neoplasia.
The biomaterials of silicone breast implants, as well as their route of placement, do not seem to interfere with the appearance of AF. The report of sparse cases is justified, given the rarity of the association of AF with silicone breast implants. The enlarged resection of the entire thickness of the affected chest wall with lateral margins of 5 cm and deep margin including the parietal pleura was adequate, promoting cure of the lesions in three patients submitted to a mean follow-up of 45 months.
ACKNOWLEDGMENTS
The authors thank Dr. Gil Patrus Mundim Pena for the preparation of the photomicrographs and Dr. Fernando Elói de Almeida Filho for the participation in the surgeries of the present paper.
COLLABORATIONS
AFSF Final approval of the manuscript; conception and design of the study; completion of surgeries and/or experiments; writing the manuscript or critical review of its contents.
JCRRA Analysis and/or interpretation of data; statistical analyses; final approval of the manuscript; conception and design of the study; completion of surgeries and/or experiments; writing the manuscript or critical review of its contents.
EHP Analysis and/or interpretation of data; statistical analyses; final approval of the manuscript; conception and design of the study; writing the manuscript or critical review of its contents.
RPLF Analysis and/or interpretation of data; statistical analyses.
ACMA Statistical analyses.
NAP Final approval of the manuscript.
FLS Completion of surgeries and/or experiments.
EFC Completion of surgeries and/or experiments.
JSAF Completion of surgeries and/or experiments.
REFERENCES
1. McFarlane J. Clinical reports of the surgical practices of Glasgow Royal Infirmary. Glasgow: D. Robertson; 1832. p. 63-66.
2. Dorland's Illustrated Medical Dictionary. 26th ed. Philadelphia: WB Saunders; 1981.
3. Müller J. Ueber den feinem Bau und die Formen der krankhaften Geschwulste. Berlin: Reimer; 1838. 60p.
4. Kallam AR, Ramakrishna BV, Roy GK, Karthik KR. Desmoid tumours: our experience of six cases and review of literature. J Clin Diagn Res. 2014;8(10):NE01-4. DOI: http://dx.doi.org/10.7860/JCDR/2014/9610.5032
5. From L, Assad D. Neoplasms, pseudoneoplasms, and hyperplasia of the dermis. In: Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, et al. eds. Fitzpatrick's Dermatology in General Medicine. New York: McGraw Hill; 1999. p. 1161-75.
6. Singhal H. Gardner syndrome. Medscape. Updated: May 24, 2016 [cited 2017 Aug 15]. Available from: http://emedicine.medscape.com/article/190486-overview
7. Weiss LM. Soft tissues. In: Weidner N, Cote RJ, Suster S, Weiss LM, eds. Modern Surgical Pathology. Philadelphia: Saunders Elsevier; 2009. p. 1717-83.
8. Rosen PP, Ernsberger D. Mammary fibromatosis. A benign spindle-cell tumor with significant risk for local recurrence. Cancer. 1989;63(7):1363-9. PMID: 2920364 DOI: http://dx.doi.org/10.1002/1097-0142(19890401)63:7<1363::AID-CNCR2820630722>3.0.CO;2-B
9. Dale PS, Wardlaw JC, Wootton DG, Resnick JI, Giuliano AE. Desmoid tumor occurring after reconstruction mammaplasty for breast carcinoma. Ann Plast Surg. 1995;35(5):515-8. PMID: 8579271 DOI: http://dx.doi.org/10.1097/00000637-199511000-00012
10. Khanfir K, Zouhair A. Review: aggressive fibromatosis and breast implant. Clin Adv Hematol Oncol. 2008;6(9):693-4.
11. Mátrai Z, Tóth L, Gulyás G, Szabó E, Szentirmay Z, Kásler M. A desmoid tumor associated with a ruptured silicone breast implant. Plast Reconstr Surg. 2011;127(1):1e-4e. PMID: 21200191
12. Caldeira LNC, Quinsan MP, Tostes ROG, Andrade Júnior JCCG, Silva KDA, Neves LJVA. Tumor desmoide pós-lipoaspiração abdominal estética: relato de caso. Rev Bras Cir Plást. 2006;21(1):53-5
13. Jewett ST Jr, Mead JH. Extra-abdominal desmoid arising from a capsule around a silicone breast implant. Plast Reconstr Surg. 1979;63(4):577-9. PMID: 424469 DOI: http://dx.doi.org/10.1097/00006534-197904000-00031
14. Schuh ME, Radford DM. Desmoid tumor of the breast following augmentation mammaplasty. Plast Reconstr Surg. 1994;93(3):603-5. DOI: http://dx.doi.org/10.1097/00006534-199493030-00028
15. Crestinu JM. Desmoid tumor of the breast. Plast Reconstr Surg. 1995;95(2):421. DOI: http://dx.doi.org/10.1097/00006534-199502000-00040
16. Schiller VL, Arndt RD, Brenner RJ. Aggressive fibromatosis of the chest associated with a silicone breast implant. Chest. 1995;108(5):1466-8. PMID: 7587464 DOI: http://dx.doi.org/10.1378/chest.108.5.1466
17. Aaron AD, O'Mara JW, Legendre KE, Evans SR, Attinger CE, Montgomery EA. Chest wall fibromatosis associated with silicone breast implants. Surg Oncol. 1996;5(2):93-9. DOI: http://dx.doi.org/10.1016/S0960-7404(96)80006-5
18. Vandeweyer E, Deraemaecker R. Desmoid tumor of the breast after reconstruction with implant. Plast Reconstr Surg. 2000;105(7):2627-8. PMID: 10845326 DOI: http://dx.doi.org/10.1097/00006534-200006000-00065
19. Abraham SC, Reynolds C, Lee JH, Montgomery EA, Baisden BL, Krasinskas AM, et al. Fibromatosis of the breast and mutations involving the APC/beta-catenin pathway. Hum Pathol. 2002;33(1):39-46. PMID: 11823972 DOI: http://dx.doi.org/10.1053/hupa.2002.30196
20. Khanfir K, Guinebretiere JM, Vanel D, Barreau-Pouhaer L, Bonvalot S, Le Cesne A. Unusual problems in breast cancer and a rare lung cancer case. Case 2. Aggressive fibromatosis of the chest wall arising near a breast prosthesis. J Clin Oncol. 2003;21(11):2216-8. DOI: http://dx.doi.org/10.1200/JCO.2003.04.189
21. Jandali AR, Wedler V, Meuli-Simmen C, Künzi W, Meyer VE. Breast implant and desmoid tumor: is there an etiological relation? Handchir Mikrochir Plast Chir. 2004;36(6):343-7. PMID: 15633076
22. Gandolfo L, Guglielmino S, Lorenzetti P, Fiducia G, Scenna G, Bosco V. Chest wall fibromatosis after mammary prosthesis implantation. A case report and review of the literature. Chir Ital. 2006;58(5):655-60. PMID: 17069197
23. Neuman HB, Brogi E, Ebrahim A, Brennan MF, Van Zee KJ. Desmoid tumors (fibromatoses) of the breast: a 25-year experience. Ann Surg Oncol. 2008;15(1):274-80. DOI: http://dx.doi.org/10.1245/s10434-007-9580-8
24. Jamshed S, Farhan MI, Marshall MB, Nahabedian MY, Liu MC. Fibromatosis of the breast after mammary prosthesis implantation. Clin Adv Hematol Oncol. 2008;6(9):687-94.
25. Balzer BL, Weiss SW. Do biomaterials cause implant-associated mesenchymal tumors of the breast? Analysis of 8 new cases and review of the literature. Hum Pathol. 2009;40(11):1564-70. PMID: 19540553 DOI: http://dx.doi.org/10.1016/j.humpath.2009.03.020
26. Mazzocchi M, Onesti MG, Di Ronza S, Scuderi N. Breast desmoid tumor after augmentation mammoplasty: two case reports. Acta Chir Plast. 2009;51(3-4):73-8. PMID: 20514891
27. Chummun S, McLean NR, Abraham S, Youseff M. Desmoid tumour of the breast. J Plast Reconstr Aesthet Surg. 2010;63(2):339-45. DOI: http://dx.doi.org/10.1016/j.bjps.2008.09.024
28. Henderson PW, Singh SP, Spector JA. Chest wall spindle cell fibromatosis after breast augmentation. Plast Reconstr Surg. 2010;126(2):94e-5e. DOI: http://dx.doi.org/10.1097/PRS.0b013e3181de23ce
29. Gergelé F, Guy F, Collin F, Krausé D. A desmoid tumour associated with a breast prosthesis. Diagn Interv Imaging. 2012;93(3):e200-3.
30. Hammoudeh ZS, Darian VB. Desmoid tumor (fibromatosis) of the breast after augmentation with saline implants. Plast Reconstr Surg. 2012;129(4):753e-4e. PMID: 22456408 DOI: http://dx.doi.org/10.1097/PRS.0b013e318245e918
31. Jeong WS, Oh TS, Sim HB, Eom JS. Desmoid tumor following augmentation mammoplasty with silicone implants. Arch Plast Surg. 2013;40(4):470-2. DOI: http://dx.doi.org/10.5999/aps.2013.40.4.470
32. Shim HS, Kim SJ, Kim OH, Jung HK, Kim SJ, Kim W, et al. Fibromatosis associated with silicone breast implant: ultrasonography and MR imaging findings. Breast J. 2014;20(6):645-9. DOI: http://dx.doi.org/10.1111/tbj.12340
33. Seo YN, Park YM, Yoon HK, Lee SJ, Choo HJ, Ryu JH. Breast fibromatosis associated with breast implants. Jpn J Radiol. 2015;33(9):591-7. DOI: http://dx.doi.org/10.1007/s11604-015-0461-y
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
2. Hospital Felício Rocho, Belo Horizonte, MG, Brazil
3. Instituto de Cirurgia Plástica Avançada, Belo Horizonte, MG, Brazil
4. Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brazil
5. Universidade de Itaúna, MG, Brazil
6. Hospital e Maternidade Therezinha de Jesus, Juiz de Fora, MG, Brazil
7. Hospital João XXIII, Belo Horizonte, MG, Brazil
8. Hospital Júlia Kubitscheck, Belo Horizonte, MG, Brazil
9. Faculdade de Ciências Médicas de Minas Gerais, Belo Horizonte, MG, Brazil
Institution: Hospital Felício Rocho, Belo Horizonte, MG, Brazil.
Corresponding author:
Erick Horta Portugal
Rua Santa Maria de Itabira, 217 - Bairro Sion
Belo Horizonte, MG, Brazil - Zip Code 30310-600
E-mail: erickhphp@yahoo.com.br
Article received: April 10, 2017.
Article accepted: August 7, 2017.
Conflicts of interest: none.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter