

Original Article - Year 2013 - Volume 28 -
Evaluation of neurosensory disturbance in patients with craniofacial syndrome subjected to horizontal chin advancement
Avaliação de déficit neurossensorial em pacientes com síndromes craniofaciais submetidos a avanço horizontal do mento
ABSTRACT
BACKGROUND: Although neurosensory disturbances after genioplasty have been evaluated in different studies, standardization for testing and grading of neurosensory injuries is lacking. For this reason, the incidence of neurosensory disturbance varies from 0% to 100%, depending on the definition of nerve damage, the sensitivity of the diagnostic test method, and the follow-up period. Therefore, the aim of this study was to perform an objective evaluation of the permanent neurosensory disturbances in patients who underwent horizontal chin advancement.
METHODS: A retrospective study of all patients who underwent horizontal chin advancement at the Hospital SOBRAPAR between 2009 and 2010 was conducted. The objective neurosensory assessment of the lower lip and chin was performed using 2 neurological tests, namely the Semmes-Weinstein pressure and thermal sensitivity tests. Permanent neurosensory disturbance was defined as abnormal clinical test results obtained at least 12 months after surgery.
RESULTS: Thirteen patients (8 with craniofacial syndrome) were evaluated. The prevalence of the patients who showed normal results for sensitivity to pressure/touch and thermal sensitivity (warm and cold; P < 0.05) was significantly high. The analysis of the data of the patients with craniofacial syndrome revealed that most of the patients had normal pressure sensitivity test results (P < 0.003). Results from the thermal sensitivity tests showed no significant difference between these patients (P = 0.317). No significant differences were observed between the anatomical regions with abnormal sensitivity test results (P > 0.05).
CONCLUSIONS: Tactile sensitivities of the lower lip and chin to pressure and temperature were preserved in most of the patients 12 months after horizontal chin advancement.
Keywords: Genioplasty. Chin/surgery. Orthognathic surgical procedures. Mandibular nerve.
RESUMO
INTRODUÇÃO: Embora os distúrbios neurossensoriais após genioplastias tenham sido avaliados em diferentes estudos, não existe uma padronização de como testar e classificar tais alterações. Por essa razão, a incidência de distúrbios neurossensoriais varia de 0 a 100%, dependendo da definição da lesão dos nervos, da sensibilidade do método diagnóstico e do período de seguimento. Portanto, o propósito deste estudo foi avaliar objetivamente o déficit neurossensorial permanente em pacientes submetidos a avanço horizontal do mento.
MÉTODO: Foi realizado estudo retrospectivo de todos os pacientes submetidos a avanço horizontal do mento no Hospital SOBRAPAR, no período de 2009 a 2010. A avaliação neurossensorial objetiva do lábio inferior e do mento foi realizada com dois testes neurológicos (teste dos limiares de pressão de Semmes-Weinstein e teste de sensibilidade térmica). O déficit neurossensorial permanente foi definido como testes clínicos anormais com no mínimo 12 meses de pós-operatório.
RESULTADOS: Foram avaliados 13 pacientes, sendo 8 deles portadores de síndromes craniofaciais. Houve predomínio de pacientes com os testes de sensibilidade tátil à pressão e térmica (quente e frio) normal (P < 0,05). A análise dos pacientes sindrômicos revelou que a maioria teve o teste de sensibilidade tátil à pressão normal (P < 0,003), não existindo diferenças no teste de sensibilidade térmica (P = 0,317). Não foram identificadas diferenças entre as regiões anatômicas com testes de sensibilidade anormais (P > 0,05).
CONCLUSÕES: A maioria dos pacientes apresenta sensibilidade tátil (pressão e temperatura) do lábio inferior e mento preservada 12 meses após terem sido submetidos a avanço horizontal do mento.
Palavras-chave: Mentoplastia. Queixo/cirurgia. Procedimentos cirúrgicos ortognáticos. Nervo mandibular.
The chin occupies a prominent position in the lower third of the face and represents an element that contributes to overall facial appearance and harmony1. Therefore, disturbances in chin shape, anatomic position, and size can cause aesthetic disharmony among the bone components of the craniofacial skeleton2. Different surgical approaches have been used to alter chin size and spatial position1-3.
Although alloplastic chin implants are used most commonly for correction of minimal sagittal chin deficiencies (usually less than 5 mm), the horizontal osteotomy of the symphysis (commonly designated as osseous genioplasty or simply genioplasty4) is a far more versatile procedure. In this case, the chin can be repositioned in multiple planes, allowing for long-lasting results3,4.
In fact, genioplasty is the only surgical solution for asymmetric and vertical disturbances, as well as for significant sagittal deficiencies. Moreover, this technique represents the only alternative to correct chin malformations in patients with craniofacial syndromes (e.g., Treacher-Collins-Franceschetti syndrome, Nager syndrome, and craniofacial microsomia). These patients often require extensive chin advancement associated with chin repositioning in multiple planes to correct their complex asymmetries3,4. Furthermore, the horizontal osteotomy of the chin may result in effective traction of the genioglossus and geniohyoid muscles and increase in the chin-neck angle, thus improving breathing in these syndromic patients5.
Regardless of the technique used, complications such as hematoma, seroma, infection, osteonecrosis, ptosis of the lip/chin, heterotopic ossification, dental complications, irregular mandibular contours, and neurosensory disturbances of the lower lip and/or chin can arise from chin surgery1,2. Of particular interest are these neurosensory disturbances, as they can compromise oral functions such as salivation and retention of food particles, and also lead to local trauma accompanied by negative psychosocial impact on these patients6.
Even though there are studies that examined neurosensory disturbances after genioplasty7-9, several limitations have been described10. Comparison of results is often complicated because these studies use not only different methods of analysis but also a lack of standardization of the tests used to detect neurosensory abnormalities10,11. Thus, the incidence of neurosensory disturbances is extremely variable (0% to 100%), depending on the definition of nerve damage, the sensitivity of the diagnostic test method, and the follow-up period10,11. Studies that use standard methods of analysis and follow-up periods are necessary10,11. So far, the national literature comprises no descriptive studies addressing neurosensory loss in syndromic patients who underwent horizontal advancement of the chin. Therefore, the purpose of this study was to perform an objective evaluation of the permanent neurosensory disturbances in patients who underwent horizontal chin advancement.
METHODS
This study was approved by the Committee of Ethics and Human Research of the Hospital SOBRAPAR and complied with the Declaration of Helsinki of 1975, revised in 1983.
A retrospective study of all patients who underwent horizontal advancement of the chin at the Hospital SOBRAPAR between February 2009 and September 2010 was conducted.
The medical records of all the patients subjected to genioplasty were analyzed. All the patients who underwent horizontal chin advancement performed by the same group of surgeons (same education and philosophy) and with a minimal postoperative follow-up of 12 months were included in this study. Patients with a previous history of factors that could interfere with the sensitivity of the lower lip and chin, such as diabetes mellitus, smoking, facial trauma, neurosensory disturbances prior to genioplasty, and/or damage of the inferior alveolar nerves or of the mental nerves detected during the surgical intervention were excluded from the study.
A total of 13 patients, between 18 and 36 years of age, met the inclusion criteria. Of the patients, 8 (61.54%) were male and 5 (38.46%) were female. Eight (61.54%) patients had craniofacial syndrome, 6 (46.15%) had Treacher-Collins-Franceschetti syndrome, and 2 (15.38%) had Crouzon syndrome. The mean hospitalization time was 1.46 ± 1.20 days, and the mean postoperative follow-up period was 22.69 ± 5.82 months, ranging from 13 to 29 months.
Surgical Strategy
All surgical plans (repositioning of the chin in vertical, horizontal, or side planes and advancement size) were based on previously established recommendations (clinical and photocephalometric analyses)2,3,12. Additional details were reported by our group5.
Surgical Anatomy
A few anatomic sites such as the course of the inferior alveolar nerve from the point where it enters the mandibular foramen (medial section of the ramus) up to where it exits the mental foramen (between the second and third premolars) are extremely important to minimize neurosensory disturbances. The mandibular foramen is situated 0 mm to 8 mm vertically and 20 mm below the lingula and incisura, respectively. In terms of anteroposterior position, the mandibular foramen is situated approximately 20 mm off the anterior margin of the ramus, and its depth corresponds to approximately two-thirds of the total width of the ramus. The channel makes its way inside the mandible until it exits into the mental foramen, situated 10 to 15 mm off the inferior border. An important aspect of the genioplasty is the fact that before emerging in the mental foramen, there is an approximately 5 mm caudal extension of the channel12.
Surgical Technique
All surgeries were performed according to the same standards1-5,12. A solution containing a local anesthetic was first injected in the oral mucosa area, toward the second premolar tooth. The surgery started with an incision in the oral mucosa, perpendicular to the mentalis muscle and to the mental periosteum. Detachment of the anterior bone area was performed until the alveolar nerve was visualized, which is normally found between the first and second premolar and inferior to the depressor anguli oris muscle. The nerve was subsequently protected before initiating the osteotomies. The medial region was then delineated with methylene blue. An oscillating saw was used to perform the medial osteotomy of the chin. A reciprocating saw was used in the lateral areas to complete the horizontal osteotomy of the chin. The osteotomy was performed at a distance of 5 mm from the canine root and 6 mm below the mentonian nerve. Once the horizontal osteotomy of the chin was finalized, an elevator was used in the caudal direction to complete the fracture. The fractured distal chin segment was kept connected to the genioglossus and geniohyoid muscles, which is responsible for the blood supply to the fractured area. The most forward-projecting cephalometric area of the chin (pogonion) did not surpass the imaginary line perpendicular to the lower lip, thus ensuring a natural and harmonious result. Chin fixation was performed with plates and 2-mm screws (Figure 1).
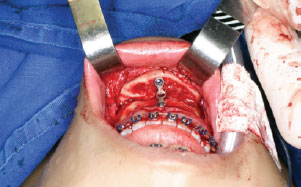
Figure 1 - Intraoperative aspect showing horizontal chin advancement using sliding genioplasty and fixation with a 2.0-mm plate and three titanium screws.
Objective Neurosensory Evaluation
All the participants were tested with eyes closed in a comfortable position (dorsal decubitus) at room temperature in a room without any acoustic or visual perturbation. The objective neurosensory evaluation of the lower lip and chin was performed with 2 neurological tests (objective and qualitative Semmes-Weinstein threshold test and objective thermal sensitivity test). These tests measure the decrease in sensitivity perceived by the patients. Tests were conducted at the same order and by the same investigator in all the patients. Before each neurosensory test, the application of each instrument was tested in a skin area with normal sensitivity. A stimulus was also applied to a skin area with normal sensitivity at every 2 or 3 stimuli non-identified by the patient, thus allowing the patient to have a reference for a normal cutaneous sensation.
For test standardization, the anatomic area innervated by the inferior alveolar nerve (specifically the mental nerve) was divided into 4 areas as follows:
1. Medial section of the chin and lower lip, situated 1 cm bilaterally from the cutaneous and mucosal side, starting from the mandibular symphysis.
2. Paramedial section, situated 2 cm bilaterally from the cutaneous and mucosal side, starting from the mandibular symphysis.
3. Mental foramen section, situated 3 cm bilaterally from the cutaneous and mucosal side starting from the mandibular symphysis, a few millimeters below the roots of the fourth and fifth teeth.
4. Border region of the lower lip vermillion (Figure 2)13.
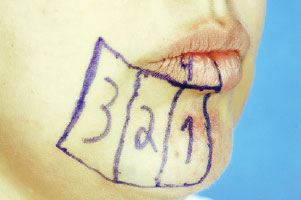
Figure 2 - Anatomical regions tested were: (1) the medial, (2) paramedial, and (3) mental foramen sections of the chin and (4) the lower lip vermillion section.
Both sides of the face were tested separately. Permanent neurosensory disturbance was defined as abnormal clinical test results obtained at least 12 months after surgery10.
Semmes-Weinstein Threshold Test
The objective and quantitative evaluations of tactile sensitivity were performed using the Semmes-Weinstein pressure aesthesiometer (SORRI®, Bauru, SP, Brazil), according to procedures previously described in similar studies14,15. All the tests were performed with the same group of nylon monofilaments of increasing diameter and followed the same standardization.
The test started with the thinner filament, followed by filaments with progressively increasing thickness. The patient was asked to answer yes when the touch of the monofilament was felt. Then, a filament immediately thicker was tested, but only in the areas without positive response to the previous filament. Each stimulus was maintained for approximately 1.5 seconds, and articulate movements were avoided during use of the monofilaments. For each tested monofilament, the stimuli were applied 4 times in each area of interest. The stimulus response was considered positive when there were at least 3 (75%) correct answers (3 of 4 correct stimuli). The Semmes-Weinstein monofilaments 2.83 and 3.22 were selected as the upper limit of normality for the detection thresholds for the lower lip and chin, respectively14,16.
Test of Thermal Sensitivity
Thermal sensitivity was evaluated using the hot-cold discrimination test13-15. Two small glass test tubes containing cold (15ºC to 20ºC) or hot (40ºC to 45ºC) water were used. The test area was randomly touched 10 times with the test tube and the patient had to report whether the stimulus was hot or cold. Test results were considered normal when 80% of the answers were correct.
Statistical Analysis
All data were subjected to statistical analysis using the test for equality of 2 proportions with the software Statistical Package for Social Sciences, version 16, for Windows (SPSS, Chicago, IL, USA). A p value < 0.05 was considered as statistically significant with a confidence interval of 95%.
RESULTS
A total of 13 patients were subjected to genioplasty. The horizontal advancement of the chin ranged from 8 to 12 mm, with a mean of 10.23 ± 1.09 mm. Full or partial incision of the inferior alveolar nerve and/or of the mental nerve was not evident in any of the osteotomies performed. No surgical complications were detected during the postoperative period, and there was no receding in the chin position, as shown by imaging and cephalometric measures (Figures 3 and 4).
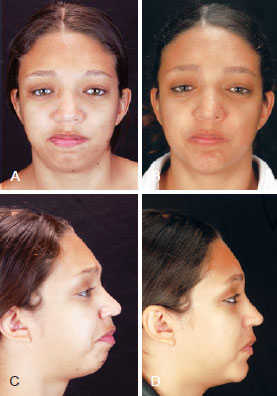
Figure 3 - In A and C, frontal view and right profile, respectively, of a patient with Crouzon syndrome showing hypomentonism in the preoperative period after undergoing frontofacial monobloc advancement and immediately before the horizontal chin advancement. In B and D, frontal view and right profile, respectively, after chin surgery for horizontal advancement.
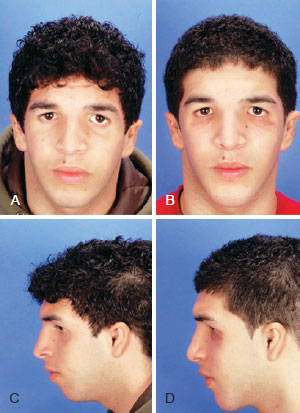
Figure 4 - Patient with Treacher-Collins-Franceschetti syndrome. In A and C, frontal and left profile views, respectively, of the preoperative aspect showing hypomentonism; In B and D, frontal and left profile views, respectively, of postoperative aspect after facial reconstruction and horizontal chin advancement.
Semmes-Weinstein Threshold Test
Most of the patients showed a normal response when subjected to the tactile point pressure sensitivity test using the Semmes-Weinstein monofilaments (p < 0.05). Most of the syndromic patients also showed a normal response to the test (p < 0.003). No significant differences were observed between the anatomical regions (1 and 2) with abnormal pressure sensitivity test results (p > 0.05, Table 1). Tactile point pressure neurosensitivity test results revealed that 1 patient (7.69%) with Treacher-Collins-Franceschetti syndrome (12-mm advancement) and 1 patient (7.69%) with craniofacial syndrome (10-mm advancement) showed disturbances in region 1 and regions 1 and 2, respectively.
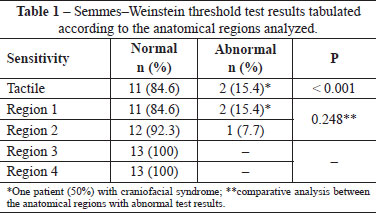
Thermal Sensitivity Results
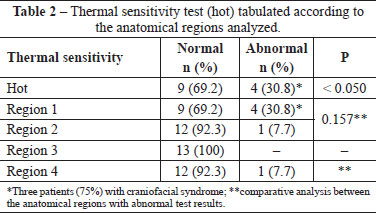
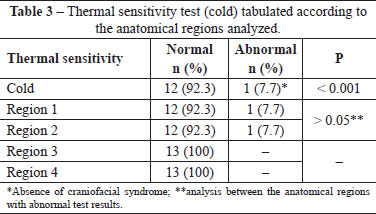
The prevalence of the patients who showed a normal response to the hot/cold thermal sensitivity test was significantly high (p < 0.05). No significant differences were detected when the syndromic patients were considered (p = 0.317). No significant differences were observed between the anatomical regions (1, 2, and 4; p > 0.05) with abnormal thermal sensitivity test results (Tables 2 and 3). Results for thermal sensitivity to heat revealed that 3 patients (23.08%) with craniofacial syndrome (1 with Crouzon syndrome and 2 with Treacher-Collins-Franceschetti syndrome subjected to 10- and 12-mm advancements, respectively) and 1 (7.69%) without craniofacial syndrome (10-mm advancement) demonstrated neurosensory disturbances in regions 1 and regions 1, 2, and 4, respectively. The same patient without craniofacial syndrome (10-mm advancement) demonstrated neurosensory disturbances in regions 1 and 2 when subjected to a cold sensitivity test.
DISCUSSION
Genioplasty, whether in combination or not with other orthognathic procedures, has been commonly used to correct functional and aesthetic chin malformations4,17. Although excellent functional and aesthetic results can be obtained4,17, this surgical intervention may cause neurosensory disturbances during the postoperative period1,2. The alterations in lower lip and chin sensitivity have been attributed to lesions in 1 or both mental nerves. Lesions may occur during mucosal incision, musculoperiosteal dissection, osteotomy execution, bone segment repositioning, and/or stabilization of the fractured bone fragments13,15. On the basis of these considerations, caution should be taken while manipulating mental nerves during tissue dissection. This has a significant impact on the conception of osteotomies and on the surgeon's ability to safely isolate and protect the mental nerves18. Most of the neurosensory abnormalities detected during the postoperative period are reversible13-15,18,19. However, neurosensory disturbances that persist even a year after a mandibular osteotomy should be considered more severe and permanent10. For this reason, only patients with at least 12 months of postoperative follow-up were analyzed in this study.
The results of the present study are substantially similar to those of other studies13,15. Most of the patients with and those without craniofacial syndromes had their neurosensory sensitivity preserved 1 year after the surgery. Only 3 patients (23.08%) with craniofacial syndrome and 1 (7.69%) without this syndrome had permanent neuro sensory disturbances. One patient with Treacher-Collins-Franceschetti syndrome subjected to a 12-mm horizontal chin advancement and 1 patient without syndrome subjected to a 10-mm horizontal chin advancement demonstrated tactile and thermal (hot and cold) neurosensory disturbances, respectively. The literature reports that neurosensory disturbance rates after genioplasty ranges from 0% to 28.5%, which depends in part on the method used to detect the neurosensory abnormalty3-15,18,19. As there is no consensus in the method used to evaluate these neurosensory disturbances12, a wide variety of tools and methods (e.g., questionnaires, tactile point pressure sensitivity test, discrimination between 2 points, and thermal discrimination) have been used in different studies12-15,18,19. Generally, the tool and method used to document the neurosensory disturbance should be specific and able to detect a wide variety of disturbances (from complete anesthesia to near-normal sensitivity). In addition, these tools/methods need to be sensitive and reproducible across time12. In this study, the Semmes-Weinstein threshold test was the tool selected to analyze neurosensory disturbances, on the basis of a recommendation from a recent systematic review10. According to other authors, this is the most sensitive and clinically useful neurosensory test because it provides quantitative data and reliable objectives12,14. However, it is important to note that more advanced and complex methods to characterize neurosensory disturbances are available. Methods that include electric stimulation, electromyography, somatosensoryevoked potential, and thermal quantitative sensory testing may characterize neurosensory disturbances more adequately11. Moreover, this study also used thermal sensitivity testing as a secondary tool, as used by other authors15.
In the present study, all the patients who showed sensitive disturbances were subjected to genioplasty with advancements greater than 10 mm (defined as extensive20). It is interesting that anatomical region 1 was involved in all the cases with permanent neurosensory disturbances. The authors hypothesized that this was because region 1 was the most distal area of mental innervation and, consequently, more susceptible to damage during the large tissue distension inherent to the extensive chin advancement. As the regions more affected and those less affected were not statistically correlated, future studies are needed to confirm this hypothesis.
Another aspect relevant to the execution of advancement genioplasty is that the ability, experience, and knowledge in a specific surgical anatomy may influence the risk for postoperative neurological complications17. Thus, it is important to teach this technique to all plastic surgery residents in training4.
Two limitations of this study can be pointed out as follows: lack of neurosensitivity evaluation of the chin and lower lip prior to genioplasty and lack of use of more modern and sophisticated methods to analyze neurosensitivity. Future studies should evaluate these aspects.
CONCLUSIONS
This retrospective observational study demonstrated that most of the patients with and those without craniofacial syndrome had their tactile (pressure and temperature) neurosensory sensitivity preserved for 12 months after undergoing horizontal chin advancement.
REFERENCES
1. Nahai FR. Surgery of the chin. Facial Plast Surg. 2012;28(1):34-9.
2. Guyuron B. MOC-PS(SM) CME article: genioplasty. Plast Reconstr Surg. 2008;121(4 Suppl):1-7.
3. Ward JL, Garri JI, Wolfe SA. The osseous genioplasty. Clin Plast Surg. 2007;34(3):485-500.
4. Wolfe SA, Rivas-Torres MT, Marshall D. The genioplasty and beyond: an end-game strategy for the multiply operated chin. Plast Reconstr Surg. 2006;117(5):1435-46.
5. Farias DB, Silva MPA, Menon D, Almeida F, Raposo-do-Amaral CA, Buzzo CL, et al. Estratégias cirúrgicas para o avanço horizontal do mento em pacientes sindrômicos. Rev Bras Cir Craniomaxilofac. 2010;13(3):127-31.
6. Guyot L, Layoun W, Richard O, Cheynet F, Gola R. Alteration of chin sensibility due to damage of the cutaneous branch of the mylohyoid nerve during genioplasty. J Oral Maxillofac Surg. 2002;60(11):1371-3.
7. Lindquist CC, Obeid G. Complications of genioplasty done alone or in combination with sagittal split-ramus osteotomy. Oral Surg Oral Med Oral Pathol. 1988;66(1):13-6.
8. Nishioka GJ, Mason M, Van Sickels JE. Neurosensory disturbance associated with the anterior mandibular horizontal osteotomy. J Oral Maxillofac Surg. 1988;46(2):107-10.
9. Posnick JC, Al-Qattan MM, Stepner NM. Alteration in facial sensibility in adolescents following sagittal split and chin osteotomies of the mandible. Plast Reconstr Surg. 1996;97(5):920-7.
10. Antonarakis GS, Christou P. Quantitative evaluation of neurosensory disturbance after bilateral sagittal split osteotomy using Semmes-Weinstein monofilaments: a systematic review. J Oral Maxillofac Surg. 2012;70(12):2752-60.
11. Poort LJ, van Neck JW, van der Wal KG. Sensory testing of inferior alveolar nerve injuries: a review of methods used in prospective studies. J Oral Maxillofac Surg. 2009;67(2):292-300.
12. Patel PK, Novia MV. The surgical tools: the LeFort I, bilateral sagittal split osteotomy of the mandible, and the osseous genioplasty. Clin Plast Surg. 2007;34(3):447-75.
13. Gianni AB, D'Orto O, Biglioli F, Bozzetti A, Brusati R. Neurosensory alterations of the inferior alveolar and mental nerve after genioplasty alone or associated with sagittal osteotomy of the mandibular ramus. J Craniomaxillofac Surg. 2002;30(5):295-303.
14. Teerijoki-Oksa T, Jääskeläinen S, Forssell K, Virtanen A, Forssell H. An evaluation of clinical and electrophysiologic tests in nerve injury diagnosis after mandibular sagittal split osteotomy. Int J Oral Maxillofac Surg. 2003;32(1):15-23.
15. Nishioka GJ, Zysset MK, Van Sickels JE. Neurosensory disturbance with rigid fixation of the bilateral sagittal split osteotomy. J Oral Maxillofac Surg. 1987;45(1):20-6.
16. Baas EM, de Lange J, Horsthuis RB. Evaluation of alveolar nerve function after surgical lengthening of the mandible by a bilateral sagittal split osteotomy or distraction osteogenesis. Int J Oral Maxillofac Surg. 2010;39(6):529-33.
17. Hoenig JF. Sliding osteotomy genioplasty for facial aesthetic balance: 10 years of experience. Aesthetic Plast Surg. 2007;31(4):384-91.
18. Troulis MJ, Kearns GJ, Perrott DH, Kaban LB. Extended genioplasty: long-term cephalometric, morphometric and sensory results. Int J Oral Maxillofac Surg. 2000;29(3):167-75.
19. Westermark A, Bystedt H, von Konow L. Inferior alveolar nerve function after mandibular osteotomies. Br J Oral Maxillofac Surg. 1998;36(6):425-8.
20. Polido WD, Bell WH. Long-term osseous and soft tissue changes after large chin advancements. J Craniomaxillofac Surg. 1993;21(2):54-9.
1. Aspiring member, in training, at the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery - SBCP), plastic surgery resident at the "Prof. Dr. Cassio M. Raposo do Amaral" do Instituto de Cirurgia Plástica Crânio Facial do Hospital SOBRAPAR (Prof. Dr. Cassio M. Raposo do Amaral Plastic Surgery Service, Craniofacial Surgery Institute of the Hospital SOBRAPAR), Campinas, SP, Brazil
2. Physician and research fellow at the Instituto de Cirurgia Plástica Crânio Facial do Hospital SOBRAPAR (Craniofacial Surgery Institute of the Hospital SOBRAPAR), Campinas, SP, Brazil
3. Plastic surgeon, associate member of the SBCP and previous resident at the Prof. Dr. Cassio M. Raposo do Amaral Plastic Surgery Service, Craniofacial Surgery Institute of the Hospital SOBRAPAR, Campinas, SP, Brazil
4. Plastic surgeon, registered member of the SBCP and the Brazilian Society for Craniomaxillofacial Surgery (ABCCMF), M.Sc. in Surgery from the State University of Campinas (UNICAMP), manager of the Prof. Dr. Cassio M. Raposo do Amaral Plastic Surgery Service, Craniofacial Surgery Institute of the Hospital SOBRAPAR, Campinas, SP, Brazil
5. Plastic surgeon, registered member of the SBCP, preceptor of residents of the Prof. Dr. Cassio M. Raposo do Amaral Plastic Surgery Service, Craniofacial Surgery Institute of the Hospital SOBRAPAR, Campinas, SP, Brazil
6. Registered member of the SBCP and ABCCMF, Ph.D. from the Clinical Surgery Program of São Paulo University (USP), vice president at the Craniofacial Surgery Institute of the Hospital SOBRAPAR, Campinas, SP, Brazil
Correspondence to:
Cassio Eduardo Raposo-do-Amaral
Instituto de Cirurgia Plástica Crânio Facial do Hospital SOBRAPAR
Av. Adolpho Lutz, 100 - Cidade Universitária
Campinas, SP, Brazil - CEP 13083-880 - Caixa postal 6028
E-mail: cassioraposo@hotmail.com
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: April 27, 2013
Article accepted: June 2, 2013
This study was performed at the Instituto de Cirurgia Plástica Crânio Facial do Hospital SOBRAPAR (Craniofacial Surgery Institute of the Hospital SOBRAPAR), Campinas, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter