

Original Article - Year 2012 - Volume 27 -
Modification of the mastopexy technique described by Longacre
Mastopexia a Longacre modificada
ABSTRACT
BACKGROUND: The functional preservation of the breasts should always be a concern for the plastic surgeon. The technique presented in this paper aims to reduce the impact of mammoplasty on breastfeeding by considering the superior vascular pedicle technique with parsimonious gland resection to be the best choice for cases with little breast hypertrophy.
METHODS: We report on 46 patients with mild to moderate breast hypertrophy who underwent mastopexy, with or without breast reduction, using the superior vascular pedicle technique as per the modified technique described by Longacre. Small gland resections were made so as to preserve the functional and sensory integrity of breasts.
RESULTS: In the 92 breasts on which we operated, we observed a proper filling of the upper pole through use of a flap, and breast shaping that satisfied the patients' desires.
CONCLUSIONS: The technique presented, in which the lower pole of the breast is used to fill the upper pole, is another technical possibility for mastopexies in patients with a high-positioned nipple-areolar complex.
Keywords: Mammaplasty. Breast/surgery. Breast feeding.
RESUMO
INTRODUÇÃO: A preservação funcional das mamas deve ser uma preocupação constante para o cirurgião plástico. A técnica apresentada neste trabalho busca reduzir o impacto da mamoplastia no aleitamento materno, considerando a técnica de pedículo vascular superior, com parcimoniosas ressecções glandulares, a de escolha para os casos de pequena hipertrofia mamária.
MÉTODO: Foram estudadas 46 pacientes portadoras de hipertrofia mamária leve a moderada, submetidas a mastopexia, com ou sem redução mamária, utilizando técnica de pedículo vascular areolar superior, segundo a técnica descrita por Longacre modificada. As pequenas ressecções glandulares foram realizadas de forma a preservar a integridade funcional e sensitiva das mamas.
RESULTADOS: Nas 92 mamas operadas, observou-se adequado preenchimento do polo superior pelo retalho e conformação mamária, que satisfizeram os anseios das pacientes.
CONCLUSÕES: A técnica apresentada constitui mais uma possibilidade técnica para mastopexias em pacientes com o complexo areolopapilar em posicionamento alto, utilizando-se o polo inferior da mama para preencher o polo superior.
Palavras-chave: Mamoplastia. Mama/cirurgia. Aleitamento materno.
The functional preservation of the breasts must be a constant concern for the plastic surgeon. The psychological stress experienced by adolescents and young women who are unhappy with their breasts is so intense that they visit plastic surgeons to diminish these frustrations. However, they are so involved in the aesthetic breast solution that they involuntarily ignore the future importance of breastfeeding when they become mothers. Interest in clarifying the difficulties of breastfeeding after mammoplasty is increasing, as many of these women feel frustrated when they cannot breastfeed; they often state that they did not receive enough information during the preoperative period.
For these patients, the postoperative reorganization of the breast structure causes problems with breastfeeding, as a result of the interruption of the continuity between the gland tissue itself and the lactiferous ducts.
The advent of breast reductions with several vascular pedicles for the areolar tissue supply, with techniques having been described by Strömbeck1 (horizontally bipediculated), Skoog2 (lateral pedicle), McKissock3 (vertical bipediculated), and Pitanguy4 (superior areolar pedicle), allowed relative maintenance of the anatomic relation between the gland and the nipple-areolar complex (NAC). The more invasive techniques (or less functional) that report areolar amputation and subsequent free graft in a new position, as described by Thorek5, are not used as frequently anymore. Nonetheless, the result of these breast resections must preserve the anatomic continuity of the gland system in order to retain the possibility of breastfeeding. Thus, regardless of the type of blood supply, the thickness of this pedicle and the higher breast gland integrity and its sensory innervation are essential to obtaining good functional results in mastopexies.
Some studies do not mention differences in breastfeeding ability when evaluating different surgical techniques; however, they do reinforce the importance of preserving areolar sensibility, because the neurohormonal reflex during breastfeeding starts with sensory stimulation of the areola6.
A study by Marshall et al.7 emphasized that the most important thing in preserving breastfeeding ability is the amount of mammary tissue that is preserved in connection with the NAC (in the group with thin pedicles, breastfeeding was not impossible, but was always insufficient for a child's full development).
Strömbeck1, with his bilateral horizontal areolar pedicle technique, showed that breastfeeding is always possible after this surgery, although the ability is reduced. Other authors8,9 report the maintenance of breastfeeding after reduction mammoplasties (Pitanguy's technique), without, however, establishing a minimum period for considering this process efficient.
Thus, the purpose of this paper is to demonstrate the technique described by Longacre10,11, which is performed in this study with a small adjustment, that being an areolar flap of an increased superior pedicle that preserves intact gland tissue extending to the NAC, similar to the mammoplasty with safety pedicle type II described by Ribeiro12. This study also aims to emphasize the need for preoperative orientation as to the potential interference with breastfeeding that may be caused by tissue breakdown after surgery.
METHODS
Case Selection
We performed 46 interventions (for a total of 92 breasts) with a modification of the mastopexy technique as described by Longacre during the period from February 2006 to July 2011. The patients ranged in age from 24 to 42 years.
All patients were evaluated during the preoperative period with imaging exams (breast ultrasound and/or mammography), laboratory tests (hemogram, coagulogram, blood glucose, urea, creatinine, and urine summary), cardiac evaluation tests, and preanesthesia evaluation.
Skin marking
In the Longacre10,11 technique, a crescent-shaped ellipsis is marked from the inframammary fold, using the areola as a limit, and continues to the area of de-epithelialization, with release of the dermoglandular flap and its rolling rotation to the retro-gland area, and without release of the NAC; this results in a scar located at the inframammary fold.
For the patients in this study, skin marking of areas A, B, and C was added similar to that described by Pitanguy4 and Ribeiro12, where the A area represents the projection of the inframammary fold and the B and C areas are obtained after cross-clamping, provided that these latter areas are projected on the ellipsis described by Longacre (Figure 1).
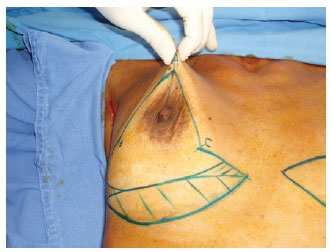
Figure 1 - Marking using the Longacre technique plus areas A, B, and C proposed in this paper.
Surgical technique
With the patient in a semi-sitting position of 45 degrees and with the arms abducted, sedation was administered along with epidural anesthesia or intercostal block (prepared by the surgeon with lidocaine 0.4% + epinephrine 1:100,000) (Figure 2), in joint decision with the anesthetist.
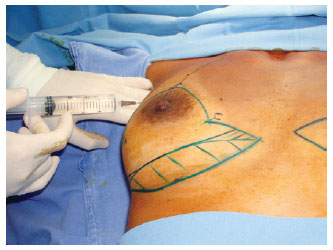
Figure 2 - Intercostal anesthetic block.
The areas of incisions and detachments were infiltrated, with epinephrine solution 1:200.000 + lidocaine 0.2%, and normal saline solution (Figure 3).
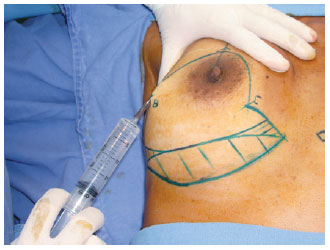
Figure 3 - Infiltration of the incision lines with epinephrine.
Prophylaxis with antibiotics was established, starting with 1 gram of cefazolin (Kefazol) during the anesthetic induction and continuing with 1 gram every 6 hours for the first 24 hours. The Schwarzmann maneuver was then performed, extending the de-epithelialization for all areas previously marked. Glandular tissue was cleared off the pectoral muscle fascia, in the caudocranial direction up to the level of the third rib (Figures 4 to 6), and the excess at the ends of the dermoglandular flap was excised. After hemostasis was achieved, the flap was "somersaulted" (Figures 7 and 8), without fixation to the pectoral fascia and with closure of the lateral and medial pillars, using 3-0 mononylon thread (Figure 9). Only then were the breasts reassembled, with repositioning of the NAC as close as possible to the "A" area previously delimited (Figures 10 and 11).
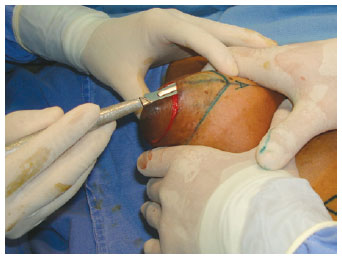
Figure 4 - Schwarzmann maneuver.
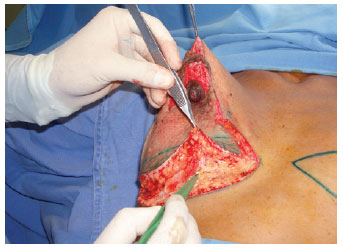
Figure 5 - Glandular clearance.
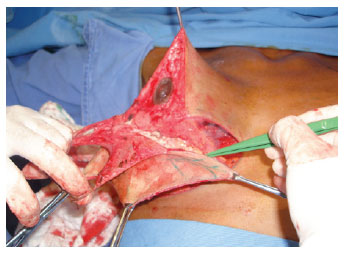
Figure 6 - De-epithelialization.
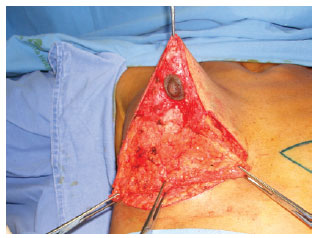
Figure 7 - Definition of the higher base flap.
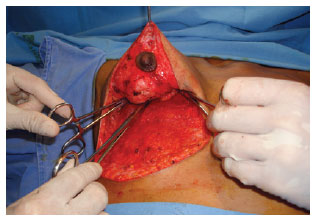
Figure 8 - Flap in somersault.
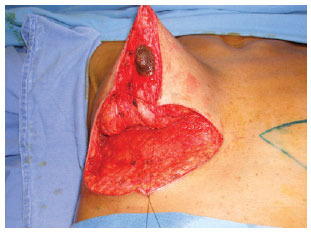
Figure 9 - Suturing of areas B and C.
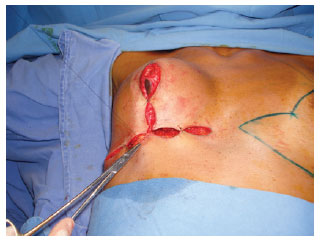
Figure 10 - Suturing of the deep layers to eliminate the dead space.
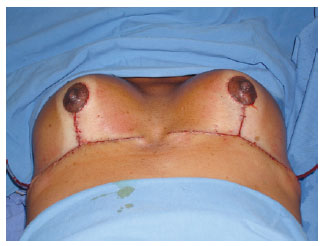
Figure 11 - Final aspect of the breasts.
In this technique, the "A" area is only a reference for the new areolar positioning. It is not static, as the elevation of the NAC up to this level will depend on the new breast shape that is achieved after the resections and assembly of the flaps. Dermocutaneous sutures were placed using 5-0 poliglecaprone (Monocryl) and tube drainage was maintained for 24 hours.
Micropore tape was used over the incisions and a compression garment was placed on the patient. The first dressing change and the removal of the drain were done after 24 hours. The subsequent reviews took place at the surgeon's office, according to the Brazilian Society of Plastic Surgery's recommendations13. Lymphatic drainage was performed on the third postoperative day.
Graduated compression stockings and intermittent pneumatic compression boots (Phlebo Press) were used routinely for antithrombotic prophylaxis.
RESULTS
All 92 breasts on which we operated showed proper filling of the upper pole by the flap, as well as a shape that met the expectations of the patients.
As complications, there were 5 cases of partial dehiscence in the horizontal scar at the union of areas B and C, which were treated conservatively and healed by second intention with a good result. There were 6 cases of seroma, all located just above the horizontal scar. There were no cases of keloid, hematoma, or wound infection.
The patients' subjective evaluation of postoperative breast shape was positive (Figures 12 and 13).
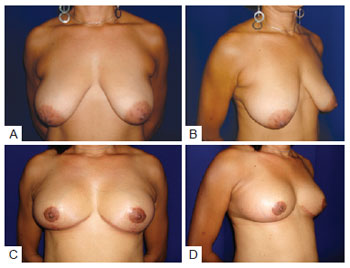
Figure 12 - Patient 1. In A, Preoperative view, frontal position. In B, Preoperative view, oblique position. In C, Postoperative view at 6 months, frontal position. In D, Postoperative view at 6 months, oblique position.
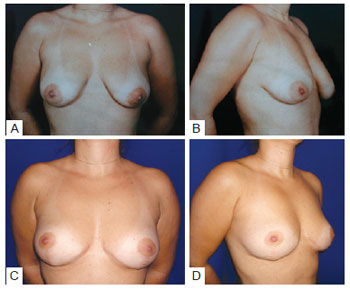
Figure 13 - Patient 2. In A, Preoperative view, frontal position. In B, Preoperative view, oblique position. In C, Postoperative view at 1½ years, frontal position. In D, Postoperative view at 1½ years, oblique position.
One patient presented with moderate galactorrhea during the third postoperative week, which we attributed to the stimulation caused by the postoperative lymphatic drainage. She was treated with cabergoline (Dostinex), to inhibit prolactin secretion.
DISCUSSION
Surely, shape and harmony are essential in the success of mammoplasties. However, the functional preservation of breast sensitivity and the ability to lactate must always be in the mind of the plastic surgeon, as breastfeeding is a natural desire of most women. Guidance is required, especially for the younger patients, with respect to the possible implications of breast surgery for their future plans to breastfeed. Thus, we should be concerned about preserving the anatomical continuity between the mammary glands and their lactiferous ducts through the maintenance of a thick glandular pedicle; the mammary incisions do not prevent lactation, but the resultant scarring makes it difficult for the milk to drain up to the nipple.
The patient must be aware that, under normal conditions, lactation will occur after mammoplasty, but the efficiency of breastfeeding may be compromised. This attitude is more honest and is likely to avoid future disturbance that could occur as a result of misinformation.
Studies show that up to about 18% of primiparous women with no history of breast surgery have difficulty breastfeeding14,15. Could it be possible that the breastfeeding deficit in operated patients is not already present in these women? The answer is that the existing conditions of agalactia or hypogalactia would present in the same way in similar study groups in all other aspects and it is therefore difficult to distinguish agalactia or hypogalactia due to surgery from that in the absence of surgery.
The abovementioned technique described by Longacre10,11 (who, was one of Ivo Pitanguy's16 supervisors early in his career as a surgeon in the 1950s), has the merit of preserving the lactiferous anatomy through a dermoglandular flap with a higher base that remains in continuity with the NAC, thus granting excellent vascularization. The author of the technique reports the lack of signs of absorption or necrosis of the flaps, which is also corroborated by Ribeiro's work12 as the type II "safety pedicle". The association with the marking of the areas A, B, and C of Pitanguy's technique has as a limitation the fact that area A cannot be positioned more than 2 to 3 centimeters off the NAC, as its mobilization is more restricted than that used in other types of pedicles. During the case selection, the author realized that seromas could be avoided through dermal-fat flap areas of approach, filling the dead space resulting from the somersault rotation of the flap (Figure 10).
CONCLUSIONS
With the results obtained by the modified use of Longacre's technique, it can be concluded that:
While the breastfeeding indexes of the patients subject to mammoplasties are generally lower than those of women who have not had breast surgery, there is less of a deficit when using this technique that is based on the anatomical preservation of the lactiferous connections. The Longacre technique is based on simple principles of anatomical preservation; however, these restrict the indication for the procedure to cases with little ptosis and breast hypertrophy, so that there is proper elevation of the NAC. As for all mammoplasty techniques, a thorough explanation of the possible repercussions on breastfeeding must be given to patients, so as to avoid future demands or frustrations during maternity. The technique presented constitutes another technical possibility for mastopexies in patients who have the NAC in a high position and uses the lower pole of the breast to fill out the upper pole.
REFERENCES
1. Strömbeck JO. Late results after reduction mammaplasty. In: Goldwyn RM, ed. Long term results in plastic and reconstructive surgery. Boston: Little, Brown; 1980. p. 723-32.
2. Skoog T. A technique of breast reduction: transposition of the nipple on a cutaneous vascular pedicle. Acta Chir Scand. 1963;126:453-65.
3. McKissock PK. Reduction mammaplasty with a vertical dermal flap. Plast Reconstr Surg. 1972;49(3):245-52.
4. Pitanguy I. Mamaplastias: estudo de 245 casos consecutivos e apresentação de técnica pessoal. Rev Bras Cir. 1961;42(4):201-20.
5. Thorek M. Plastic surgery of the breast and abdominal wall. Springfield: Charles C. Thomas; 1942.
6. Deutinger M, Deutinger J. Breast feeding after aesthetic mammary operations and cardiac operations through horizontal submammary skin incision. Surg Gynecol Obstet. 1993;176(3):267-70.
7. Marshall DR, Callan PP, Nicholson W. Breastfeeding after reduction mammaplasty. Br J Plast Surg. 1994;47(3):167-9.
8. Aboudib Junior JH, Coelho RS, Castro CC, Cupello AM. Mamoplastia redutora: alterações provocadas pela gravidez. J Bras Ginecol. 1989; 99(9):395-8.
9. Castro CC, Salema R, Aboudib Junior JHC, Calixto CA. Mamaplastia redutora na mulher jovem: análise crítica e evolutiva a longo prazo. J Bras Ginecol. 1986;96(9):471-3.
10. Longacre JJ, De Stefano GA, Holmstrand K. Breast reconstruction with local derma and fat pedicle flaps. Plast Reconstr Surg. 1959;24:563-76.
11. Longacre JJ. Correction of the hypoplastic breast with special reference to reconstruction of the "nipple type breast" with local dermo-fat pedicle flaps. Plast Reconstr Surg. 1954;14(6):431-41.
12. Ribeiro L. Pedículos em mamoplastia: atlas e texto. Rio de Janeiro: Guanabara Koogan; 2005. p. 49-62.
13. Sociedade Brasileira de Cirurgia Plástica. Orientações pré e pós-operatórias. Disponível em: http://www.cirurgiaplastica.org.br.
14. Jaeger MR, Barcellos CH, Terres MM, Ely PB, Ely JF. Aleitamento materno após a cirurgia plástica redutora da mama. ACM Arq Catarin Med. 2000;29(Supl. 1):275-7.
15. Jaeger MR, Terres M, Souza EP, Ely PB. É possível o aleitamento materno após a cirurgia plástica redutora da mama? Rev Bras Mastol. 1998;8(4):185-8.
16. Pitanguy I. Aprendiz do tempo: histórias vividas. Rio de Janeiro: Nova Fronteira; 2007. p. 66-8.
1. Full member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery) - SBCP, plastic surgeon, Recife, PE, Brazil.
2. Full member of SBCP, teacher of residency in plastic surgery at Hospital Agamenon Magalhães and Member of SBCP's Award Commission, Recife, PE, Brazil.
Luiz Felipe Duarte Fernandes Vieira
Av. Boa Viagem, 296 - apto. 1004 - Boa Viagem
Recife, PE, Brazil - CEP 51011-000
E-mail: luizfelipedfv@uol.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: November 17, 2011
Article accepted: March 7, 2012
Study conducted at the Hospital Agamenon Magalhães, Recife, PE, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter